The Huber Needles market is anticipated to grow at a CAGR of 6.1% with USD 50.72 Mn in 2026 and is expected to reach USD 76.7 Mn in 2033. The market is growing as more people have long-term illnesses that need IV treatments, more hospitals and clinics are opening, and doctors are using easier, less invasive ways (minimally invasive procedures 64 Mn) to give medicine. Better needle designs, stronger safety rules, and more healthcare in developing countries are also helping the market grow.
Curved Huber Needles is projected to account for the largest share of product type in 2026, representing approximately 60% of the total volume. As they work well, are more comfortable for patients, and are widely used in many medical treatments. The curved design of these needles shows a significant advancement in implantable port access technology, offering healthcare providers improved maneuverability and reduced tissue trauma during insertion procedures.
90″ curved Huber Needle Is made with Highest Quality imported from Japan. The tubing is made up of Pellethane Polyurethane material, ideal for chemotherapy application. This design advantage has been particularly recognized by major healthcare institutions as well as nursing organizations, with the Oncology Nursing Society highlighting the benefits of curved Huber needles in reducing insertion-related complications and improving patient outcomes in their clinical practice guidelines.
Based on application, dialysis dominates the market, accounting for a significant 55% share in 2026, owing to the high prevalence of chronic kidney diseases (CKD) and end-stage renal disease (ESRD) globally. According to data from the World Health Organization (WHO) and the Global Kidney Health Atlas, hundreds of millions of people suffer from kidney-related ailments, with many getting regular dialysis treatments to manage their condition. According to the National Kidney Foundation, 10% of the population worldwide is affected by chronic kidney disease (CKD), and millions die each year because they do not have access to affordable treatment.
Dialysis centers heavily rely on Huber needles because they allow for safer, more efficient, and less painful access to implanted ports and catheters. These needles consist of beveled tips and a special design to prevent damage or coring of the port septum, which is critical in repeated use scenarios such as dialysis. The safety and reliability associated with Huber needles ensure optimum patient care and reduce complications like infections or port failure, which might result from standard needles.
To learn more about this report, Request Free Sample
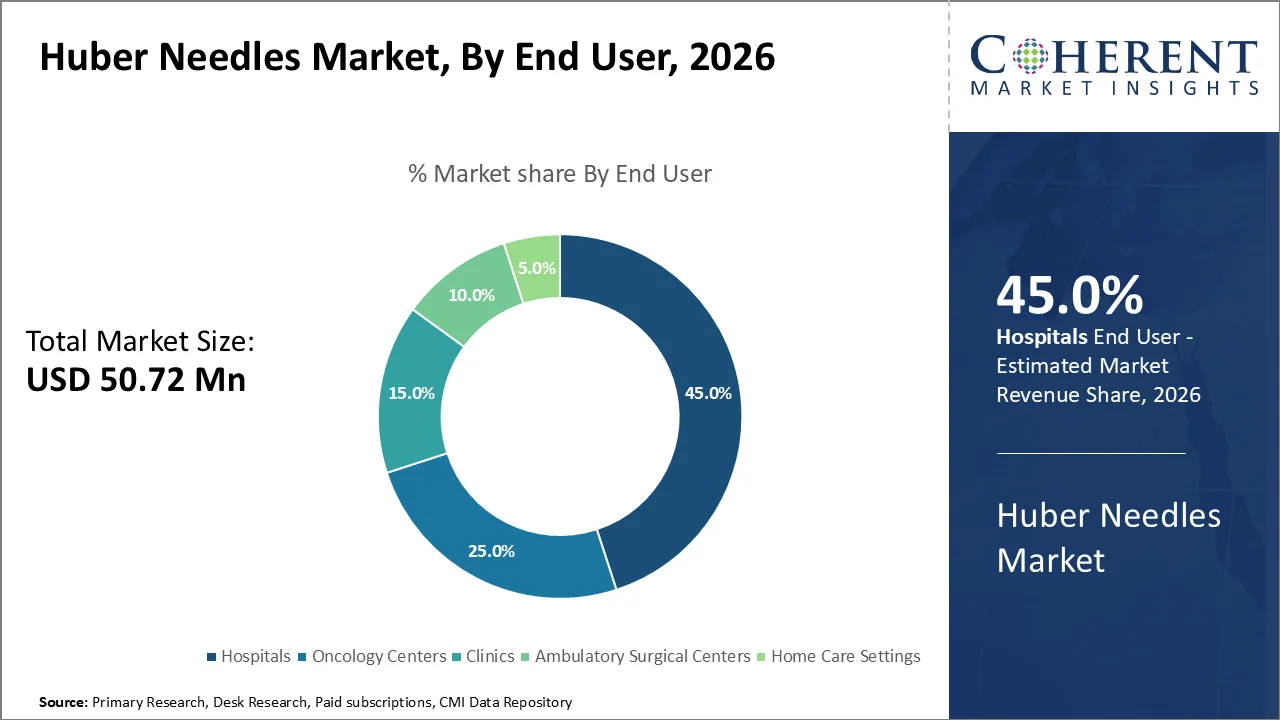
Based on end user, hospitals dominate the market, accounting for a significant 45% share in 2026, primarily due to their expansive role in delivering complex as well as high-volume healthcare services. These institutions typically handle a wide variety of medical conditions requiring frequent and precise vascular access, such as chemotherapy administration, blood sampling, parenteral nutrition, and long-term intravenous therapies.
Huber needles, which are specifically designed to prevent damage to implantable ports and reduce patient discomfort, are highly favored in hospital settings where patients often require repeated or continuous access to central venous catheters.
For instance, institutions like the Mayo Clinic, Cleveland Clinic, etc., utilize Huber needles extensively across multiple departments, from oncology wards to intensive care units, to maintain the safety along with efficiency of venous access.
Metric: American adults with chronic condition (in Per cent)
|
Age |
Per cent |
|
35–64 |
75% have at least one condition |
|
18–34 |
60% have at least one condition |
|
65 and older |
90% have at least one chronic condition |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Modern Huber needles use advanced coatings to improve safety, comfort, and biocompatibility. Hydrophilic coatings reduce friction during insertion, antimicrobial coatings prevent bacterial growth, as well as silicone-based coatings minimize tissue damage (BD, B. Braun, Smiths Medical). Companies like BD with PowerLoc needles, B. Braun with Ultrasite needles, and Smiths Medical with Gripper needles have adopted these technologies, showing reductions in insertion force, bacterial adhesion, and improved patient comfort.
The impact includes 30–40% less patient discomfort, 25% fewer catheter-related bloodstream infections, extended indwelling time from 5–7 to 7–14 days, and a 35% surge in healthcare provider satisfaction (BD, B. Braun, Smiths Medical).
Safety-engineered Huber needles use both passive and active mechanisms to protect healthcare workers from needlestick injuries. Passive safety shields activate automatically when the needle is withdrawn, while active systems require manual activation. Retractable needles fully retract into the housing, and blunting technologies dull the needle tip after use.
Companies like ICU Medical with the CLAVE NeedleFree Connector, Teleflex with the Arrow HuberPLUS, and AngioDynamics with BioFlo PICC have adopted these technologies, leading to an 80% reduction in needlestick injuries in hospitals as well as cancer centers. Overall, these safety innovations have reduced needlestick injuries by 70–85%, saved about USD1.2 billion annually in related costs, improved compliance with safety protocols by 45%, and cut liability and workers’ compensation claims by 60%.
|
Current Event |
Description and its Impact |
|
FDA Updates Infusion Device Safety Guidelines (2025) |
|
|
Expansion of Reimbursement Policies for Home Care Infusions in Europe (2026) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
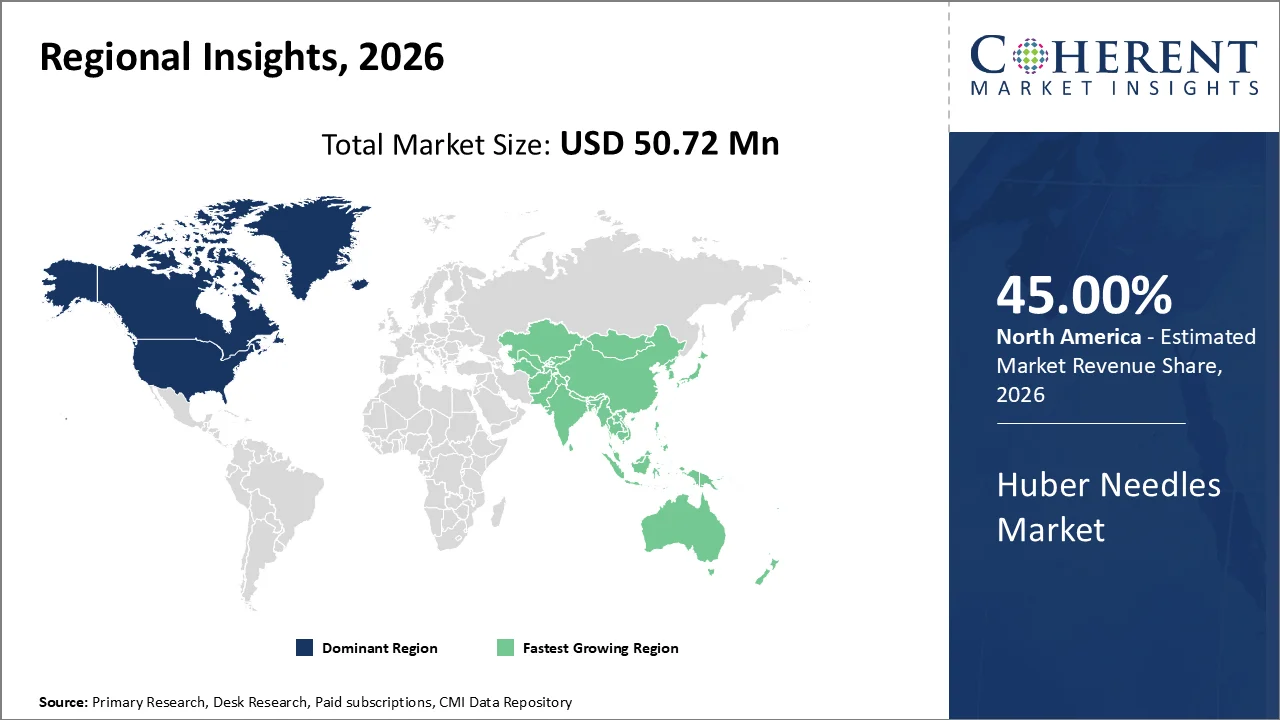
North America account 45% market share in 2026, North America, particularly the United States and Canada, hosts a robust network of advanced hospitals, oncology centers, and infusion therapy clinics that extensively utilize Huber needles due to their specialized design for safe, repeated access to implantable ports and catheters.
Many cases of cancer and long-term chronic diseases have increased the need for Huber needles. For example, the National Cancer Institute (NCI) estimated nearly 1.9 million new cancer cases in the U.S. in recent years. This makes Huber needles important because they are safe, reduce infection risk, and make chemotherapy more comfortable for patients.
The Asia-Pacific region is poised to be the fastest-growing region through 2026-2033, expanding at a CAGR of approximately 7.5%. The region presents a unique landscape marked by stark variations in healthcare infrastructure, which fuels demand for a broad spectrum of Huber needle products, from ultra-high precision needles for specialized oncology centers in Japan as well as South Korea to cost-effective, mass-produced variants tailored to meet basic medical needs across India, Indonesia, and Vietnam.
For example, India’s rapid expansion in outpatient chemotherapy centers and homecare services, supported by government programs such as Ayushman Bharat, will have a defined benefit cover of Rs. 5 lakh per family per year which necessitates widespread usage of Huber needles designed for long-term vascular access.
Furthermore, the Chinese government is spending a lot on improving healthcare through its ‘Healthy China 2030’ plan, focusing on better cancer care and increasing the need for advanced IV devices.
Many top hospitals, cancer centers, and specialty clinics create a high demand for precise medical devices like Huber needles, which are used to access ports for chemotherapy and apheresis treatments.
For example, the U.S. National Cancer Institute reports that approximately 1.9 million new cancer cases were diagnosed in the country in the past year, resulting in a substantial requirement for reliable venous access devices to facilitate chemotherapy treatments.
This creates sustained demand for Huber needles, which are widely preferred for their non-coring design that minimizes port damage and patient discomfort.
The highly advanced medical infrastructure as well as stringent quality standards governing medical device usage is working as the growth inducing factor for the growth of the market. The country’s robust healthcare system, backed by initiatives by government authorities that emphasize patient safety and precision in invasive medical procedures, has created substantial demand for superior quality Huber needles primarily used for accessing implantable ports.
Japan has one of the oldest populations in the world, which leads to many people having cancer and chronic diseases. These conditions often need chemotherapy or IV nutrition, where Huber needles are very important. For instance, according to Japan’s Ministry of Health, Labour and Welfare, cancer remains one of the leading causes of mortality, along with an estimated 1 million patients undergo chemotherapy annually, necessitating reliable and safe vascular access devices like Huber needles.
Some of the major key players in Baxter International Inc., B. Braun Medical Inc., Smiths Medical, NIPRO Medical Corporation, AngioDynamics, Inc., and C. R. Bard, Inc.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 50.72 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.1% | 2033 Value Projection: | USD 76.7 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Baxter International Inc., B. Braun Medical Inc., Smiths Medical, NIPRO Medical Corporation, AngioDynamics, Inc., and C. R. Bard, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients