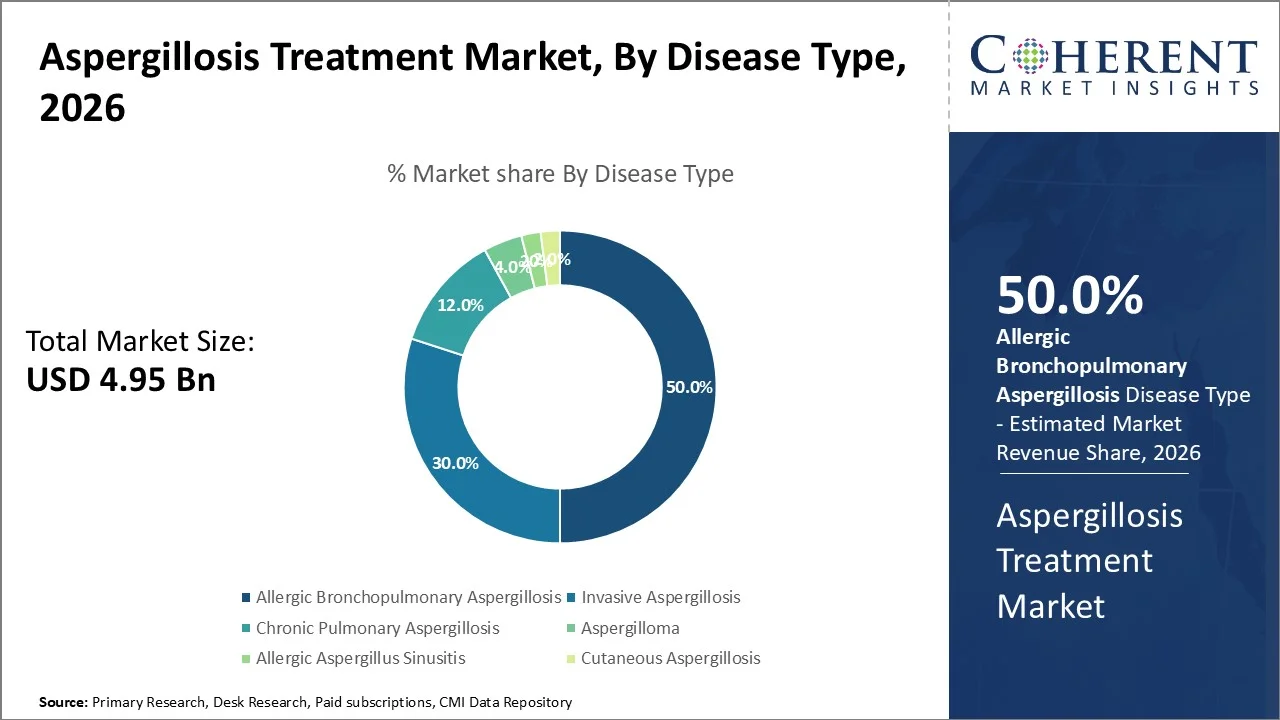
The Aspergillosis Treatment Market is projected to witness a CAGR of 4%, with a market value of USD 4.95 Bn in 2026 and a forecast of USD 6.51 Bn in 2033. Factors contributing to the growth of the market include an increase in the prevalence of fungal infections, a rise in the number of immunocompromised patients, and advancements in antifungal medications.
The anti-fungal segment is expected to hold the highest market share of the aspergillosis treatment market in 2026, accounting for nearly 80% of the total share of drugs used for the condition. The dominance of the segment can be credited to it being the primary and preferred method of treatment for all types of aspergillosis infections, including invasive, chronic, and allergic forms.
The high dependence on antifungal therapy is further supported by the growing incidence of invasive fungal infections in immunocompromised populations, where prompt antifungal intervention is critical for patient survival.
In June 2024, F2G announced FDA accepted the New Drug Application (NDA) for review Olorofim for the treatment of invasive fungal infections, including aspergillosis.
To learn more about this report, Request Free Sample
The allergic bronchopulmonary aspergillosis segment (containing ABPA) is expected to dominate the market, representing more than 50% of the market's total share by 2026. This is due to the high prevalence of allergic bronchopulmonary aspergillosis compared to other types of aspergilloses like invasive and cutaneous aspergillosis. The ABPA form of aspergillosis is mainly prevalent among patients with asthma and cystic
According to the World Health Organization (WHO), asthma alone affects more than 260 million people worldwide, creating a large at-risk population for allergic fungal complications, including aspergillosis-related conditions.
The rise in immunocompromised patients is another important factor that increases the requirement for aspergillosis management worldwide. Patients who are under chemotherapy, organ transplant or corticosteroids therapy have increased chances of developing invasive fungal infections. This factor necessitates the development of antifungal drugs and diagnostic tools for managing aspergillosis.
Additionally, clinical research shows that aspergillosis occurs in up to 2–26% of hematopoietic stem cell transplant patients and 1–15% of solid organ transplant recipients, demonstrating the high infection risk in immunocompromised populations.
Increasing cases of fungal infections worldwide have played an important role in driving the growth of the market for treatments for aspergillosis. Increased admissions, healthcare expenses, and outpatient visits due to fungal infections have increased the need for efficient antifungal treatments. Hospitals and other organizations involved in healthcare are focusing on early diagnosis and treatment of these infections to lower mortality rates and financial expenses.
According to the World Health Organization, chronic respiratory diseases affect over 300 million people globally, creating a large at-risk population for complications such as fungal infections including aspergillosis.
|
Current Event |
Description and its Impact |
|
Rising Antifungal Drug Resistance (Global Health Concern 2025–2026) |
|
|
Growing Transplant and Cancer Treatment Volumes Globally |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
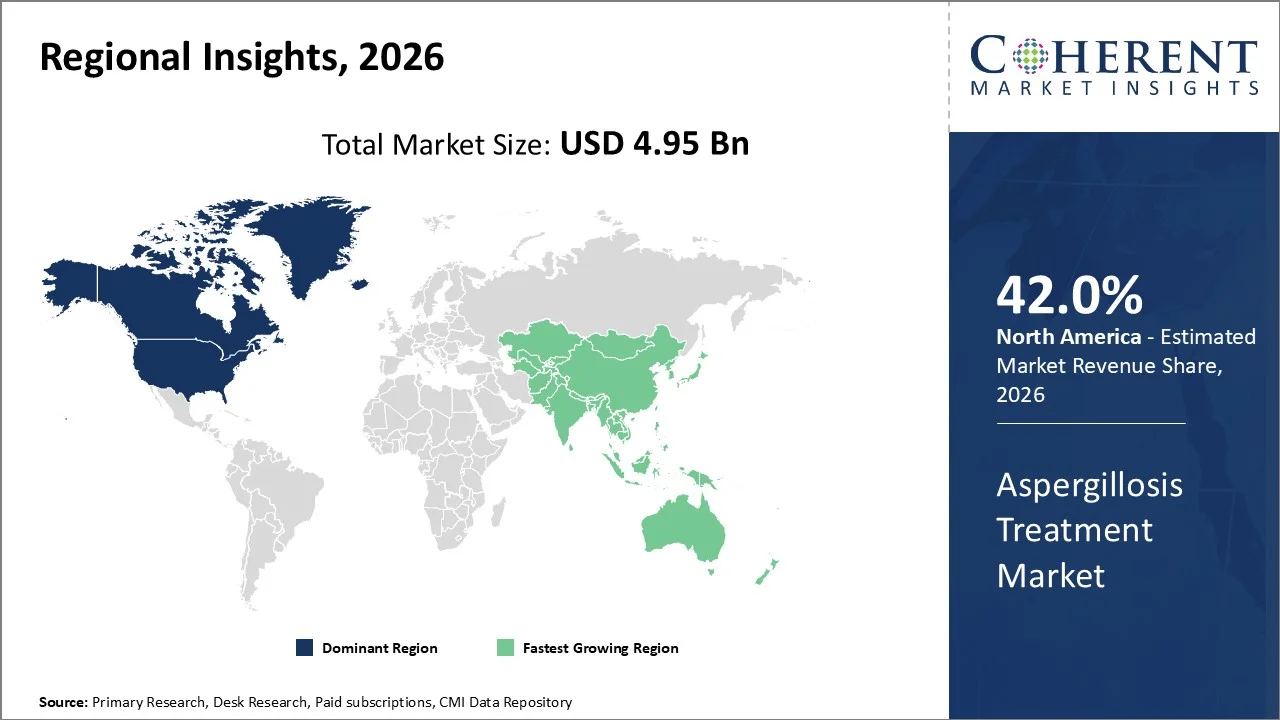
North America is estimated to hold the largest market share of around 42% in 2026 owing to advanced healthcare facilities, awareness, early diagnosis, and availability of antifungals in the region. Additionally, North America has a high number of immunocompromised people, such as cancer patients, transplant recipients, and ICU patients, resulting in high demand for aspergillosis drugs in this region.
In September 2024, F2G announced continued advancement of its Phase III clinical program for Olorofim, a novel antifungal therapy targeting invasive aspergillosis, with strong focus on U.S. regulatory submission and commercialization planning.
The Asia Pacific region is expected to be the fastest-growing market for antifungals between 2026 and 2033 owing to the high prevalence of respiratory diseases, high number of hospitalizations, and better diagnosis methods. The rapid growth of healthcare facilities in nations such as China and India will boost antifungal consumption.
According to the World Health Organization (WHO), chronic respiratory diseases affect hundreds of millions of people in the Asia Pacific region, creating a large at-risk population for fungal complications such as aspergillosis.
The U.S. aspergillosis treatment market is expected to witness sustained growth over the forecast period. Such growth will be attributable mainly to the growing number of immunosuppressed patients, the growing number of fungal infections acquired in hospitals, and an abundance of antifungal drugs. Other factors include early diagnosis facilities and the adoption of precise diagnostic technologies, such as PCR-based fungal detection and biomarkers.
In January 2025, the U.S. Food and Drug Administration (FDA) finalized updated antifungal susceptibility breakpoints for azole drugs such as voriconazole and isavuconazole used in invasive aspergillosis treatment.
China is expected to remain a key and rapidly growing market for aspergillosis therapy in the forecast period. The growth will be supported by the increase in the prevalence of chronic respiratory diseases, the rise in hospitalization rates, advancements in diagnostics, and the availability of antifungal drugs.
In July 2025, China’s National Medical Products Administration (NMPA) reported that it had approved a record number of new drugs during 2025, totaling 120 first-time marketing authorizations, including multiple anti-infective and antifungal therapies supporting the treatment of invasive fungal diseases such as aspergillosis
Some of the major key players in Aspergillosis Treatment are Abbott laboratories, AstraZeneca plc, Eli Lilly and Company, GlaxoSmithKline, Johnson & Johnson Services, Inc., Mylan Pharmaceuticals, Inc., Novartis AG, Pfizer, Inc., and Takeda Pharmaceutical Company Ltd.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4.95 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4% | 2033 Value Projection: | USD 6.51 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Abbott laboratories, AstraZeneca plc, Eli Lilly and Company, GlaxoSmithKline, Johnson & Johnson Services, Inc., Mylan Pharmaceuticals, Inc., Novartis AG, Pfizer, Inc., and Takeda Pharmaceutical Company Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients