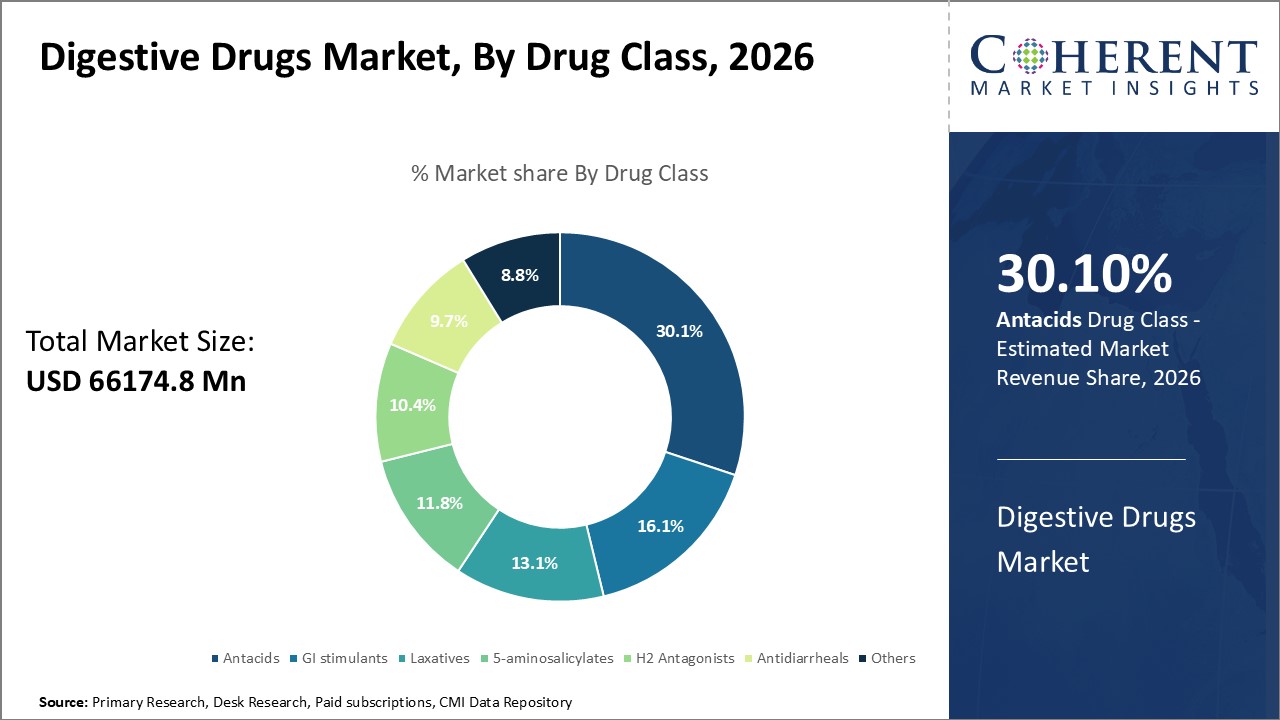
Digestive Drugs Market is estimated to be valued at USD 66,174.8 Mn in 2026 and is expected to reach USD 99,897.3 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 6.1% from 2026 to 2033.
The Digestive Drugs Market is witnessing steady growth, driven by rising prevalence of gastrointestinal disorders, including acid reflux, ulcers, and irritable bowel syndrome. Increasing geriatric population, lifestyle changes, and expanding healthcare access fuel demand. Pharmaceutical companies are investing in novel formulations and combination therapies to improve patient outcomes, boosting the Digestive Drugs Market demand globally.
In terms of type, the generic segment is projected to hold 64.4% share of the market in 2026, due to its cost-effectiveness, growing consumer awareness, and patent expirations of key branded drugs. Generic medications enable wider access to treatments for gastrointestinal disorders like acid reflux, ulcers, and IBS. Their rapid adoption, especially in emerging markets, positions generics as the leading segment globally.
For instance, in August 2026, Dr Reddy's Laboratories introduced Colozo, a generic version of the USFDA-approved drug Linaclotide, to treat chronic idiopathic constipation (CIC), irritable bowel syndrome with constipation (IBS-C) in adults, and functional constipation in children aged 6–17. This launch strengthens the company's gastrointestinal portfolio, following the introduction of BixiBat in India last year.
To learn more about this report, Request Free Sample
In terms of drug class, the antacids segment is projected to account for the highest share of the market in 2026, due to their widespread use in treating GERD, heartburn, and acid-related disorders. Rising prevalence of gastrointestinal conditions, easy over-the-counter availability, and growing consumer awareness drive their demand. Other segments, such as Laxatives and 5-Aminosalicylates, are also growing, showing strong market potential.
For instance, in July 2026, Abbott introduced Digene Insta On The Go in India, a water-free antacid solution offering relief from acidity, heartburn, and indigestion within 2 seconds. Packaged in single-use 10 ml mint-flavoured sachets, it’s priced at ₹50 for a five-sachet pack.
In terms of route of administration, the oral segment is expected to lead the market in 2026, due to its convenience, cost-effectiveness, and widespread patient preference. Oral formulations, including tablets, capsules, and liquids, are extensively used to treat gastrointestinal disorders such as acid reflux, ulcers, and irritable bowel syndrome. Intravenous and other routes hold smaller shares, primarily used in acute or specific treatment scenarios.
For instance, in July 2026, Dr. Reddy’s has struck a non-exclusive patent-licensing deal with Takeda to introduce Vonoprazan tablets, an oral acid-blocker in India under the brand name VONO™. Available in 10 mg and 20 mg doses, the drug aims to treat reflux esophagitis and other acid-peptic conditions, affecting nearly 37-39% of Indians aged 18-59.
In terms of application, the Gastroesophageal Reflux Disease (GERD) segment is projected to hold the biggest share of the market in 2026. This is due to the high prevalence of GERD globally and the availability of effective treatments, such as proton pump inhibitors (PPIs) and H2 receptor antagonists. These factors contribute to GERD's significant market share within the digestive drugs sector.
For instance, in July 2026, the FDA approved vonoprazan (brand name Voquezna) for daily use to relieve heartburn in adults with non-erosive GERD. Clinical trials showed it significantly increases heartburn-free days vs placebo, and its safety profile includes typical side effects like nausea, abdominal pain, and constipation.
In terms of distribution channel, the Retail Pharmacies segment is expected to capture the greatest share of the market in 2026, due to their wide accessibility, convenience, and established role in dispensing medications. Patients prefer retail pharmacies for prescription refills, consultations with pharmacists, and reliable access to digestive drugs, making them the primary distribution channel and a key contributor to the market’s overall demand and growth.
For instance, in March 2026, Eli Lilly launched Mounjaro, its weight-loss and type-2 diabetes injection, in India after receiving approval from the country’s drug regulator. The medicine, administered once weekly, works by slowing digestion and increasing feelings of fullness. It will be available via pharmacies with 2.5 mg and 5 mg doses priced at approximately ₹3,500 and ₹4,375 respectively.
To learn more about this report, Request Free Sample
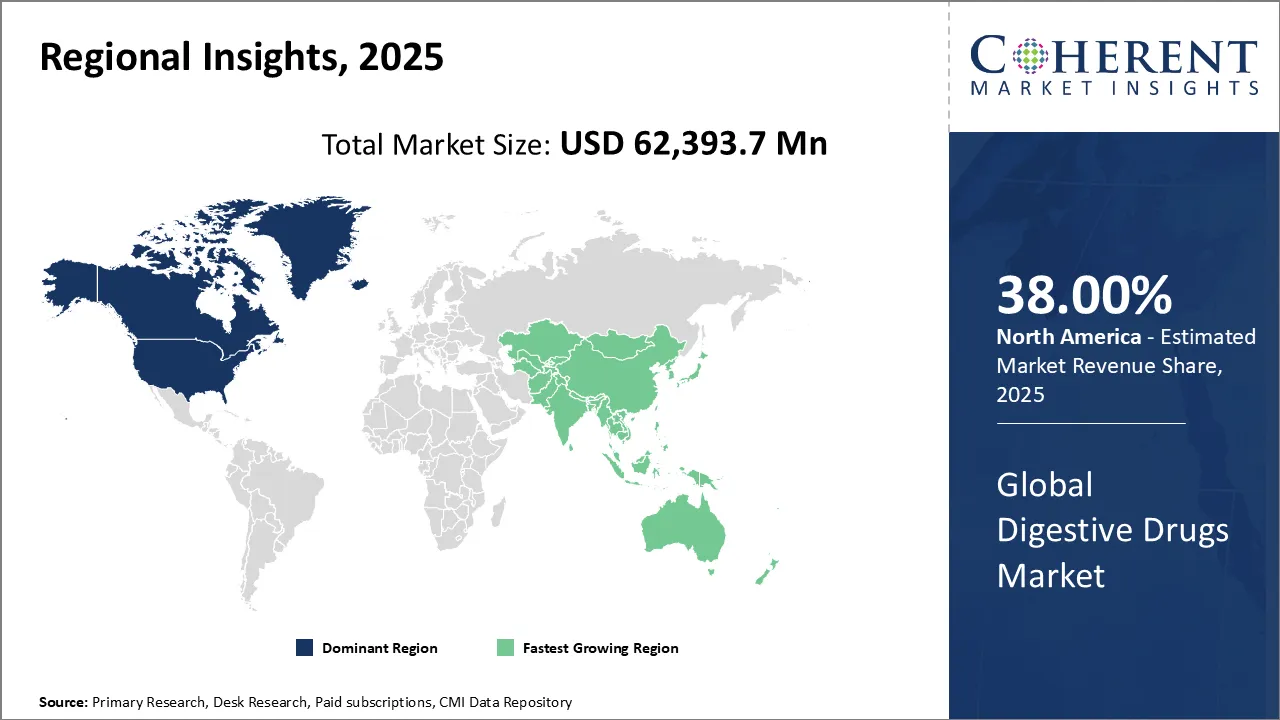
North America dominates the digestive drugs market in 2026, holding about 38% of global share. This leadership is supported by high prevalence of gastrointestinal disorders, advanced healthcare infrastructure, and widespread access to innovative therapies. Strong pharmaceutical R&D, rapid adoption of branded drugs, and favorable reimbursement policies further reinforce North America’s leading position in this market.
For instance, in February 2026, Sandoz launched Pyzchiva® (ustekinumab-ttwe), a biosimilar to Stelara®, in the US, expanding treatment options for around 12 million patients. The drug targets chronic inflammatory conditions such as plaque psoriasis, psoriatic arthritis, Crohn’s disease and ulcerative colitis.
Asia-Pacific is the fastest-growing region in the digestive drugs market, driven by expanding healthcare access, increasing prevalence of digestive diseases, and growing middle-class populations. Rapid pharmaceutical infrastructure development, government-backed healthcare initiatives, and rising demand for cost-effective generics accelerate regional growth. Countries like China and India play a key role in fueling the market’s expansion across Asia-Pacific.
For instance, in June 2024, Akums Drugs & Pharmaceuticals introduced Rabeprazole + Levosulpiride SR Capsules, a novel, DCGI-approved treatment for gastrointestinal disorders in India. The formulation combines rabeprazole (40 mg) and levosulpiride (75 mg) to ease GERD symptoms, improve digestive motor function, and offer faster relief with fewer side effects.
The United States holds a significant share in the digestive drugs market, driven by the high prevalence of gastrointestinal disorders such as GERD, IBS, and ulcers. Advanced healthcare infrastructure, widespread access to both prescription and over-the-counter drugs, and strong adoption of innovative therapies further support demand. Additionally, consumer awareness of digestive health contributes to sustained market growth.
For instance, in March 2026, U.S. FDA approved Tremfya (guselkumab) for adults with moderately to severely active Crohn’s disease. It is the first IL-23 inhibitor in the U.S. to offer both subcutaneous and intravenous induction treatment options. The approval follows Phase 3 trials showing Tremfya’s effectiveness even in patients who failed or were intolerant to conventional or biologic therapies.
China is expected to lead the global digestive drugs market in 2026. This growth is driven by a rapidly expanding middle class, heightened awareness of digestive health, and a rising prevalence of gastrointestinal disorders such as GERD, IBS, and acid reflux. Increasing healthcare access and adoption of both prescription and over-the-counter therapies further support market demand.
For instance, in June 2026, Ecnoglutide, a novel GLP-1 weight-loss drug developed by China's Sciwind Biosciences, has shown promising results in Phase 3 trials. Participants lost up to 15% of their body weight over 48 weeks, with nearly 93% achieving at least a 5% reduction. The drug also demonstrated significant reductions in liver fat.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 66,174.8 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.1% | 2033 Value Projection: | USD 99,897.3 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
GlaxoSmithKline plc, AbbVie Inc. (Allergan), Takeda Pharmaceutical Company Limited, Pfizer Inc, AstraZeneca plc, Marksans Pharma, Tillotts Pharma AG, Salix Pharmaceuticals, Otsuka Pharmaceutical Co., Ltd., Merck & Co., Inc., Lexicon Pharmaceuticals, Inc., Bayer AG, Cadila Healthcare Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Key players in the Digestive Drugs Market are actively expanding their portfolios by obtaining approvals for new digestive drugs, including advanced formulations for GERD, IBS, and other gastrointestinal disorders. These approvals enable companies to meet growing patient demand, enhance therapeutic efficacy, and strengthen their market presence, contributing significantly to the overall Digestive Drugs Market size and revenue growth.
The rising prevalence of gastrointestinal disorders, including GERD, irritable bowel syndrome (IBS), and acid reflux, is a key driver for the Digestive Drugs Market growth. An aging population, changing dietary habits, sedentary lifestyles, and stress contribute to increased digestive health issues. This growing patient base boosts demand for both prescription and over-the-counter digestive medications globally.
Expansion of OTC Digestive Products – Rising consumer awareness about digestive health presents opportunities for over-the-counter (OTC) medications, dietary supplements, and herbal remedies, allowing companies to tap into preventive care and self-medication trends in both developed and emerging markets.
Emerging Markets Growth – Increasing healthcare access, rising disposable incomes, and growing prevalence of gastrointestinal disorders in countries like India, China, and Brazil offer significant opportunities for market expansion and higher adoption of digestive drugs.
The digestive drugs market value is undergoing a significant transformation, driven by a confluence of factors that are reshaping its landscape. A notable trend is the increasing integration of artificial intelligence (AI) in drug development and diagnostics. AI's ability to analyze vast datasets accelerates the identification of novel therapeutic targets and the development of personalized treatment regimens, enhancing the efficacy of digestive disorder interventions.
In the realm of product innovation, the market is witnessing a surge in the development of combination therapies that address multiple gastrointestinal issues simultaneously. For instance, the emergence of dual-action medications that combine proton pump inhibitors with prokinetic agents offers a comprehensive approach to managing conditions like gastroesophageal reflux disease (GERD) and delayed gastric emptying. This innovation not only improves patient compliance but also optimizes treatment outcomes.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients