Peripheral intravenous catheters market is estimated to be valued at USD 9.41 Bn in 2026 and is expected to reach USD 16.65 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 8.5% from 2026 to 2033.
The global peripheral intravenous catheters (PIVC) market is witnessing steady growth, fuelled by rising hospital admissions, increasing surgical procedures, and the growing need for efficient vascular access devices. Short PIVCs dominate due to ease of use, lower complication rates, and widespread application in fluid and medication administration. Ongoing innovations in catheter safety, material design, and insertion techniques continue to drive market demand across various healthcare settings.
|
Current Event |
Description and its impact |
|
Product Innovation and Clinical Safety Enhancements |
|
|
Strategic Expansions and Industry Collaborations |
|
|
Regulatory Shifts and Healthcare Policy Updates |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Increasing incidence of major diseases such as cancer, cardiac disorders, orthopaedic disorders, etc. is expected to propel growth of the global peripheral intravenous catheter market over the forecast period.
The short peripheral intravenous catheters (Short PIVC) segment is projected to dominate the global PIVC market, capturing approximately 59.2% of the total market share in 2026. This segment’s leadership is driven by its widespread use in both emergency and routine medical settings due to ease of insertion, cost efficiency, and reduced complication rates.
Short PIVCs are primarily utilized for the administration of medications, fluids, and blood products, making them an essential component in hospital wards, outpatient departments, and ambulatory surgical centres. Their popularity is further supported by advancements in catheter design, such as the integration of blood control features, antimicrobial coatings, and improved biocompatible materials that enhance safety and comfort.
Additionally, the growing emphasis on infection prevention and the availability of user-friendly insertion tools are expected to drive continued adoption. These factors position short PIVCs as the most preferred choice among healthcare providers worldwide through and beyond 2025.
To learn more about this report, Request Free Sample
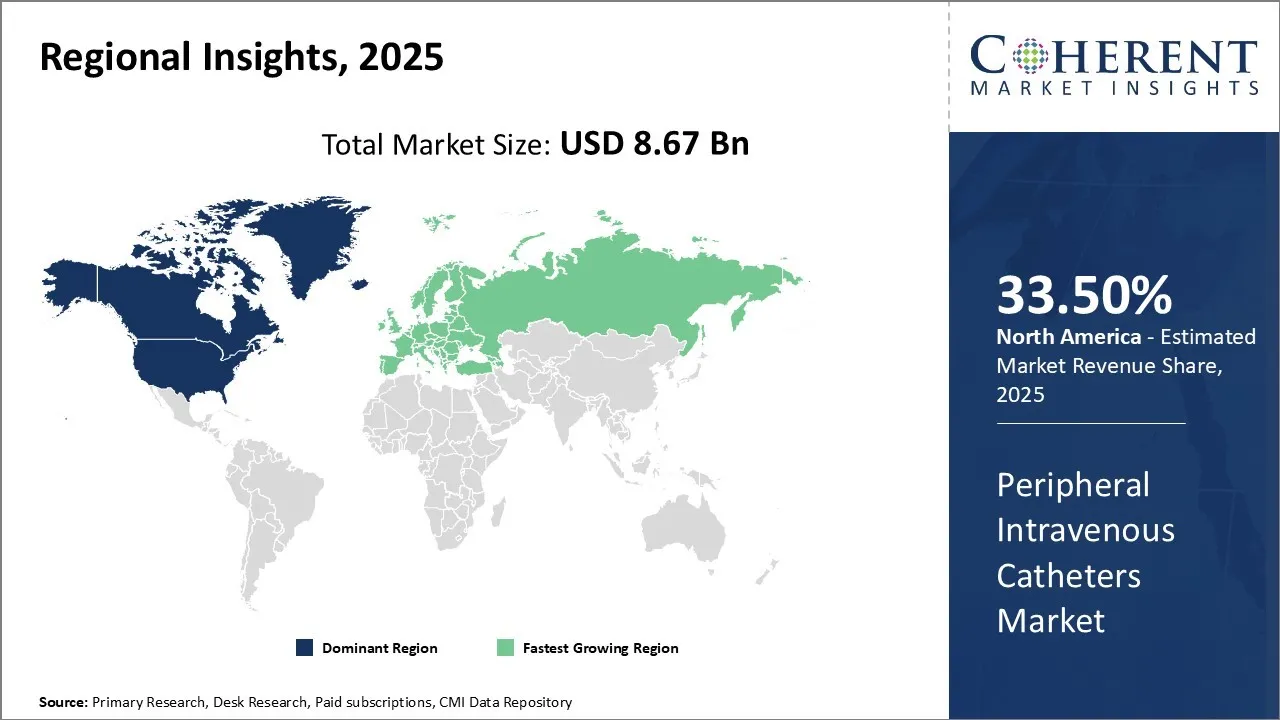
North America is projected to hold the largest regional share in the global peripheral intravenous catheters (PIVC) market, accounting for an estimated 33.50% of the total market in 2026. This regional dominance is supported by a combination of high hospitalization rates, advanced clinical practices, and a strong emphasis on patient safety and infection control.
The region boasts a well-developed healthcare infrastructure, with a widespread adoption of vascular access devices in both inpatient and outpatient settings. Technological advancements in catheter design such as safety-engineered tips, antimicrobial coatings, and improved insertion techniques further enhance clinical outcomes and reduce complications, contributing to the high demand for PIVCs.
Favourable reimbursement frameworks, coupled with continuous training programs for healthcare professionals, promote the use of advanced peripheral IV catheters across hospitals and ambulatory care centres. Additionally, the presence of leading medical device manufacturers and consistent R&D investments ensure that North America remains at the forefront of innovation and adoption in the PIVC market throughout the forecast period.
The United States dominates the global peripheral intravenous catheters (PIVC) market, significantly contributing to North America's projected 38.5% market share in 2026. This leadership is supported by a high volume of hospital admissions, an advanced healthcare system, and a strong regulatory focus on patient safety and infection control.
Germany is a key player in the European PIVC market, benefiting from its strong healthcare infrastructure, stringent quality standards, and high demand for advanced medical technologies. The country’s emphasis on clinical excellence and infection prevention drives adoption of safety-engineered PIVCs, particularly in surgical and intensive care settings.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 9.41 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.5% | 2033 Value Projection: | USD 16.65 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
BD, ICU Medical, Inc., Surmodics, Inc., B. Braun SE, Greiner Bio-One International GmbH, Terumo Corporation, Vygon, Healthline Medical Products, Retractable Technologies, Inc., Nipro Medical Corporation, Medsource Labs., Argon Medical Devices., Integral Lifesciences Holdings Corporation, Intra Special Catheters GmbH, Medical Components, Inc., Merit Medical Systems., Heka s.r.l., RAUMEDIC AG, Cook, Teleflex Incorporated |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients