The North America and Europe Acute Bacterial Skin and Skin Structure Infections (ABSSSI) market is anticipated to grow at a CAGR of 5.5% with USD 1,615.5 Mn in 2026 and is expected to reach USD 2,111.7 Mn in 2033. The major driver for the growth of the North America and Europe acute bacterial skin and skin structure infections (ABSSSI) market is research and development of products and novel treatments by pharmaceutical (U.S. pharmaceutical, USD 721.16 Bn in 2025, and Germany, USD 172 billion (EUR 159.4 billion)) and biopharmaceutical companies.
Oral & parenteral antibiotics is projected to account for the largest share of drug type in 2026, representing approximately 70% of the total volume. owing to their superior efficacy in treating severe as well as complicated ABSSSI cases that require systemic intervention.
The dominance of this segment is primarily attributed to the high prevalence of methicillin-resistant Staphylococcus aureus (MRSA) infections, which have become a major concern in both North American and European healthcare systems. Approximately 2% of the population carries MRSA. The societal economic burden of MRSA in the USA was estimated to range from USD 1.4 billion to USD13.8 billion.
According to the Centers for Disease Control and Prevention (CDC), MRSA accounts for a substantial proportion of skin and soft tissue infections in the United States, 5.4 million patients experienced 9.1 million SSTI episodes, with an incidence of 77.5 (95% confidence interval, 77.4–77.5) per 1000 person-years of observation (PYO), necessitating the use of potent systemic antibiotics like vancomycin and linezolid that can potentially penetrate deep tissue layers as well as achieve therapeutic concentrations at infection sites.
To learn more about this report, Request Free Sample
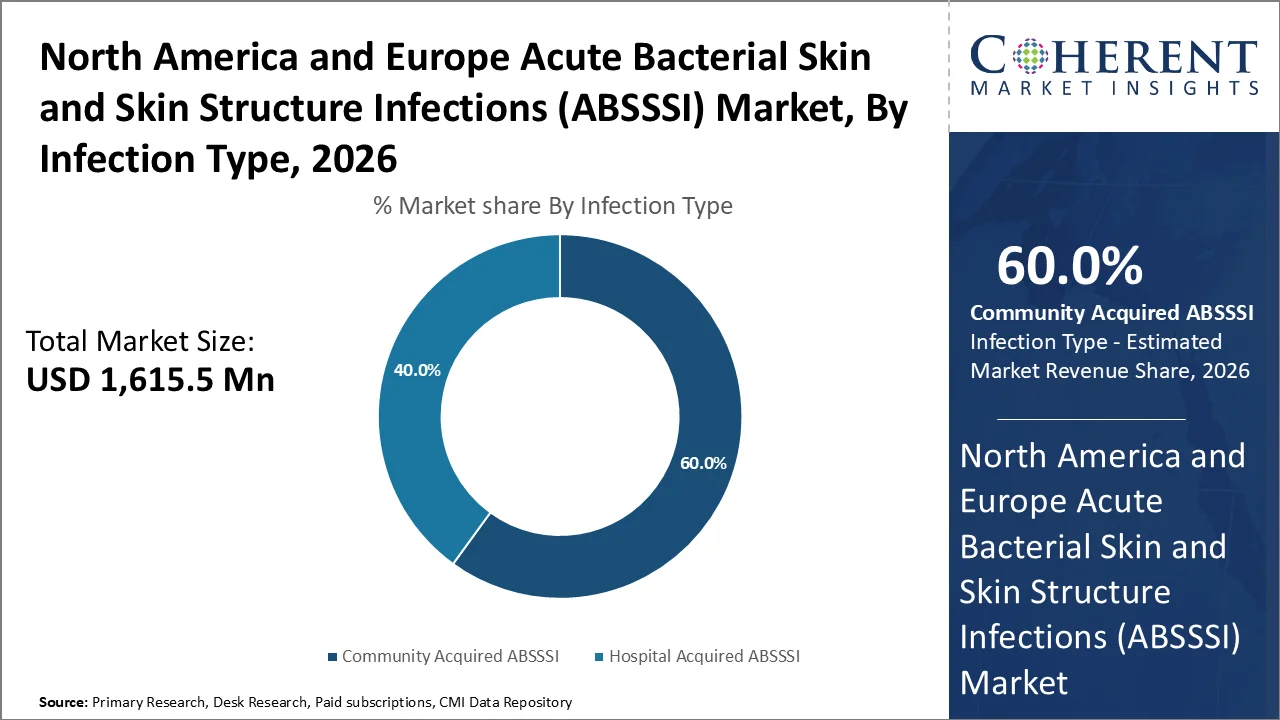
Based on infection type, Community Acquired ABSSSI dominates the market, accounting for a significant 60% share in 2026, owing to its significantly higher prevalence in the general population as well as the widespread nature of risk factors that predispose individuals to these infections outside healthcare settings.
The prevalence of Community Acquired ABSSSI is fundamentally driven by a high incidence of conditions that compromise skin integrity as well as immune function in the general population. Diabetes mellitus, which affects millions of individuals across North America and Europe, serves as a primary risk factor for community-acquired skin and soft tissue infections.
According to the International Diabetes Federation, the rising prevalence of diabetes creates a substantial population at risk for developing ABSSSI due to impaired wound healing, reduced peripheral circulation, and compromised immune responses. Diagnosed diabetes 29.1 million, estimated number of people with diagnosed diabetes in the United States, including 28.8 million adults aged ≥18 years.
Based on route of administration, parenteral dominate the market, accounting for a significant 45% share in 2026, owing to the critical nature of severe ABSSSI cases that require immediate as well as effective therapeutic intervention. The parenteral route, which includes intravenous as well as intramuscular administration, offers superior bioavailability and rapid onset of action compared to other delivery methods, making it the preferred choice for hospitalized patients and those with complicated infections. There are over 14 million cases of a common bacterial skin infection called cellulitis each year in the U.S. alone.
According to the Centers for Disease Control and Prevention (CDC), complicated skin and soft tissue infections usually involve deeper tissue layers, necrotizing fasciitis, or infections in immunocompromised patients, conditions that necessitate aggressive parenteral antibiotic therapy to prevent systemic complications and potential mortality.
Based on distribution channel, hospital pharmacies dominate the market, accounting for a significant 45% share in 2026, as ABSSSI treatments are very important and severe cases need quick medical help, hospital pharmacies are the main place where strong antibiotics are given, especially when patients need IV treatment or special monitoring. In 2025, retail, mail, long-term care, and specialty pharmacies collectively dispensed an estimated USD293.4 billion in specialty pharmaceuticals a 9.6% surge over the revised 2024 figure.
Hospital pharmacies are important because ABSSSI cases are often emergencies that need quick hospital care. The European Centre for Disease Prevention and Control says serious skin and tissue infections often need antibiotics given through a drip, which must be done safely in hospitals by medical staff.
Advanced diagnostic technologies, particularly molecular diagnostics and PCR-based systems, are transforming the detection of bacterial pathogens in acute bacterial skin and skin structure infections (ABSSSI). Real-time and multiplex PCR platforms can identify pathogens and resistance genes within 1–2 hours, compared to the 24–72 hours required for traditional culture methods. Leading systems include the Cepheid GeneXpert (used by Mayo Clinic, Johns Hopkins, and NHS trusts), bioMérieux FilmArray (Cleveland Clinic, Massachusetts General Hospital, Charité Berlin), and BD Max (Kaiser Permanente and Swedish healthcare system).
Adoption of these technologies majorly improves clinical outcomes as well as reduces costs. Hospitals report a 40–60% faster initiation of appropriate antibiotic therapy, a 25% reduction in patient length of stay, and savings of USD2,000–4,000 per ABSSSI episode. Additionally, using antibiotics the wrong way goes down by 30–45%, showing that fast molecular tests help both patients and healthcare work better.
Matrix-Assisted Laser Desorption/Ionization Time-of-Flight (MALDI-TOF) technology enables rapid bacterial identification using protein fingerprinting, dramatically shortening the time required for diagnosis. Key systems include the Bruker MALDI Biotyper, used at the University of Pennsylvania Health System, Toronto General Hospital, as well as major German university hospitals, and the bioMérieux VITEK MS, adopted by Intermountain Healthcare and French hospital networks.
The adoption of MALDI-TOF has majorly improved laboratory efficiency and clinical outcomes. Accuracy in bacterial identification reaches 85–95%, with identification times reduced from 24–48 hours to just 2–6 hours. This translates to a 20% improvement in appropriate empirical therapy selection and annual laboratory cost savings of USD150,000–300,000, highlighting both clinical as well as economic benefits.
|
Current Event |
Description and its Impact |
|
EU MDR 2024 Implementation Updates |
|
|
U.S. FDA Antimicrobial Stewardship Guidelines (2025) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
North America account 40% market share in 2026, owing to the robust healthcare infrastructure, advanced diagnostic capabilities, as well as well-established antibiotic development programs that have positioned the region as the dominant force in addressing Acute Bacterial Skin and Skin Structure Infections (ABSSSI). The Centers for Disease Control and Prevention (CDC) reports that skin and soft tissue infections account for approximately 14 million outpatient visits annually in the United States, with ABSSSI cases showing a major portion requiring intensive antibiotic therapy.
Hospital admissions for SSTI impose a substantial burden in the US, with SSTIs accounting for roughly 10% of all infection-related hospitalizations.
The region's leadership stems from its comprehensive approach to combating antibiotic-resistant pathogens, particularly methicillin-resistant Staphylococcus aureus (MRSA), which affects nearly 80,000 Americans annually according to CDC surveillance data.
The Europe region is poised to be the fastest-growing region through 2026-2033, driven by high domestic pharmaceutical innovation as well as strengthened regulatory frameworks that support local drug development initiatives. European pharmaceutical companies such as GSK, Sanofi, Novartis, etc., have targeted their efforts to develop region-specific ABSSSI treatments made for local resistance patterns. This focus is supported by the persistent prevalence of antibiotic-resistant infections in the EU.
In 2024, about 11 out of every 100,000 people in the EU had bloodstream infections from E. coli that resist third-generation cephalosporins. This is 5.9% higher than in 2019 and more than the 2030 goal of 9.38 per 100,000, showing that better treatments are still needed.
Owing to its extensive healthcare infrastructure, advanced medical research capabilities, and substantial healthcare expenditure allocation. The U.S. healthcare system's sophisticated diagnostic capabilities enable early identification as well as comprehensive treatment of ABSSSI cases, with the Centers for Disease Control and Prevention (CDC) reporting significant investment in antimicrobial resistance surveillance programs specifically targeting skin and soft tissue infections.
The presence of world-renowned medical institutions such as the Mayo Clinic, Cleveland Clinic, and Johns Hopkins has established the U.S. as a global leader in infectious disease management protocols. Recently, the FDA has made faster ways to approve new ABSSSI treatments. For example, some new medicines that fight MRSA infections were approved quickly.
Germany contributes the highest share in the market in Europe owing to its robust healthcare infrastructure, advanced medical research capabilities, as well as comprehensive antimicrobial stewardship programs that have positioned it as the leading segment for Acute Bacterial Skin and Skin Structure Infections (ABSSSI) treatment.
The German healthcare system's emphasis on evidence-based medicine and its well-established network of specialized dermatology centers have created an optimal environment for ABSSSI management. For example, the German Society for Dermatology (DDG) has implemented standardized treatment protocols that have resulted in improved patient outcomes as well as reduced hospital readmission rates.
Some of the major key players in North America and Europe Acute Bacterial Skin and Skin Structure Infections (ABSSSI), include Merck & Co., Inc., Pfizer Inc., GlaxoSmithKline plc, Teva Pharmaceutical Industries Ltd., Paratek Pharmaceuticals, Inc., Melinta Therapeutics, Inc., Arrevus, Inc., AbbVie Inc., Aurobindo Pharma Ltd., Amneal Pharmaceuticals LLC, and Sun Pharmaceutical Industries Ltd.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,615.5 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.5% | 2033 Value Projection: | USD 2,111.7 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Merck & Co., Inc., Pfizer Inc., GlaxoSmithKline plc, Teva Pharmaceutical Industries Ltd., Paratek Pharmaceuticals, Inc., Melinta Therapeutics, Inc., Arrevus, Inc., AbbVie Inc., Aurobindo Pharma Ltd., Amneal Pharmaceuticals LLC, and Sun Pharmaceutical Industries Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients