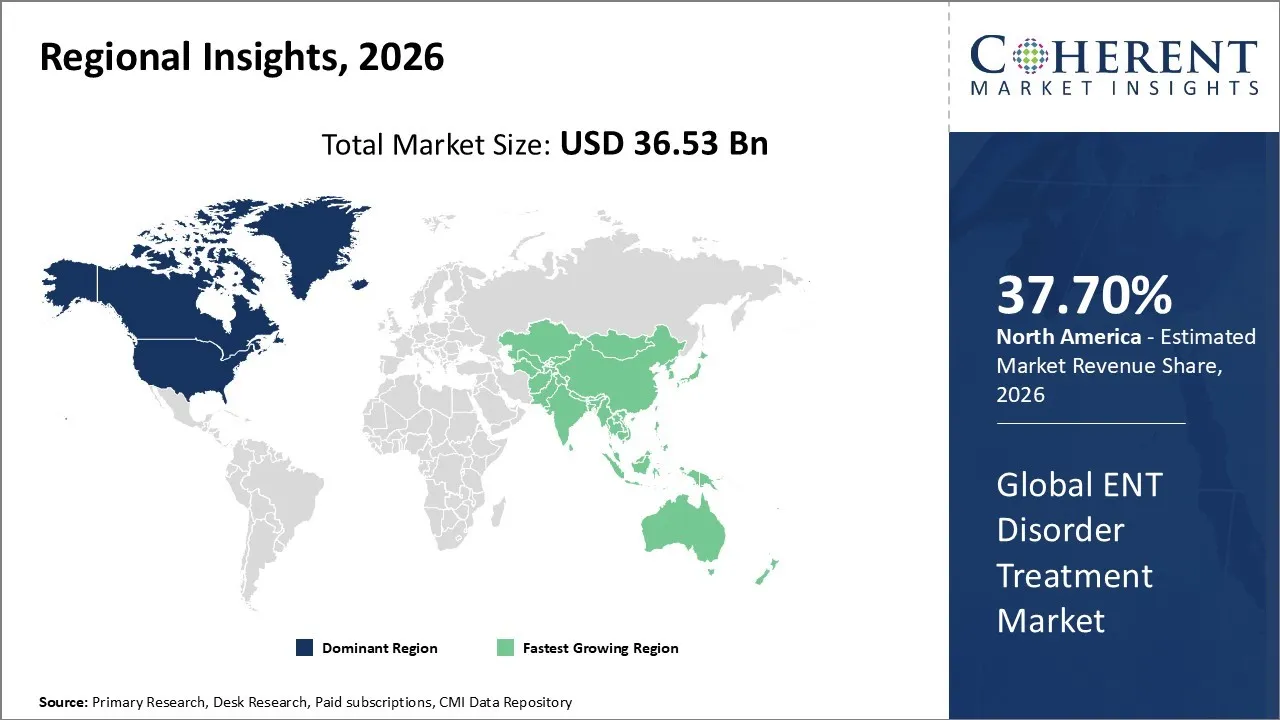
The ENT Disorder Treatment Market is estimated to be valued at USD 36.53 Bn in 2026 and is expected to reach USD 57.30 Bn by 2033, growing at a compound annual growth rate (CAGR) of 6.6% from 2026 to 2033.
The ENT disorder treatment market is advancing significantly driven by the rising global prevalence of chronic sinusitis, otitis media, and age-related hearing loss. The growing patient awareness regarding long-term respiratory health and a strategic shift toward minimally invasive surgeries are expected to propel market growth over the forecast period. The cutting-edge diagnostic and surgical devices have become the cornerstone of modern otolaryngology, moving away from traditional invasive methods toward high-precision, etch-enabled interventions.
|
Current Event |
Description and the Impact |
|
Technological Advancements and Innovations |
|
|
Public Health Trends and Epidemiological Shifts |
|
|
Environmental and Societal Factors |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of treatment type, the drugs segment contributes the highest share of 57.20% in 2026 of the market. The growth is because pharmacological interventions remain the primary and most accessible line of defense for the majority of patients. This segment is driven by the high volume of prescriptions for common ailments such as acute sinusitis, otitis media, and allergic rhinitis, which typically require antibiotics, antihistamines, or corticosteroids rather than surgical intervention. The continual innovation in next-generation antimicrobial drug development is playing a critical role in propelling the market growth amid increasing antibiotic resistance. In addition, the affordability and ease of self-administration of drug regimens ensure their continued preference among both clinicians and patients for the management of acute and chronic ENT conditions.
In terms of organ type, the ears segment contributes the highest share of 38.10% in 2026 of the market. The growth is owing to the massive global prevalence of hearing loss and auditory infections. The World Health Organization estimates that hundreds of millions of people globally require rehabilitation for disabling hearing loss, a figure that is increasing sharply due to the growing geriatric population. This demographic shift fuels a continual demand for both pharmaceutical treatments for conditions like tinnitus and medical devices like hearing aids and cochlear implants. Furthermore, the increasing exposure of younger populations to noise pollution and personal audio devices has created a new, consistent stream of patients seeking auditory care. These factors combine to make ear-related treatments the most significant revenue generator within the ENT specialty.
For instance, in July 2025, the Cochlear Limited, the worldwide pioneer in implantable hearing aids, announced that the Cochlear™ Nucleus® Nexa™ System has received approval from the U.S. Food and Drug Administration (FDA). It is acknowledged as the first and only intelligent cochlear implant system in the world.
In terms of end user, the hospital segment contributes the highest share of 44.60% in 2026 of the market because these institutions serve as the primary centers for complex and specialized ENT care. Hospitals possess the advanced diagnostic infrastructure, such as high-resolution imaging and robotic surgical systems, required for intricate procedures like cochlear implantation and head and neck oncology treatments. While outpatient clinics and ambulatory centers are gaining popularity for minor procedures, the comprehensive care and emergency capabilities of hospitals ensure they manage the highest volume of severe cases. In addition, long-standing reimbursement arrangements systematically favor hospital-based inpatient care, institutionalizing hospitals as the cornerstone of ENT disorder management on a global scale.
For instance, in December 2024, Apollo Hospitals has introduced an Integrated Neuro-ENT Vertigo and Balance Disorders Clinic at Apollo One. This innovative clinic brings together the expertise of neurologists and ENT specialists to provide advanced care for patients who experience vertigo, balance disorders, and a fear of falling.
To learn more about this report, Request Free Sample
North America has remained the dominant region with 37.70% in 2026 of the global ENT disorder treatment market over the past decade. The growth is owing to a mature healthcare infrastructure and high public health investments. The market is marked by a high prevalence of chronic conditions like sinusitis, allergic rhinitis, and otitis media. In addition, a substantial demographic shift toward an aging population is driving sustained demand for treatments related to age-related hearing loss and balance disorders. The technological breakthroughs are pivotal in this region, with a strong emphasis on AI-powered diagnostics, robotic-assisted surgeries, and minimally invasive procedures like balloon sinuplasty that minimize patient recovery times.
The competitive landscape is dominated by established medical technology firms. These companies are placing a greater emphasis on research and development to create innovative devices like next-generation hearing aids and high-definition endoscopes. The leading firms in the region are Medtronic, Stryker, Olympus, and Cochlear Limited. The favorable reimbursement policies and heightened patient awareness regarding early diagnosis and specialized care are also contributing to the region's growth in the market.
For instance, in July 2021, McLaren and Karmanos have introduced a comprehensive ear, nose, and throat (ENT) service line. The service line is designed to offer ENT services to patients in Genesee and Lapeer Counties and beyond.
The Asia Pacific region is recognized as the fastest-growing market for ENT disorder treatment. This growth is largely due to a large population base and an increase in healthcare spending. A rising geriatric population, particularly vulnerable to hearing loss and chronic conditions, primarily drives the rapid expansion. Additionally, increasing urbanization and pollution levels have contributed to a higher incidence of respiratory and allergic disorders, such as sinusitis and allergic rhinitis. A major trend in this market is the use of new technologies, with more and more people wanting procedures that are less invasive. These include robotic-assisted handheld surgical devices and advanced endoscopic systems, which provide shorter recovery times and enhanced precision.
The key drivers at the country level include China and Japan, which together hold a dominant share of the market. Meanwhile, India is expected to experience the highest growth rate owing to its developing healthcare infrastructure and initiatives like Ayushman Bharat. The market leadership is maintained by well-established global companies such as Cochlear Ltd, Olympus Corp, and Medtronic, which are increasingly pursuing strategic alliances and establishing local manufacturing facilities to tap into the substantial volume
The US is currently at the forefront of the ENT disorder treatment market owing to a massive aging population. The chronic conditions like sinusitis create a consistent demand for both pharmacological treatments and minimally invasive surgical options. The market is shifting toward outpatient settings, where procedures like balloon sinuplasty offer faster recovery times and benefit from favorable reimbursement policies provided by Medicare and private insurance. The technological advancements are essential, particularly with the incorporation of artificial intelligence (AI) and robotics in diagnostic endoscopes and surgical navigation systems. The companies like Stryker, Olympus Corporation, and Cochlear Limited are investing in research and development, as well as pursuing strategic acquisitions to strengthen their technological advantage.
For instance, in September 2025, Olympus Corporation has introduced the VISERA™ S OTV-S500 imaging platform in the U.S. This platform combines advanced diagnostic capabilities for ear, nose, and throat (ENT) as well as urology applications, providing flexibility for use in both office and outpatient settings.
China is rapidly emerging as the fastest-growing market for treatments of ENT disorder treatment. The growth is owing to the country’s aging demographic and the health consequences of rapid urbanization. The demand for hearing aids and cochlear implants is surging, thereby driving the ENT device market.
The environmental factors, specifically high levels of air pollution in industrial hubs, have led to a high prevalence of chronic sinusitis and allergic rhinitis. This situation has positioned China as one of the largest consumers of nasal sprays and endoscopic surgical tools worldwide. The market is undergoing significant transformation due to the Healthy China 2030 initiative. This initiative has sped up the modernization of healthcare infrastructure and encouraged the adoption of minimally invasive procedures in tier-2 and tier-3 cities. In addition, National Medical Products Administration (NMPA) has streamlined the approval process for innovative medical devices. This has led to a significant increase in localized AI-integrated diagnostic tools and robotic-assisted ENT platforms that address China's specific clinical volume and patient needs.
According to the National Library of Medicine, by 2060, an estimated 242 million people in China will experience moderate to complete hearing loss. This is increasing the demand for growth in the China ENT Disorder Treatment Market.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 36.53 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.6% | 2033 Value Projection: | USD 57.30 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Sanofi, AstraZeneca, Novartis AG, Pfizer Inc., Mylan N.V., Teva Pharmaceutical Industries Ltd., GlaxoSmithKline Pharmaceuticals, Otonomy Inc., Merck & Co., Dr. Reddy’s Laboratories Ltd., Allergan plc, Cochlear Ltd., Sonova Holding AG, Siemens Healthcare, Starkey Laboratories Inc., William Demant Holding A/S, Widex A/S, GN ReSound A/S, Sonic Innovations Inc., Panasonic Corp., Beltone, Rexton Inc., Avada Hearing Care, Miracle-Ear Inc., MED-EL GmbH, Nuear Hearing Aids Inc., Audiosync Inc., Bernafon, American Hearing Systems Inc., Unitron Hearing Inc., and Zounds Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The increasing size of the global geriatric population is a major factor fueling demand within the ENT disorder treatment market. According to the World Health Organization (WHO), the proportion of the world’s population over 60 years is likely to nearly double by 2050. This demographic shift impacts healthcare because aging naturally triggers degenerative changes in the ear, nose, and throat. The high prevalence of sensory impairments, especially hearing loss, among older adults is accelerating global demand for medical interventions and assistive technologies. In addition to auditory disorders, the aging population exhibits a higher prevalence of chronic conditions such as sleep apnea and laryngeal disorders. Together, these factors are accelerating a market shift toward targeted care pathways and sophisticated diagnostic solutions for the growing aging population.
The ENT Disorder Treatment Market is driven by the high prevalence of ear, nose, and throat conditions and rapid adoption of advanced clinical solutions. Chronic sinusitis affects an estimated 12% of adults globally, while hearing loss impacts a significant portion of the population, particularly in aging demographics. These incidence levels support steady treatment volumes across hospitals, outpatient clinics, and specialized ENT centers.
Demographic trends further reinforce market growth. Increasing geriatric populations experience higher rates of age-related hearing impairment and chronic ENT conditions, creating demand for pharmacological therapies, surgical interventions, and rehabilitative devices. Expanding healthcare access in emerging regions also contributes to broader patient coverage and uptake of treatments.
Technological advancements are reshaping clinical management. Minimally invasive surgical techniques, endoscopic systems, image-guided procedures, and AI-enhanced hearing devices improve treatment outcomes and procedural efficiency. Non-invasive therapies, such as nasal therapeutics and allergy immunotherapy, complement device-based interventions, reflecting a diversified treatment landscape.
Regional dynamics show North America leveraging high healthcare infrastructure and spending, while Asia-Pacific markets are growing due to improved access and demographic shifts. Cost constraints and limited adoption of premium technologies in resource-constrained regions remain challenges, but overall, the market is supported by strong clinical demand, demographic momentum, and technology-driven treatment expansion.
Definition: The ENT disorder treatment market comprises the global sector responsible for the provision of medical solutions for ear, nose, and throat conditions. The market includes the manufacturing and distribution of pharmaceutical drugs, surgical instruments, and prosthetic devices such as hearing aids and cochlear implants. It addresses a wide range of common and complex ailments, including chronic sinusitis, hearing impairment, tonsillitis, and sleep apnea. the market growth is consistently fueled by an aging global population, rising levels of environmental pollutants, and technological advancements in minimally invasive surgical techniques. Ultimately, the sector is integral to modern healthcare, supplying critical tools and therapies that restore sensory functions and meaningfully improve the daily quality of life for millions of patients worldwide.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients