Fungal otitis externa is a condition that causes inflammation (redness and swelling) of the external ear canal. Fungal otitis externa is often referred to as swimmer's ear, as repeated exposure to water can make the ear canal more vulnerable to inflammation. The most common pathogen is Aspergillus (80 to 90% of cases), followed by Candida. Fungal infection is the result of prolonged treatment of bacterial otitis externa that alters the flora of the ear canal.
The global fungal otitis externa market is estimated to be valued at US$ 169.40 million in 2021 and is expected to exhibit a CAGR of 2.9% over the forecast period (2021-2028).
Figure 1. Global Fungal Otitis Externa Market Share (%) in Terms of Value, By Drug Type, 2021
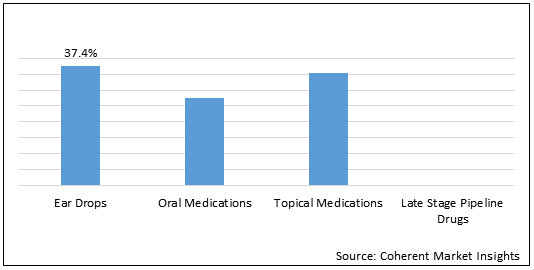
To learn more about this report, Request Free Sample
Increasing incidence and prevalence of fungal otitis externa are expected to drive the market growth over the forecast period.
Increasing incidence of fungal otitis externa is expected to drive the growth of global fungal otitis externa market over the forecast period. For instance, according to the article published in the National Center for Biotechnology Information, in March 2019, fungal otitis externa is common all over the world, with a higher incidence in tropical than in temperate zones owing to higher temperature and humidity. Its lifetime prevalence is estimated at 10%. It affects adults commonly, and children very rarely generally in the age range between seven to 12 years. Similarly, according to the same studies from the Netherlands and the U.K. have shown an annual incidence of 1%.
Figure 2. Global Fungal Otitis Externa Market Share (%), Fungus Type, 2021
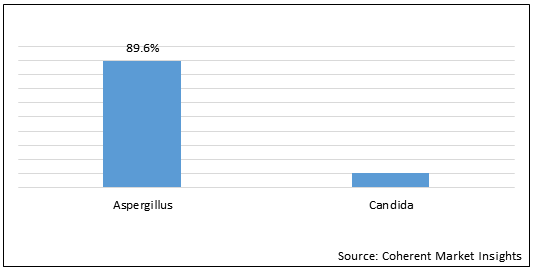
To learn more about this report, Request Free Sample
Robust product pipeline expected to offer the market growth opportunities over the forecast period.
Robust product pipeline for the treatment of fungal otitis externa which are expected to launch during the forecast period is anticipated to drive the growth of the global fungal otitis externa market. For instance, in February 27, 2020, Salvat Laboratories, a Spain-based pharmaceutical company, initiated a phase III, multicenter, randomized, doubleblind clinical trial to assess the efficacy and safety of SVT-15652 Otic Solution compared to placebo for the treatment of fungal otitis externa (otomycosis).
Fungal Otitis Externa Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2020 | Market Size in 2021: | US$ 169.40 Mn |
| Historical Data for: | 2017 to 2019 | Forecast Period: | 2021 to 2028 |
| Forecast Period 2021 to 2028 CAGR: | 2.9% | 2028 Value Projection: | US$ 206.94 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Novartis, Bayer AG, Sanofi, GlaxoSmithKline plc., AbbVie, Par Pharmaceutical, Inc., Hi-Tech Pharma, Actavis Mid-Atlantic, Hill Dermaceuticals Inc, NuLife Pharmaceuticals, Glenmark Pharmaceuticals Limited, Pfizer Inc., Janssen Pharmaceutical, McNeil Consumer Healthcare, Mylan N.V., Salvat Laboratories |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Fungal Otitis Externa Market– Impact of Coronavirus (COVID-19) Pandemic
Since the COVID-19 virus outbreak in December 2019, the disease has spread over 100 countries across the globe and the World Health Organization has declared it a public health emergency. According to the World Health Organization’s report, the manifestation of Coronavirus disease (COVID-19) has resulted in more than 189 million infected individuals worldwide as of July 19, 2021.
COVID-19 can affect the economy in three main ways; by directly affecting production and demand, by creating disruptions in distribution channels, and through its financial impact on firms and financial markets. Due to nationwide lockdowns, several countries such as China, India, Saudi Arabia, UAE, Egypt, and others are facing problems with regards to transportation of drugs from one place to another.
Furthermore, players operating in the global fungal otitis externa market are facing major challenges on various fronts due to the COVID- 19 pandemic. The major challenges include supply of raw materials required for manufacturing drug formulations due to irregularities in transportation facility. Moreover, distributors of products are experiencing irregular demand for products from the retailers due to increasing number of patients suffering from COVID-19 and other life threatening disorders.
Moreover, according to the article published in the National Center for Biotechnology Information in August 2020, the incidence of otitis externa increased in the patients suffering from COVID-19. According to the study results, increased incidence of otitis externa in COVID- 19 patients that is 18% of study group out of 257 cases were infected with COVID-19 and the symptoms were observed mainly between the fifth to eighth day of COVID-19 infection.
Global Fungal Otitis Externa Market- Restraint
Complications associated with drugs used in the treatment of fungal otitis externa and increasing generic competition is expected to hinder growth of the global fungal otitis externa market. Presence of generic drugs, at much lower price compared to branded drugs, in the market for the treatment of fungal otitis externa is a major factor, which is expected to hamper the market growth in the near future For instance, on July 2012, Mylan Inc., its subsidiary Mylan Pharmaceuticals Inc. received final approval from the U.S. Food and Drug Administration (FDA) for its Abbreviated New Drug Application (ANDA) for Itraconazole Capsules, 100 mg. This product is the generic version of Janssen Pharmaceutical Inc.'s Sporanox and is indicated for the treatment of fungal infections.
Key Players
Major players operating in the global fungal otitis externa market include Novartis, Bayer AG, Sanofi, GlaxoSmithKline plc., AbbVie, Par Pharmaceutical, Inc., Hi-Tech Pharma, Actavis Mid-Atlantic, Hill Dermaceuticals Inc, NuLife Pharmaceuticals, Glenmark Pharmaceuticals Limited, Pfizer Inc., Janssen Pharmaceutical, McNeil Consumer Healthcare, Mylan N.V., and Salvat Laboratories, among others
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients