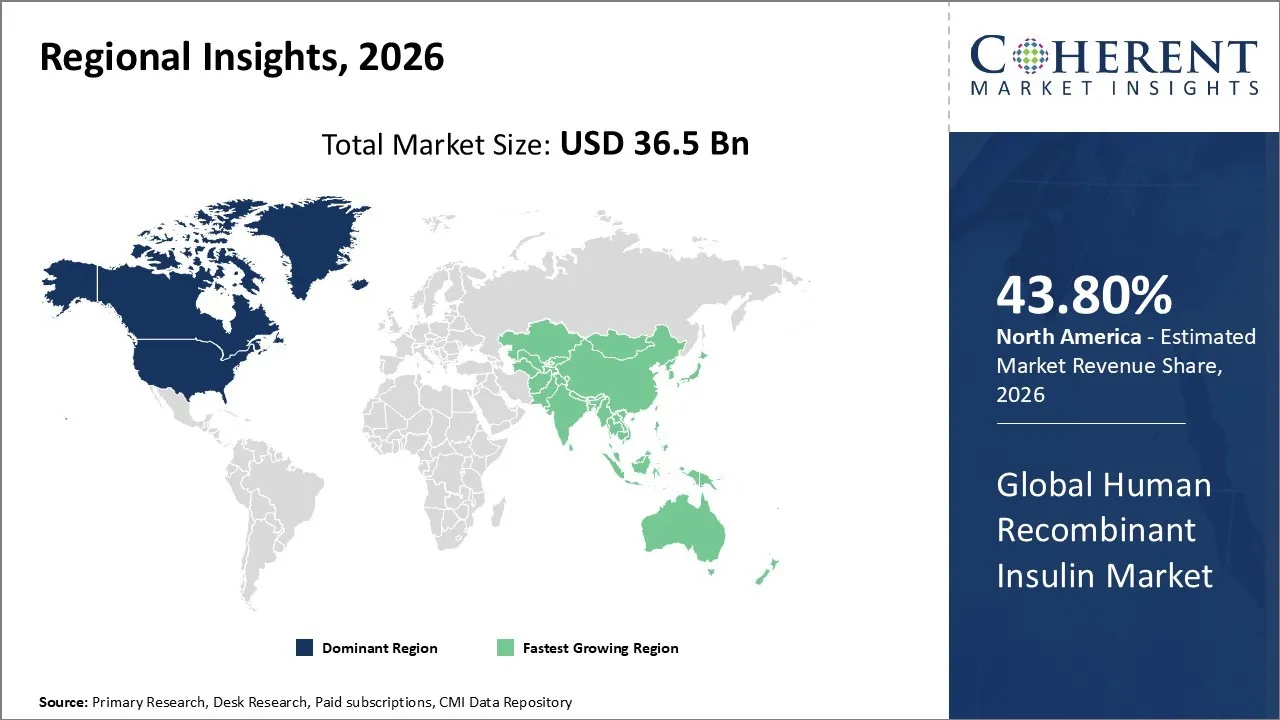
The Human Recombinant Insulin Market is estimated to be valued at USD 36.5 Bn in 2026 and is expected to reach USD 60.1 Bn by 2033, growing at a compound annual growth rate (CAGR) of 7.4% from 2026 to 2033.
The human recombinant insulin market is advancing significantly with the rising demand for laboratory-synthesized hormonal therapies and effective glycemic control solutions in the global healthcare and metabolic disorder sectors. The growing consumer awareness regarding chronic disease management and a strategic shift away from animal-derived insulin products are expected to propel market growth over the forecast period. The human recombinant insulin is produced using cutting edge recombinant DNA technology in genetically engineered microorganisms like Escherichia coli or yeast. The final product is a highly purified hormone. This makes human recombinant insulin a vital component in modern diabetes care and life-saving therapeutic formulations.
The industry is entering a transformative period owing to the use of advanced bioprocessing technologies, a surge in the prevalence of type 1 and type 2 diabetes, and the integration of insulin biosimilars into diverse healthcare systems. These advancements enhance the purity, safety, and scalability of human recombinant insulin production to meet the needs of a growing patient population. As a result, the manufacturers can supply cost-effective, high-performance alternatives to standard therapies while ensuring compliance with stringent global regulations governing pharmaceutical quality and therapeutic transparency.
|
Current Event |
Description and the Impact |
|
Geopolitical and Regulatory Developments |
|
|
Technological Innovations and Biomanufacturing Advances |
|
|
Epidemiological and Demographic Shifts |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of product type, the short-acting human insulin segment contributes the highest share of 37.40% in 2026 of the market. The growth is owing to its essential role for postprandial glucose management during mealtimes. The short-acting recombinant insulin mimics the natural bolus response to food intake. The widespread adoption is bolstered by a lower price point compared to modern rapid-acting analogs, thereby making it a critical resource in emerging economies. In addition, its versatility ensures a stronghold as a primary treatment because it is the only one suitable for intravenous use in hospital settings for emergencies like ketoacidosis.
In terms of brand, the Humulin R segment contributes the highest share of 16.80% in 2026 of the market. The growth is owing to sustained physician trust and brand loyalty established by early leadership in recombinant DNA insulin technology. The healthcare providers demonstrate a strong preference for Humulin R. This is attributable to its long-standing safety record and high purity derived from proprietary fermentation processes. This clinical confidence and extensive global supply chains, as well as differentiated pricing, have helped the brand sustain market share amid rising biosimilar penetration. The recent legislative price caps in major markets have also made this premium brand more accessible for both newly diagnosed patients and long-term users.
In terms of distribution channel, the retail pharmacies segment contributes the highest share of 43.20% in 2026 of the market. The growth is owing to a global shift toward home-based diabetes management and the increasing availability of user-friendly delivery devices like pens and prefilled syringes. The retail pharmacy chains and independent pharmacies have expanded their reach into suburban and rural markets, thus improving access to insulin for a wider patient population. The availability of automated refill services and pharmacist-led counseling has contributed to better treatment adherence. This expanded access and service integration has enabled retail pharmacies to lead the market.
To learn more about this report, Request Free Sample
North America has emerged as the leader in the global market and accounts for a higher share of 43.80% in 2026. The growth is due to an increase in diabetes access, mainly in the US. The market is marked by a strong healthcare system and comprehensive insurance coverage as well as pharmaceutical companies like Eli Lilly, Novo Nordisk, and Sanofi. A key trend shaping the industry is the rapid transition toward next-generation insulin delivery systems. The patient preference is moving away from conventional vials and syringes toward more convenient and user-friendly options like insulin pens, automated insulin pumps, and wearable patch pumps.
These technologies are frequently integrated with continuous glucose monitoring (CGM) systems and smartphone-based applications, enabling improved dosing accuracy, real-time glucose tracking, and higher levels of patient adherence. Innovation in this segment is further supported by strategic collaborations between pharmaceutical manufacturers and medical technology companies, including partnerships with firms such as Tandem Diabetes Care and Abbott. In parallel, the regulatory and economic landscape is undergoing significant transformation, with a growing emphasis on affordability and market competition.
For instance, in October 2025, Biologics Ltd., a subsidiary of Biocon Ltd., has expanded its strategic partnership with Civica, Inc. to include a new Insulin Glargine medication. The goal of this initiative is to enhance the availability of high-quality, affordable insulins for patients in the US.
The Asia Pacific region is the fastest-growing region in the global human recombinant insulin market. The growth is owing to a substantial demographic shift and rapid modernization of the electorate. The countries like China and India face a growing health burden and are generating sustained demand for affordable and scalable insulin therapies. The growth in the region is defined by a structural shift away from traditional animal-derived insulins toward human recombinant formulations. This is driven by the rising disposable incomes and expanded public reimbursement frameworks, which together are improving access to higher-quality treatments across a broader patient population.
The government intervention has become a central driver of market evolution across the region. In China, centralized volume-based procurement has resulted in lower insulin prices, thereby increasing patient access while pushing manufacturers to focus on scale-driven efficiency. India has positioned itself as a global manufacturing hub, with domestic companies like Biocon supplying competitively priced biosimilar insulins to both domestic and export markets. These regional manufacturers, along with multinational firms like Novo Nordisk and Sanofi, are investing more in local production facilities. this allows them to manage regulations in an easier way, cut supply chain costs, and improve market responsiveness.
For instance, in December 2025, Cipla Limited unveiled Afrezza Inhalation Powder in India. The Central Drugs Standard Control Organisation (CDSCO) has approved the company's exclusive distribution and marketing of Afrezza®. This rapidly acting, orally inhaled insulin provides a needle-free and convenient alternative to injectable insulin therapies.
The U.S. remains the most profitable market for human recombinant insulin globally owing to strong healthcare spending and the high number of patients with both Type 1 and Type 2 diabetes. The market is defined by strong government and regulatory pressure to improve affordability, thus leading to broad out-of-pocket cost caps for many insured patients. This shift has resulted in increased adoption of interchangeable biosimilars, which offer lower-cost alternatives to original brand-name products. The private insurers and Medicare prioritize high-cost analogs and integrated technologies over human insulin. Traditional human insulin serves as a critical low-cost bridge for the uninsured population despite the market dominance of expensive analogs. Furthermore, the American market is unique for its rapid integration of automated delivery systems and continuous glucose monitoring technologies.
For instance, in April 2023, Eli Lilly and Company has introduced its insulin glargine biosimilar, Rezvoglar, to the US market. This makes it the second interchangeable insulin biosimilar available for patients with diabetes in America.
China is the fastest growing market for human recombinant insulin owing to the world’s largest number of diabetes patients. The market is currently undergoing a shift as domestic firms challenge the historical dominance of multinational corporations. The government-led volume-based procurement programs have reduced prices to increase affordability for rural patients. The patients are transitioning from traditional human insulin toward newer insulin types and biosimilars. The national focus on biomanufacturing and research infrastructure drives steady demand growth through industry upgrades. The focus is shifting toward modern delivery systems like smart insulin pens and integrated digital health platforms that cater to a rising middle class.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 36.5 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.4% | 2033 Value Projection: | USD 60.1 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Novo Nordisk A/S, Eli Lilly and Company, Sanofi S.A., Julphar Gulf Pharmaceutical Industries, Biocon Limited, Bioton S.A., Gan & Lee Pharmaceuticals, Ltd., Zhuhai United Laboratories Co., Ltd., Wanbang Biopharmaceuticals Co., Ltd., and Dongbao Enterprise Group Co., Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The increasing primary prevalence of diabetes is the primary driver of growth in the Human Recombinant Insulin Market. The rising number of diagnoses worldwide creates a sustained and urgent demand for effective blood sugar management. As sedentary lifestyles and urbanization become more common, the need for long-term glycemic control is essential to prevent chronic complications. Human recombinant insulin has surpassed animal-derived alternatives because it offers better purity and significantly lower risk of adverse reactions. This shift is supported by a growing aging population and breakthroughs in biotechnology that allow for reliable, large-scale manufacturing. In addition, the emergence of cost-effective biosimilars and improved delivery systems is making these life-saving treatments more accessible across developing regions, solidifying insulin’s role as a fundamental pillar of modern medical care.
For instance, in February 2025, the U.S. FDA has approved Merilog as a biosimilar to Novolog to enhance glycemic control in adults and children with diabetes. Merilog is notable for being the first rapid-acting insulin biosimilar to receive U.S. FDA.
Definition: The Human Recombinant Insulin Market encompasses the global industry involved in the research, manufacturing, and sales of insulin produced via recombinant DNA (rDNA) technology. The genetically modified microorganisms, like E. coli and yeast can be used to generate human insulin. This provides a biologically identical alternative for therapeutic use. This market is mainly driven by the increasing global prevalence of diabetes and the critical need for high-purity, biocompatible treatments that minimize allergic reactions compared to older animal-sourced versions. It encompasses a broad range of products, including short-, intermediate-, and long-lasting formulations, administered through pens, vials, and advanced pump systems. It is a million-billion-dollar sector at the vital intersection of biotechnology and chronic disease management.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients