Androgen Replacement Therapy Market is estimated to be valued at USD 2,857.8 Mn in 2026 and is expected to reach USD 4,897.7 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 8.0% from 2026 to 2033.
Analysts’ Views on Global Androgen Replacement Therapy Market :
Increasing research and development activities are expected to boost the growth of the global androgen replacement therapy market over the forecast period. For instance, according to a study published by Cleaveland Clinic, a non-profit, multispecialty academic medical center, on June 16, 2023, in men with low testosterone, testosterone-replacement therapy does not increase incidence of major adverse cardiac events.
Figure 1. Global Androgen Replacement Therapy Market Share (%), By Distribution Channel, 2026
To learn more about this report, Request Free Sample
Global Androgen Replacement Therapy Market– Drivers
The increasing prevalence of hypogonadism
The growing prevalence of hypogonadism in adult men worldwide is major factor leading to the high demand growth of androgen replacement therapy market. For instance, according to the data published in January 2021, in PubMed, a free search engine for biological databases and medical reports, the estimated prevalence of hypogonadism varies between 2.1% and 38.7% in middle-aged and older men, but can increase to approximately 50% among individuals with diabetes or obesity globally.
Introduction of newer products in the market
The increasing introduction of newer products can drive the growth of the global androgen replacement therapy market. For instance, in June 2022, Halozyme Therapeutics, Inc., a U.S.-based biotechnology company, announced the commercial launch of TLANDO (testosterone undecanoate), an oral treatment indicated for testosterone replacement therapy in adult males for conditions associated with a deficiency or absence of testosterone (primary or hypogonadotropic hypogonadism).
Figure 2. Global Androgen Replacement Therapy Market Share (%), By Region, 2026
To learn more about this report, Request Free Sample
Global Androgen Replacement Therapy Market- Regional Analysis
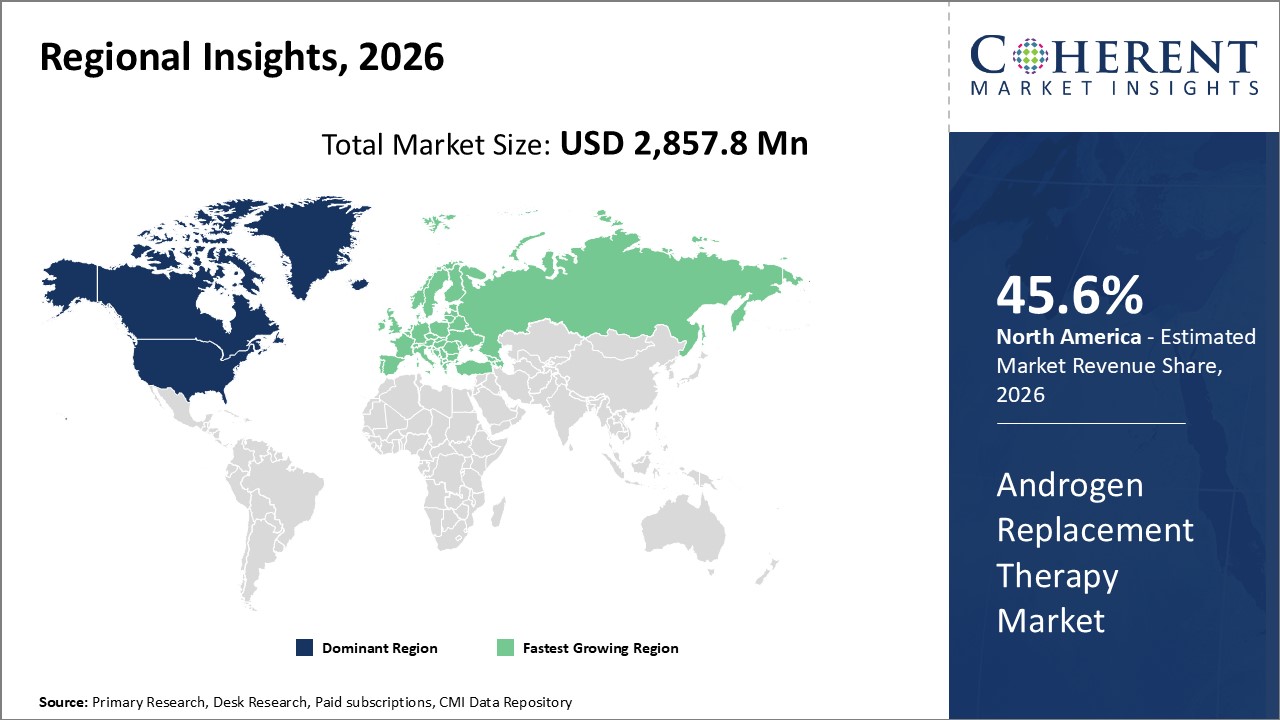
Among regions, North America is estimated to hold a dominant position in the global androgen replacement therapy market over the forecast period. This is due to the increasing number of product launches in the region. For instance, on March 2026, the U.S. Food And Drug Administartion (FDA) approved testosterone undecanoate capsules called (Tlando), an oral testosterone replacement therapy in the U.S. hypogonadism market. The approval was given to Antares Pharma, a U.S-based specialty pharmaceutical company.
Since the virus outbreak in December 2019, the disease spread to over 100 countries across the globe, and the World Health Organization declared it a public health emergency on January 30, 2020.
Androgen Replacement Therapy Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2,857.8 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.0% | 2033 Value Projection: | USD 4,897.7 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AbbVie, Inc., Bayer AG, Endo International plc, Lilly, Kyowa Kirin Co., Ltd, Novartis AG, Pfizer Inc, Clarus Therapeutics, Perrigo Company plc, Acerus Pharmaceuticals Corporation, Sawai Pharmaceutical Co.,Ltd, Dr. Reddy’s Laboratories Ltd, Sun Pharmaceutical Industries Ltd., and Simple Pharma. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Androgen Replacement Therapy Market Segmentation:
The global androgen replacement therapy market report is segmented into product type, active ingredient, distribution channel, and region.
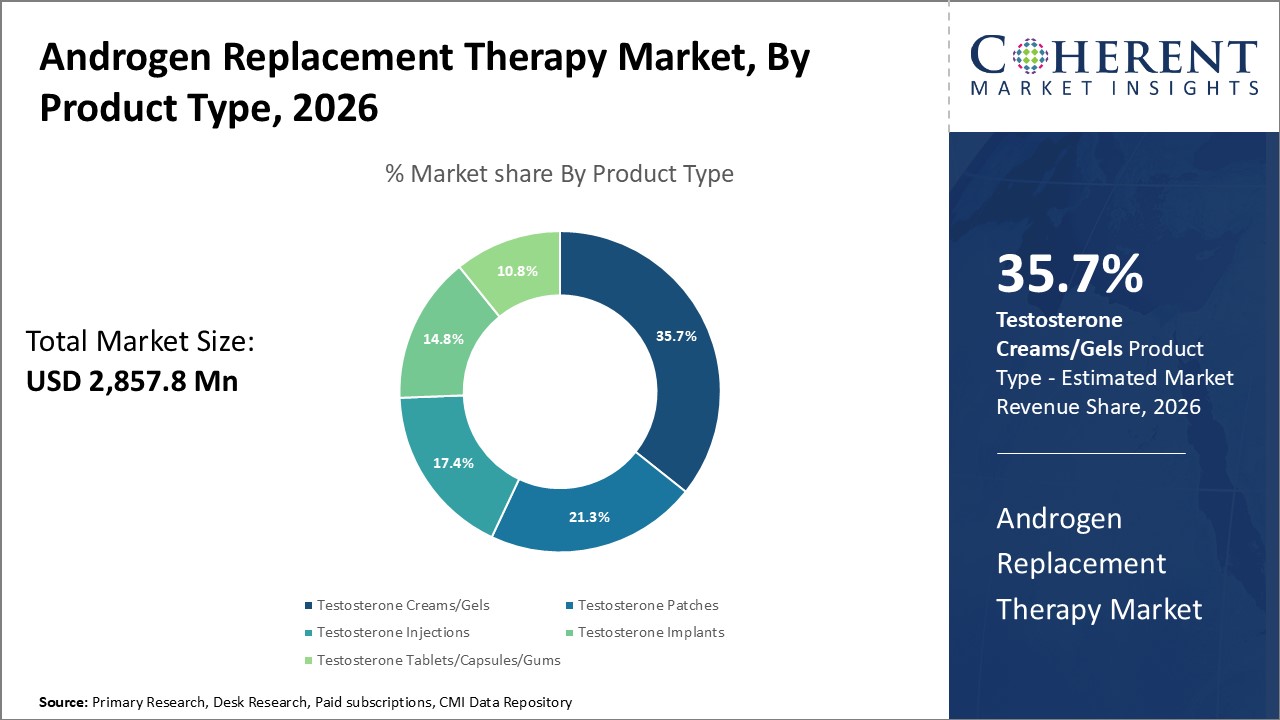
By Product Type, the market is segmented into testosterone creams/gels, testosterone patches, testosterone injections, testosterone implants, and testosterone tablets/capsules/gums. Out of which, the Testosterone Creams/Gels segment is expected to hold a dominant position in the global androgen replacement therapy market during the forecast period and this is attributed to the higher safety of this dosage form in comparison to others.
By Active Ingredient, the market is segmented into testosterone, methyl testosterone, testosterone undecanoate, testosterone enanthate, and testosterone cypionate. Out of which, the testosterone undecanoate segment is expected to hold a dominant position in the global androgen replacement therapy market during the forecast period and this is attributed to a good profile of efficacy and safety of the drug.
By Distribution Channel, the market is segmented into hospital pharmacies, retail pharmacies, and online pharmacies . Out of which, the Hospital Pharmacies segment is expected to dominate the market over the forecast period and this is attributed to the rising burden of hypogonadism in men globally and consequent hospitalization.
By Region, the market is segmented into North America, Latin America, Europe, Asia Pacific, Middle East, and Africa. Out of which, the North America is expected to dominate the market over the forecast period and this is attributed to the growing number of cases of hypogonadism in men in this region.
Among all the segmentations, the product type segment has the highest potential due to the increasing partnerships and collaborations across the world over the forecast period. For instance, on April 4, 2023, PeterMD, a telemedicine provider in North America focusing on Testosterone Replacement Therapy and Male Hormone Optimization Therapy, announced a partnership with The Beverly Hills Anti-aging Center for Men and Women & Advanced Nutrition and Hormone-Based Gynecology, a U.S.-based healthcare provider, to launch PetraMD, female focused Hormone Replacement Therapy telemedicine clinic which will manufacture hormone gels and creams.
Global Androgen Replacement Therapy Market Cross Sectional Analysis:
The Testosterone Creams/Gel segment is estimated to dominate the global androgen replacement therapy market in North America region during forecast period. For instance, in March 2022, Clarus Therapeutics, a U.S.-based pharmaceuticals company dedicated to providing resources and support for men affected by the health-eroding symptoms of hypogonadism, launched JATENZO, an oral softgel as Testosterone Replacement Therapy (TRT) in adult hypogonadal males who have low or no testosterone due to certain medical conditions.
Global Androgen Replacement Therapy Market: Key Developments
On June 1, 2023, Getlabs, Inc., a provider for at-home diagnostic collections, partnered with Fountain TRT, a provider of testosterone replacement therapy (TRT) for men, to offer convenient at-home hormone testing and treatment.
On April 3, 2023, the U.K. Department of Health and Social Care, announced the launch of a new scheme to expand access to cheaper Hormone Replacement Therapy (HRT) drugs. This initiative was introduced to cut cost burden for hormone replacement therapies in the U.K.
On May 10, 2023, FarmaKeio Pharmacy Network, a pharmaceutical outsourcing facility in the U.S., was granted a patent to produce testosterone and triamcinolone subcutaneous hormone pellets. The drug was introduced to provide a sustained release of testosterone and triamcinolone, allowing patients to enjoy the benefits of these hormones for extended periods of time.
Global Androgen Replacement Therapy Market: Key Trends
The launch of novel drug therapies and treatments can provide lucrative opportunities to the global androgen replacement therapy market. For instance, in August 2022, GAT WHP announced the launch of its new testosterone optimization product, TESTROL PLATINUM with a new formula that has ingredients shown to increase total testosterone by up to 132%.
Increasing research and development efforts are expected to boost the growth of global androgen replacement therapy market. According to an article published in Urology Times, a resource for urologists & allied health professionals offering clinical analysis, policy perspectives, & practical advice, in June 2020, Tlando, a novel lymphatically delivered oral testosterone undecanoate shows liver health benefits with 40% reduction in liver fat at 4 months.
Global Androgen Replacement Therapy Market: Restraint
Product Recalls
The growth of the androgen replacement therapy market may be hampered, owing to the increasing product recalls of testosterone products in the market. For instance, in July 2022, the U.S. Food and Drug Administration (USFDA) enforced the recall of 50,680 vials of Testosterone Cypionate Injection, used to treat low testosterone in adult males produced by Sun Pharmaceutical Industries Ltd., a multinational pharmaceutical company headquartered in Mumbai, India.
This restrain can be overcome by strict compliance with the regulatory guidelines and introduction of Good Manufacturing Practices (GMP) in production.
Risk of prostate cancer
The side-effects associated with androgen replacement therapy are restraining the growth of this market. For instance, according the data shared by researchers at the Central European University, a private research university, in June 2022, there is a positive associations between high levels of free testosterone and increased risks of prostate cancer.
This can be overcome by taking proper dosages of testosterone which would not be toxic to the human system.
Global Androgen Replacement Therapy Market- Key Players
Major players operating in the global androgen replacement therapy market include include AbbVie, Inc., Bayer AG, Endo International plc, Lilly, Kyowa Kirin Co., Ltd, Novartis AG, Pfizer Inc, Clarus Therapeutics, Perrigo Company plc, Acerus Pharmaceuticals Corporation, Sawai Pharmaceutical Co.,Ltd, Dr. Reddy’s Laboratories Ltd, Sun Pharmaceutical Industries Ltd., and Simple Pharma.
Definition: Androgen replacement treatment (ART), also known as testosterone replacement therapy (TRT), is a type of hormone therapy that supplements or replaces androgens, most often testosterone. It generally involves the administration of testosterone therapy by injections, skin creams, patches, gels, tablets, or subcutaneous pellets. ART is frequently given to treat male hypogonadism.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients