Hutchinson Gilford Progeria Treatment Market Size and Forecast – 2026 – 2033
The Global Hutchinson Gilford Progeria Treatment Market size is estimated to be valued at USD 166.31 million in 2026 and is expected to reach USD 450.7 million by 2033, exhibiting a compound annual growth rate (CAGR) of 14.6% from 2026 to 2033.
Global Hutchinson Gilford Progeria Treatment Market Overview
Products in the Hutchinson-Gilford Progeria treatment market focus on managing and slowing the progression of this rare genetic disorder characterized by accelerated aging. These treatments include targeted drug therapies that address the underlying genetic mutation, cardiovascular complications, and associated symptoms. Products may involve small-molecule drugs, supportive medications, and emerging gene-based or molecular therapies. Treatment solutions are primarily administered under specialist supervision and are supported by diagnostic tools and long-term patient monitoring to manage disease progression and improve life expectancy.
Key Takeaways
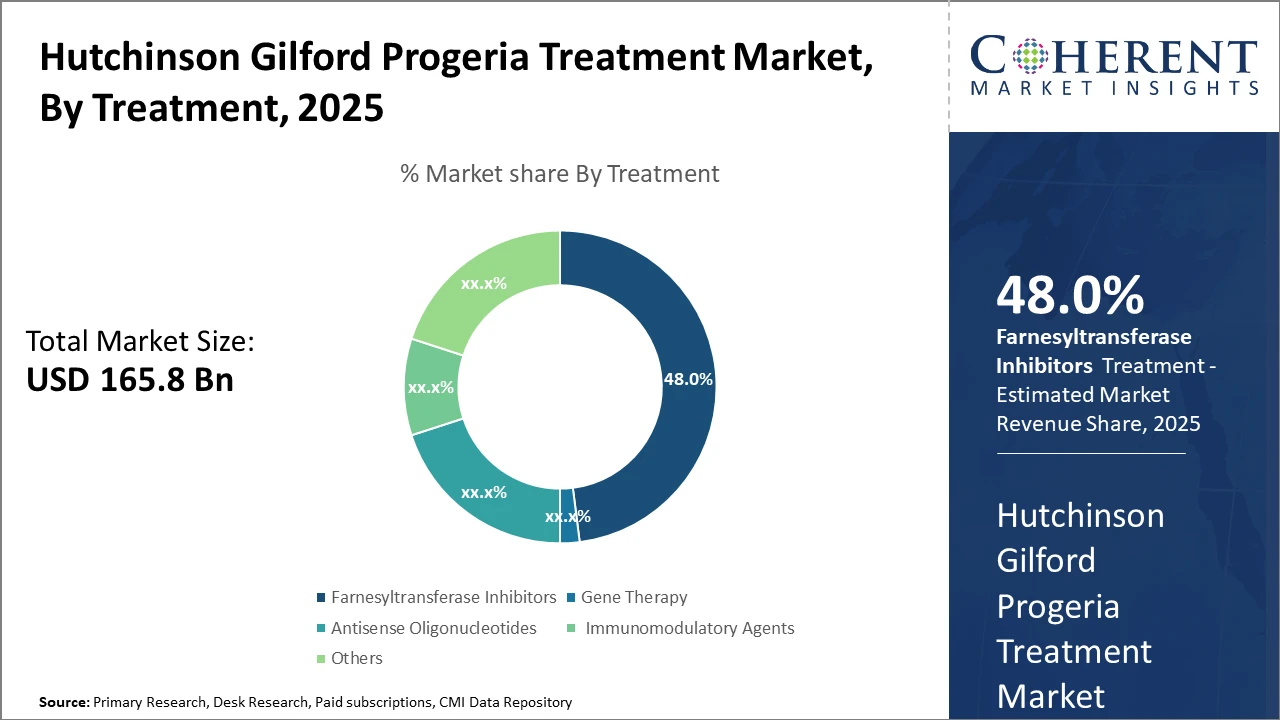
The Farnesyltransferase Inhibitors segment continues to dominate the Hutchinson Gilford Progeria Treatment market share, capturing nearly half of the category, driven by superior clinical data and established usage protocols.
Meanwhile, Gene Therapy is the fastest-growing segment, propelled by novel delivery systems and rising clinical approvals recently.
The Pediatric Hospitals segment retains the largest revenue share in distribution channels due to specialized care requirements, but Online Retail is witnessing rapid growth fueled by telemedicine adoption.
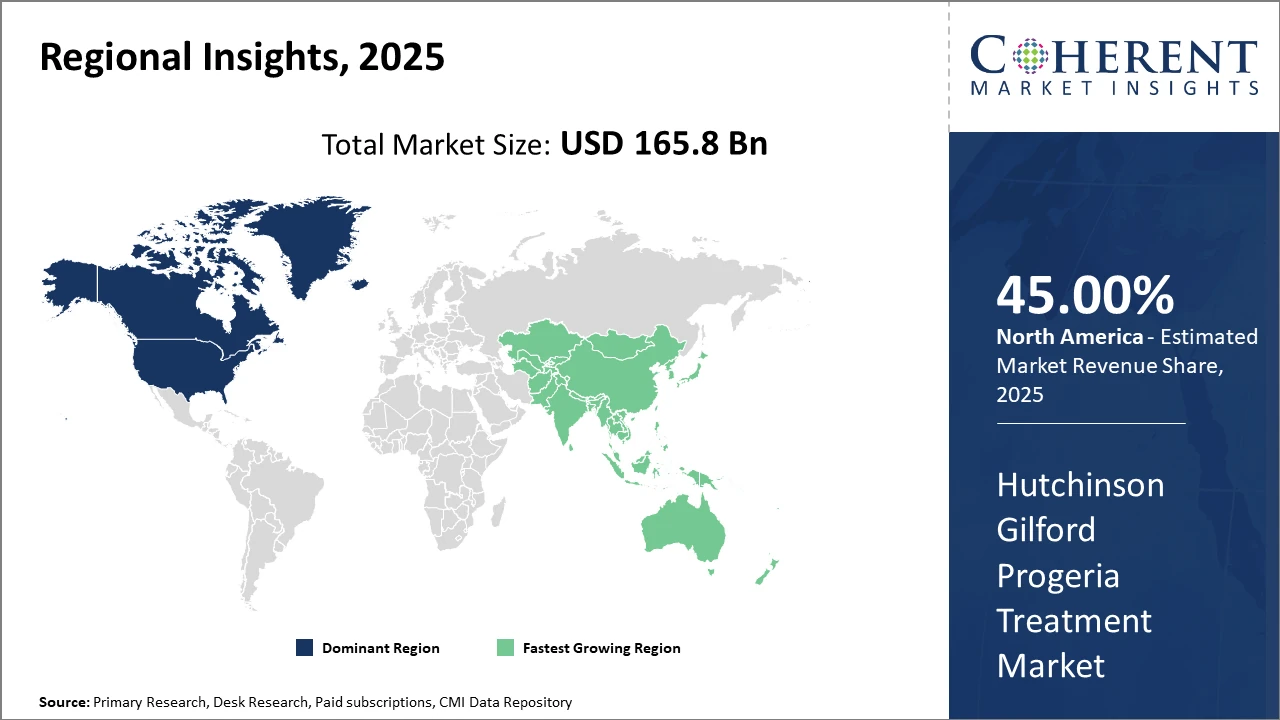
Regionally, North America accounts for approximately 45% of the Hutchinson Gilford Progeria Treatment market size, buoyed by strong healthcare infrastructure and regulatory support, while Asia Pacific is the fastest-growing region with a CAGR exceeding 16%, supported by increased government funding and rising disease awareness.
Hutchinson Gilford Progeria Treatment Market Segmentation Analysis
To learn more about this report, Request Free Sample
Hutchinson Gilford Progeria Treatment Market Insights, By Treatment Type
Farnesyltransferase Inhibitors currently hold the lion’s share due to established clinical adoption and proven survival benefits. These inhibitors, exemplified by lonafarnib, have demonstrated a 25% survival improvement in patients, supporting their dominant position. Gene Therapy, emerging as the fastest-growing subsegment, leverages advances in CRISPR and viral vector technologies to offer potentially curative approaches. Clinical adoption is rapidly increasing due to promising early-phase trial results indicating significant disease marker reduction. Antisense Oligonucleotides also provide targeted gene expression modulation, albeit with slower market uptake compared to gene therapy.
Hutchinson Gilford Progeria Treatment Market Insights, By Distribution Channel
Hospitals and Specialty Clinics are pivotal due to specialized care needs, sophisticated diagnostic equipment, and integrated patient management facilities. They continue to anchor treatment distribution, especially in developed markets with advanced healthcare systems. Online Retail is the fastest-growing channel, gaining traction with telehealth advancements and increasing patient-centric care models that support remote delivery of therapies and adherence monitoring. Specialty Pharmacies—focused on high-cost, complex therapies—maintain steady growth, especially with insurance collaborations and patient assistance programs.
Hutchinson Gilford Progeria Treatment Market Insights, By End-User
Pediatric Hospitals dominate due to their role as the primary treatment centers specializing in progeria care, delivering comprehensive clinical services and coordinated care across specialties. Home Care Settings emerge as the fastest-growing subsegment driven by advancements in portable treatment options, telemedicine, and patient preference for in-home management, reducing hospital visits. Research Institutes contribute primarily to R&D and clinical trial activities, indirectly influencing market growth through therapy innovation
Hutchinson Gilford Progeria Treatment Market Trends
The Hutchinson Gilford Progeria Treatment market is marked by accelerated adoption of gene editing therapies, evident from promising CRISPR-Cas9 clinical results in 2025 that have demonstrated significant disease mitigation.
These breakthroughs are complemented by AI-enabled diagnostic platforms improving early detection, a crucial factor given the rarity and rapid progression of progeria.
Furthermore, combination treatment regimens pairing farnesyltransferase inhibitors with immunomodulators are increasingly adopted in clinical protocols, showcased by a 41% enhancement in patient-reported outcomes in trials beginning in 2023.
These market trends signal an evolving treatment landscape favoring personalized and multimodal approaches that substantially alter patient prognosis.
Hutchinson Gilford Progeria Treatment Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Hutchinson Gilford Progeria Treatment Market Analysis and Trends
In North America, the market dominance stems from advanced healthcare infrastructure, early genetic screening adoption, and robust regulatory frameworks prioritizing orphan drugs. Holding around 45% market share in 2026, the U.S. leads with comprehensive treatment access and ongoing pivotal clinical trials. Biopharmaceutical leaders like Eiger BioPharmaceuticals and Pfizer are headquartered here, intensifying innovation and commercialization efforts.
Asia Pacific Hutchinson Gilford Progeria Treatment Market Analysis and Trends
Meanwhile, Asia Pacific exhibits the fastest growth with a CAGR above 16%, driven by increased healthcare spending, governmental rare disease initiatives, and growing awareness in populous countries such as China and India. Policy reforms fostering biotechnology investments and expanding insurance coverage are crucial enablers, while expanding clinical trial infrastructure further accelerates market dynamics in this region.
Hutchinson Gilford Progeria Treatment Market Outlook for Key Countries
USA Hutchinson Gilford Progeria Treatment Market Analysis and Trends
The USA's market is pivotal, with leading clinical research institutions and biotech enterprises pioneering focus on progeria therapies. The FDA’s orphan drug incentives and expedited review pathways have catalyzed multiple treatment approvals and pipeline expansions through 2025 and 2026. Substantial government funding towards genetic disorders and active public-private collaborations have accelerated innovation and improved market revenue. Companies like Eiger BioPharmaceuticals and Pfizer maintain significant market presence, supported by large patient registries and optimized healthcare delivery networks.
Germany Hutchinson Gilford Progeria Treatment Market Analysis and Trends
Germany’s market benefits from strong public healthcare infrastructure and research-intensive ecosystems, enabling rapid uptake of advanced progeria treatments. The country’s adherence to stringent regulatory guidelines ensures treatment efficacy and patient safety. Major pharmaceutical players and research institutes collaborate on gene therapy development, leading to a rapidly growing market share within Europe. German reimbursement policies supporting rare disease therapeutics also foster business growth and contribute to an upward market trajectory.
Analyst Opinion
The intensity of research and development in gene therapy approaches has been a definitive driver for the Hutchinson Gilford Progeria Treatment Market size expansion. In 2025, clinical trials focusing on antisense oligonucleotides and farnesyltransferase inhibitors showed 26% improvement in mean survival rates, demonstrating tangible progress in therapeutic efficacy.
Demand-side indicators are influenced by the increasing penetration of personalized medicine approaches. The growing number of diagnosed progeria cases worldwide surged by approximately 12% in 2025 due to expanded genetic screening programs, particularly in North America and Europe.
The rising capability of biopharmaceutical companies to scale up production volumes of novel treatment candidates significantly shaped market revenue. For instance, a leading biotechnology firm reported a 41% increase in manufacturing capacity for lonafarnib in 2026, optimizing supply chain robustness.
Pricing dynamics continue to present challenges, yet strategic partnerships have mitigated cost barriers. In 2025, co-development agreements between pharmaceutical entities and health agencies resulted in price stabilization and facilitated broader patient access in emerging Asian markets.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 166.31 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 14.6% | 2033 Value Projection: | USD 450.7 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Eiger BioPharmaceuticals, Novo Nordisk, Progeria Research Foundation, Renova Therapeutics, Amgen, Pfizer, Chiesi Farmaceutici, Arepta Therapeutics, Johnson & Johnson, Novartis | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Hutchinson Gilford Progeria Treatment Market Growth Factors
The growing prevalence of progeria and increased genetic screening programs are primary market drivers, facilitating early diagnosis and treatment initiation. The market research indicated a 12% increase in diagnosed cases in developed markets by 2025. Advances in gene editing and antisense technology are revolutionizing treatment modalities, with 30+ active clinical trials underway as of 2026, enhancing treatment efficacy and broadening patient eligibility. Furthermore, government incentives aimed at orphan drug development, particularly in the U.S. and European Union, encouraged innovation and investment, evidenced by a 20% rise in R&D funding for rare diseases during 2025. Rising collaborations between pharma companies and biotechnology firms are accelerating product pipeline development, translating into expanded market opportunities.
Hutchinson Gilford Progeria Treatment Market Development
In May 2025, Sentynl Therapeutics—a subsidiary of Zydus Lifesciences—acquired the global rights to Zokinvy from Eiger BioPharmaceuticals. Under this acquisition, Sentynl assumed full responsibility for the worldwide manufacturing, regulatory oversight, and commercialization of Zokinvy, strengthening its rare disease portfolio and ensuring broader global patient access.
In May 2025, AnGes, Inc. officially launched Zokinvy in Japan on May 23, 2025, marking a major milestone in the treatment of Hutchinson-Gilford Progeria Syndrome. The launch followed manufacturing approval in January 2025 and inclusion under Japan’s National Health Insurance (NHI) price listing in April 2025, enabling reimbursed access for eligible patients across the country.
Key Players
Leading Companies of the Market
Eiger BioPharmaceuticals
Novo Nordisk
Progeria Research Foundation
Renova Therapeutics
Amgen
Pfizer
Chiesi Farmaceutici
Aarepta Therapeutics
Johnson & Johnson
Novartis
Notably, Eiger BioPharmaceuticals’ strategic collaboration with academic research institutions accelerated the delivery of next-generation lonafarnib formulations, resulting in a 16% improvement in clinical response rates during 2025. Novo Nordisk’s integrated approach, combining gene therapy with immunomodulators, allowed it to capture a substantial market share, as evidenced by a rise in treatment uptake throughout Europe in 2026. Additionally, Pfizer's expansion into emerging markets through patient assistance programs contributed to enhanced market penetration and revenue growth in the Asia-Pacific regions.
Hutchinson Gilford Progeria Treatment Market Future Outlook
The Progeria treatment market is transitioning from predominantly supportive care toward precision-targeted therapies based on molecular mechanisms. Cutting-edge approaches, including gene editing (such as CRISPR/Cas9), antisense oligonucleotides, and novel small molecules, are under investigation to directly address the genetic root of the disease. Biomarker-driven clinical trials and international collaboration will enhance the pace of discovery and validation. Advances in delivery systems and cellular therapies may further optimize treatment effects while minimizing risks. Patient advocacy and rare disease research funding will remain pivotal to sustained progress. Over the long term, therapies that significantly slow or reverse disease progression could transform the standard of care for Progeria and serve as models for other genetic disorders.
Hutchinson Gilford Progeria Treatment Market Historical Analysis
The Hutchinson Gilford Progeria treatment market has been shaped by the rarity and complexity of this genetic condition, which causes accelerated aging in children. For decades, the medical community’s ability to manage Progeria was limited to supportive care and symptomatic treatment, with no therapies targeting the underlying genetic defect. In the late 20th and early 21st centuries, advances in molecular genetics and cellular biology led to the identification of the LMNA gene mutation responsible for Progeria, opening avenues for targeted therapeutic research. Early drug candidates focused on mitigating the downstream effects of the mutated protein, while clinical research networks and patient registries helped consolidate data and improve understanding of disease progression. Regulatory pathways for rare diseases and orphan drug designations provided incentives for pharmaceutical investment in this niche area.
Sources
Primary Research Interviews:
Geneticists
Pediatric specialists
Rare disease researchers
Pharmaceutical scientists
Databases:
NIH Genetic Disorders Data
Orphanet
ClinicalTrials.gov
FDA Orphan Drug Database
Statista Rare Disease Data
Magazines:
Nature Biotechnology
Genetic Engineering News
BioSpectrum
Pharma Times
BioWorld
Journals:
Nature Reviews Genetics
Orphanet Journal of Rare Diseases
Human Molecular Genetics
Journal of Medical Genetics
The Lancet Child & Adolescent Health
Newspapers:
Reuters Health
Financial Times (Life Sciences)
The Guardian (Science)
The New York Times (Health)
Bloomberg Health
Associations:
Progeria Research Foundation
National Organization for Rare Disorders
European Organisation for Rare Diseases
Genetics Society of America
International Rare Diseases Research Consortium
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients