The global OTC consumer health market is estimated to be valued at USD 221.86 Bn in 2026 and is expected to exhibit a CAGR of 4.6% during the forecast period (2026-2033).
Over-the-counter (OTC) drugs are medicines sold directly to a consumer without a requirement for a prescription from a healthcare professional, as opposed to prescription drugs, which may be supplied only to consumers possessing a valid prescription. In several countries, OTC drugs are selected by a regulatory agency to ensure that they contain ingredients that are safe and effective when used without a physician's care. OTC drugs are usually regulated according to their active pharmaceutical ingredients (API) rather than final products. By regulating APIs instead of specific drug formulations, governments of countries allow manufacturers the freedom to formulate ingredients, or combinations of ingredients, into proprietary mixtures.
|
Current Events |
Description and its impact |
|
Technological & Distribution Innovations |
|
|
Regulatory Modernization and Policy Shifts |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Based on Product type, the dermatological products segment is dominating the market with 27.4% of the global market revenue. The major drivers include the rising awareness regarding skin health, a growing trend for self-care. The consumers are also engaging in the management of minor skin conditions like acne, eczema, dryness, etc. Hence, there is a surge in the demand for easy-to-access and non-prescriptive treatment methods. Moreover, the growth of the quick e-commerce industry has expanded and established channels that have made it easier to acquire OTC products. The consumer base of the Dermatological segment ranges from teenagers to older adults, either seeking relief from acne or anti-aging solutions.
In February 2025, Embryolisse, a popular French skincare company, launched its product Filaderme Emulsion in India. The product is designed to address the distress of dry and sensitive skin.
To learn more about this report, Request Free Sample
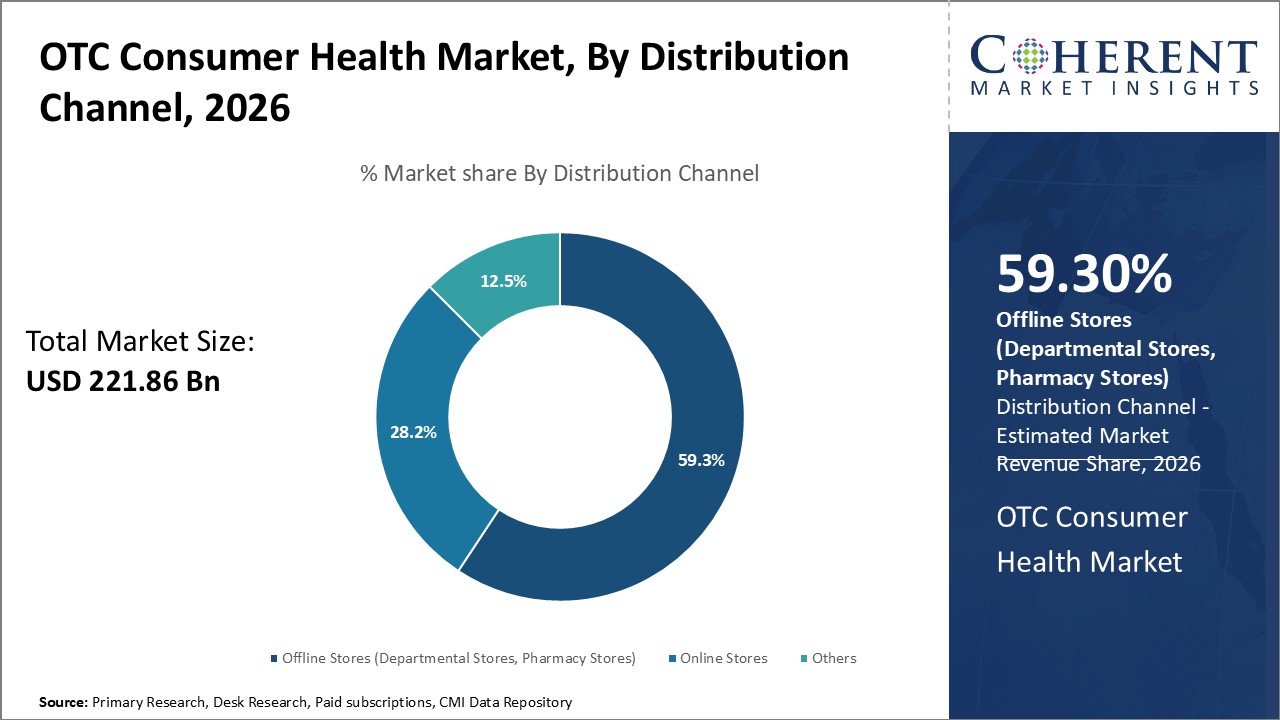
Based on the Distribution Channel, the Pharmacy Stores segment is leading the market, owing to the trust of consumers in pharmacists, who are often consulted for advice on minor health concerns. Pharmacies also offer a wide range of products, right from cold remedies to dermatological creams. Some of the key drivers of this segment include quality assurance and safety, and personalized services like consultations.
In June 2024, Walgreens launched its OTC nasal spray called Naloxone Nasal Spray. The product is significant as it is considered to be a life-saving drug that is available in all Walgreens’ stores.
To learn more about this report, Request Free Sample
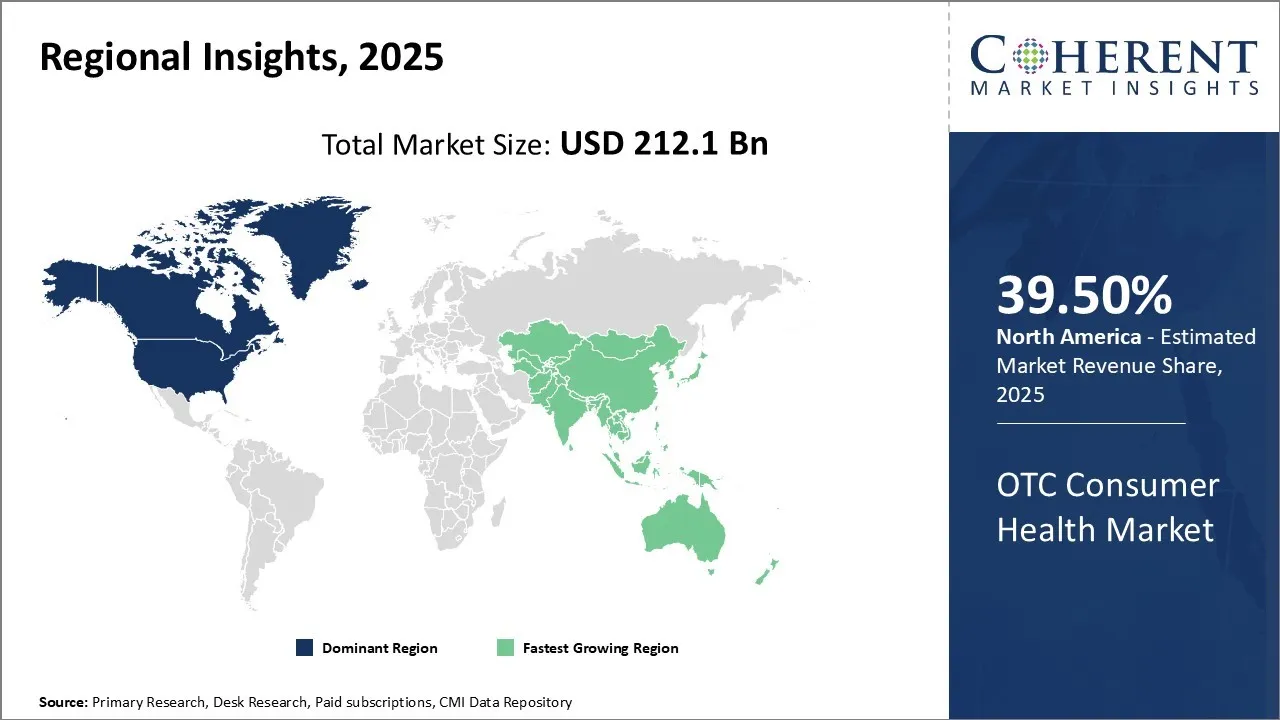
Among regions, the North America market is expected to witness significant growth, owing to key companies in the market focusing on adopting inorganic strategies such as agreements with other market players. North America has emerged as the largest market for OTC consumer health in 2026, accounting for around 39.5% of the global market share. The United Nations and Canada are the top two countries leading the market in North America. On one hand, the United States is the largest global OTC market, primarily driven by the rise in the aging population, and Digital health and DTC trends. On the other hand, Canada follows closely by expanding a health-conscious consumer base, an increase in the focus on preventive healthcare, and government regulations etc. For instance, in July 2022, Dr. Reddy’s Laboratories Ltd., a pharmaceutical company, announced that it had entered into a licensing agreement with Slayback Pharma LLC, a specialty pharmaceutical company to acquire rights in Slayback’s Brimonidine Tartrate Ophthalmic Solution 0.025%, the private label equivalent of Lumify in the U.S. Lumify is an over-the-counter (OTC) eye drop that can be used to relieve redness of the eye due to minor eye irritations.
The Asia-Pacific region is the second-largest market globally. The OTC Consumer Health Market’s growth in this region is attributed to the large population, particularly to the rise in middle-class spending and disposable income. Rapid urbanization, easy access to pharmacies, and increasing awareness regarding preventive health are equally contributing to the market’s growth. Two countries show notable contributions to the OTC Consumer Health Market’s growth: China and India. While the expansion of popular e-commerce platforms such as Alibaba and JD.com is primarily driving the market’s growth in China, the increase in use of traditional Chinese medicine in OTC, and government reforms, too, are aiding to it. Meanwhile, in India, there is a surge in the demand for ayurvedic products, inside and outside the country. For instance, in March 2024, Emcure Pharmaceuticals launched its new product Galact, a lactation supplement enriched with more than 5 different herbs. The purpose of the product is to support the production of breast milk in mothers. It is a significant move as it moves from prescription-based to OTC. The rise in disposable income is driving the OTC Consumer Health market share.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 221.86 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.6% | 2033 Value Projection: | USD 303.94 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Johnson & Johnson, Abbott Laboratories, Sanofi, GlaxoSmithKline plc. Bayer AG, Nestle Company, Procter & Gamble Corp., Danone Food Company, Colgate Palmolive Mfg. Company, Unilever, and Henkel AG & Co. KGaA |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Increasing product launches by market players are expected to drive the global OTC consumer health market growth over the forecast period. For instance, Hamdard Laboratories, a Unani pharmaceutical company has launched 12 over-the-counter (OTC) medicines that help boost immunity. The new range of products contains ingredients such as Aswagandhi, Kalonji, Gilo, and Jamun powder along with Zafran, which are known for strengthening body immunity and overall health. This is further positively influencing the OTC consumer health market share.
Increasing product approvals by regulatory authorities are expected to drive the five-year growth of the global OTC consumer health market over the forecast period. For instance, in January 2025, Sanofi’s consumer healthcare division, Opella, has received approval from the FDA to proceed with its plan to reclassify its erectile dysfunction medication, Cialis, as an over-the-counter product. This authorization aims to increase the availability of appropriately accurate and reliable OTC tests to meet public health needs and increase access to testing for consumers. This is further positively influencing the OTC consumer health market revenue.
*Definition - The term over-the-counter (OTC) refers to a medication that can be purchased without a medical prescription. In contrast, prescription drugs require a prescription from a doctor or other healthcare professional and should only be used by the prescribed individual. Some drugs may be legally classified as over-the-counter (i.e., no prescription is required), but may only be dispensed by a pharmacist after an assessment of the patient's needs or the provision of patient education. Regulations governing where drugs may be sold, who is authorized to dispense them, and whether a prescription is required vary greatly between countries.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients