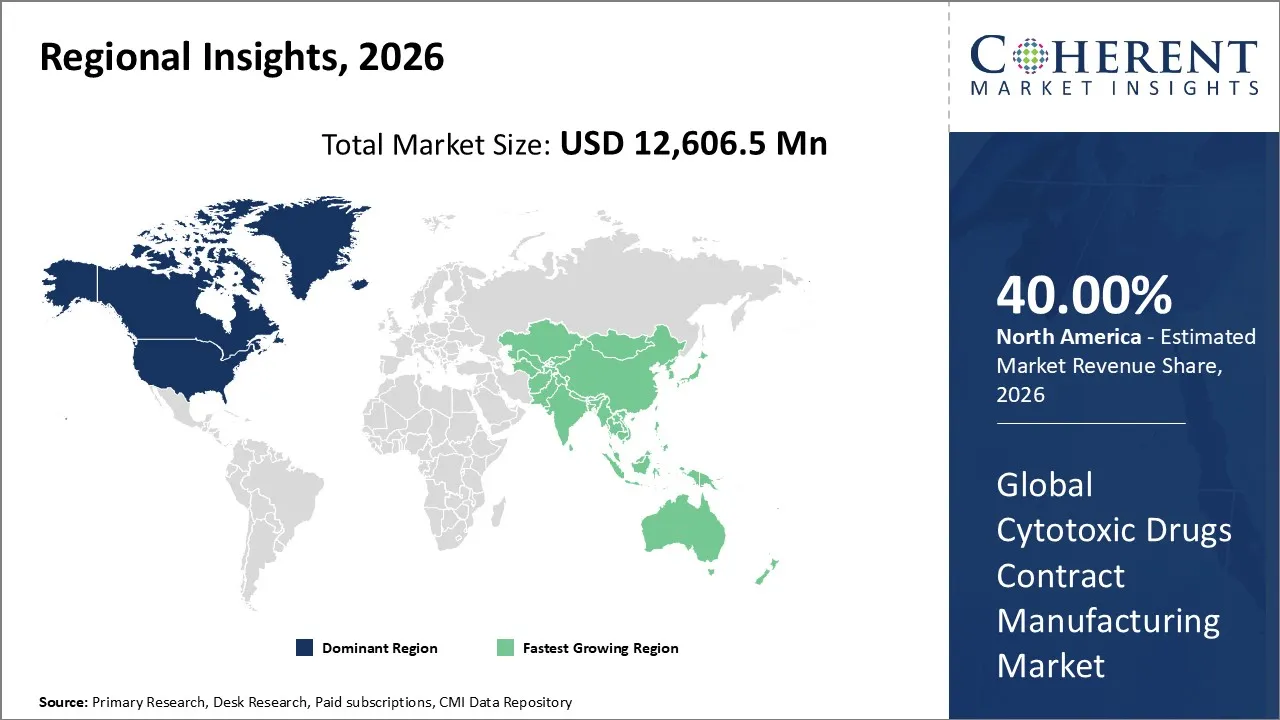
Cytotoxic drugs contract manufacturing market is estimated to be valued at USD 12,606.5 Mn in 2026 and is expected to reach USD 23,157.5 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 9.1% from 2026 to 2033.
The increasing number of cancer cases across the globe is fueling the demand for cytotoxic drugs, thus propelling the market for cytotoxic drugs contract manufacturing. The complexity involved in the manufacturing of cytotoxic drugs, as well as the regulatory requirements, is encouraging pharmaceutical companies to outsource the manufacturing of these drugs to CDMOs (Contract Development and Manufacturing Organizations). The need for injectable and oral chemotherapy drugs, as well as the growing pipeline of oncology drugs, is also fueling the market.
|
Current Events |
and its impact |
|
Regulatory Harmonization and Safety Standards Evolution |
|
|
Consolidation and Strategic Partnerships in Contract Manufacturing |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Based on production scale, the industrial scale segment is expected to account for the highest market share of 60% in 2026, due to its capability of dealing with large-scale commercial production of cytotoxic drugs. Industrial-scale facilities are better for productivity, quality control, and cost savings than pilot or lab scales. This is why pharmaceutical companies prefer to outsource their production to these types of facilities.
For instance, in May 2025, Shilpa Biologicals announced the opening of a bioconjugation suite at its facility in Dharwad, India, to facilitate the advanced manufacturing of antibody drug conjugates.
By form, the liquid segment is expected to have the largest market share of 58% in 2026, driven by the popularity of injectable oncology therapies. Liquid formulations, such as sterile injections and IV solutions, are highly important in cancer therapy and require specialized contract manufacturing facilities that can maintain sterility and contain them well.
For instance, in March 2025, LGM Pharma announced a strategic USD6 million investment to expand its CDMO manufacturing capabilities, specifically increasing capacity for liquid, suspension, semi‑solid, and suppository drug products at its Rosenberg, Texas facility.
Based on the type of product, the intravenous segment is expected to hold a 55% market share in 2026, driven by the demand for intravenous chemotherapy. Intravenous products are highly demanding in terms of handling and regulatory requirements, making CDMOs a crucial partner for pharmaceutical companies. The demand for intravenous products is driven by the rising prevalence of cancer, ongoing clinical trials, and the need for hospital-administered products.
For instance, in April 2025, PCI Pharma Services, a global CDMO, announced that it would acquire Ajinomoto Althea, Inc., a US-based sterile fill finish CDMO with expertise in injectable drug products, including prefilled syringes and cartridges, with high potency and oncology-relevant formats.
To learn more about this report, Request Free Sample
North America is anticipated to lead the cytotoxic drugs contract manufacturing market in 2026, with a market share of 40%. The dominance of the North American region can be attributed to factors such as the presence of advanced pharmaceutical infrastructure, a large number of leading CDMOs, a robust pipeline of cancer therapies, and well-established regulatory frameworks.
For instance, in January 2025,Lonza Group announced the extension of its industrial-scale cytotoxic drug manufacturing capacity at its Vacaville, California facility. This extension includes new high-containment suites and aseptic fill-finish lines for injectable and intravenous cancer therapies, further solidifying North America’s position in the global CDMO industry.
The Asia Pacific region is expected to be the fastest-growing market for cytotoxic drug contract manufacturing organizations. This is because manufacturing costs are low, investments in pharmaceutical infrastructure are rising, demand for cancer therapies is rising, and government policies in countries like China, India, and Japan are good for business. New companies in the area are learning how to make cytotoxic drugs and highly potent APIs on a large scale.
For instance, in December 2025, Shilpa Biologicals inaugurated a new bioconjugation and sterile manufacturing facility in Dharwad, India, focused on antibody-drug conjugates and other intravenous cytotoxic formulations.
The U.S. market is currently undergoing substantial growth due to the high number of top-tier CDMOs, advanced industrial-scale facilities, and a robust oncology drug pipeline. The presence of stringent regulatory environments and a well-developed pharmaceutical infrastructure in the US makes it attractive for outsourcing the production of cytotoxic drugs to experienced contract manufacturers.
For instance, in January 2025, Lonza Group expanded its Vacaville, California facility with new high-containment suites and aseptic fill-finish lines for injectable and intravenous oncology drugs.
The growing investments of China in the manufacturing of pharmaceuticals and the favorable government policies are fueling the growth of the cytotoxic drugs CDMO market. The increasing demand for cancer treatments in the domestic market and the competitive manufacturing costs are encouraging pharmaceutical companies to outsource their production.
For instance, in December 2025, WuXi AppTec inaugurated a new sterile manufacturing facility in Suzhou, China, focused on injectable cytotoxic drugs and highly potent APIs. The facility includes advanced aseptic fill-finish lines and containment systems to meet both domestic and global demand.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 12,606.5 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.1% | 2033 Value Projection: | USD 23,157.5 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Lonza Group, Piramal Group, Evonik Industries AG, Novasep Holding SAS, Merck KGaA (SAFC Pharma), Baxter Biopharma Solutions, AbbVie Contract Manufacturing, Cambrex Corporation, BSP Pharmaceuticals S.p.A., CordenPharma Internatisonal, Catalent, Inc., Albany Molecular Research Inc., Evotec, WuXi AppTec Co., Ltd., Pierre Fabre Laboratories, and Dishman Group |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The rising incidence of cancer cases globally is fueling the demand for cytotoxic drugs, thereby propelling the growth of the contract manufacturing market. The complexity and containment involved in the manufacturing of cytotoxic drugs are forcing pharmaceutical companies to outsource their manufacturing to CDMOs. The rising use of intravenous and oral chemotherapy and the development of targeted therapies for cancer are further increasing the demand for contract manufacturing.
The market offers substantial growth opportunities with the development and scaling of highly potent and advanced drug manufacturing facilities. Investment in sterile fill-finish lines, high-containment isolators, and end-to-end development services enables CDMOs to secure pharmaceutical clients from across the globe. Development of capabilities in antibody-drug conjugates (ADCs), intravenous formulations, and other complex cytotoxic products creates a platform to capitalize on the growing demand in the North America, Europe, and Asia-Pacific regions.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients