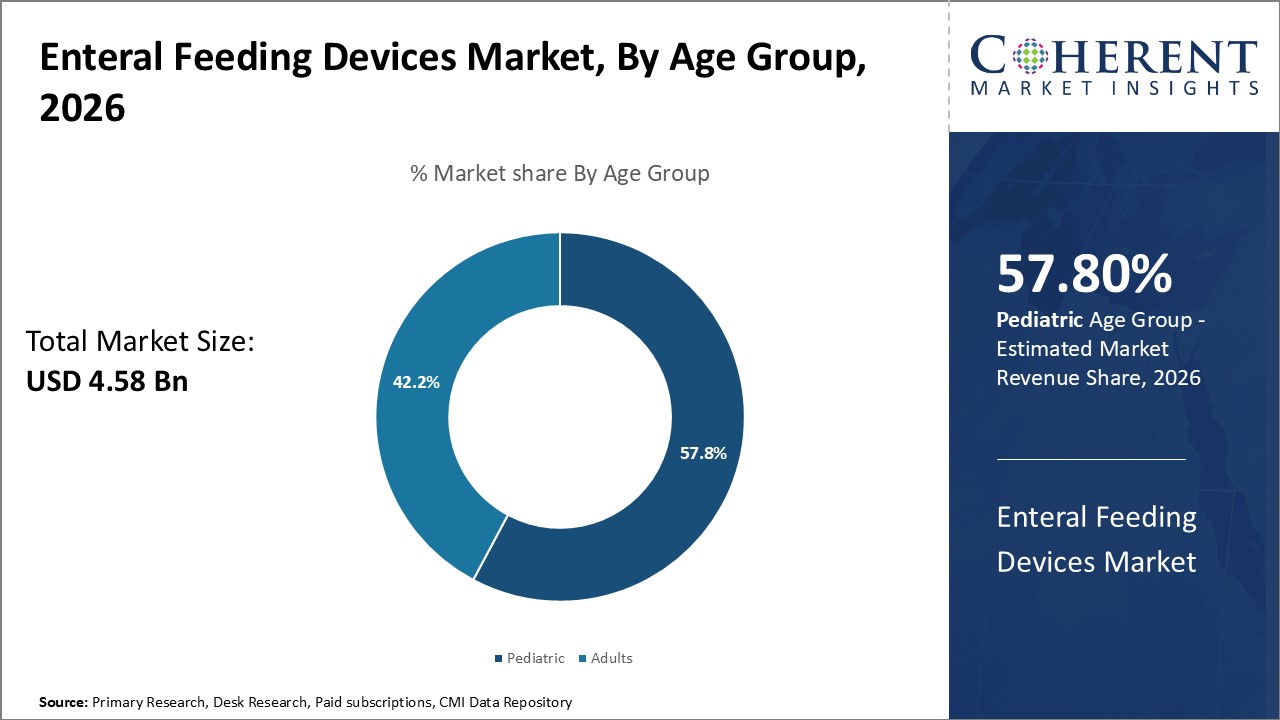
Enteral Feeding Devices Market is estimated to be valued at USD 4.58 Bn in 2026 and is expected to reach USD 7.17 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of6.6% from 2026 to 2033.
The enteral feeding devices market continues to grow as rising cases of chronic diseases, an aging population, and the need for nutritional support drive demand across healthcare settings. Healthcare providers increasingly use these devices to support patients who cannot eat orally, especially in hospitals, home care, and long-term care. Technological advancements and greater awareness of clinical nutrition further accelerate adoption. Strong healthcare infrastructure and supportive reimbursement policies in developed regions like North America and Europe also fuel market expansion.
|
Current Events |
Description and its impact |
|
Geopolitical and Regulatory Developments |
|
|
Technological and Product Innovation |
|
|
Economic Trends and Healthcare Spending |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Feeding Tubes hold the largest market share of 37.2% in 2026. The number of people with chronic illnesses like cancer, neurological disorders, and gastrointestinal ailments is still going up, which is why the enteral feeding devices market needs more feeding tubes. Healthcare professionals in hospitals and home care settings actively employ feeding tubes to give patients who can't eat orally the nutrition they need. Better tube design, more safety features, and more people knowing about the benefits of enteral nutrition are all reasons why more people are using it. Because of this, healthcare systems all across the world are using feeding tubes more and more.
To learn more about this report, Request Free Sample
The market for enteral feeding devices for kids is still growing because doctors are finding ways to help kids who were born too early, have congenital problems, or are having trouble developing. Doctors and other health care professionals actively employ enteral feeding to help babies and kids who can't eat normally develop and get better. Demand is driven by new technologies for children, more money spent on neonatal care, and easier access to specialist medical services. Also, more and more parents are realizing how important early nutritional support is, which is making the market grow even more.
For instance, Kate Farms®, the leader in plant-based medical nutrition, announced the early 2024 launch of its first whole food Pediatric Blended Meals, designed for tube feeding with resealable, direct-connect packaging.
Cancer therapies can make it hard to eat and swallow, therefore doctors often employ enteral feeding tubes to provide cancer patients the nutrients they need. Feeding tubes help patients stay strong and better handle chemotherapy, radiation, or surgery. As more and more focus is put on supportive and palliative care, professionals are using more and more specialized tools for cancer care. As more people learn about how nutrition affects recovery, hospitals and doctors are actively include enteral feeding in cancer treatment regimens to help patients get better faster.
Rockfield Medical Devices Limited, a commercial-stage company specializing in medical devices aimed at improving mobility and quality of life for tube-fed individuals, has announced that its Mobility+ Enteral Feeding System has received 510(k) clearance from the U.S. Food and Drug Administration (FDA).
Hospitals are a big part of the enteral feeding devices market since they actively treat patients with serious diseases that need nutritional support. In ICUs and surgical units, medical teams often utilize feeding tubes and pumps to help people recover after surgery, trauma, or a serious disease. Hospitals use innovative and reliable enteral feeding systems to improve patient outcomes. The use of enteral nutrition in hospitals is growing because they use qualified healthcare workers, follow updated clinical standards, and use high-quality technology.
To learn more about this report, Request Free Sample
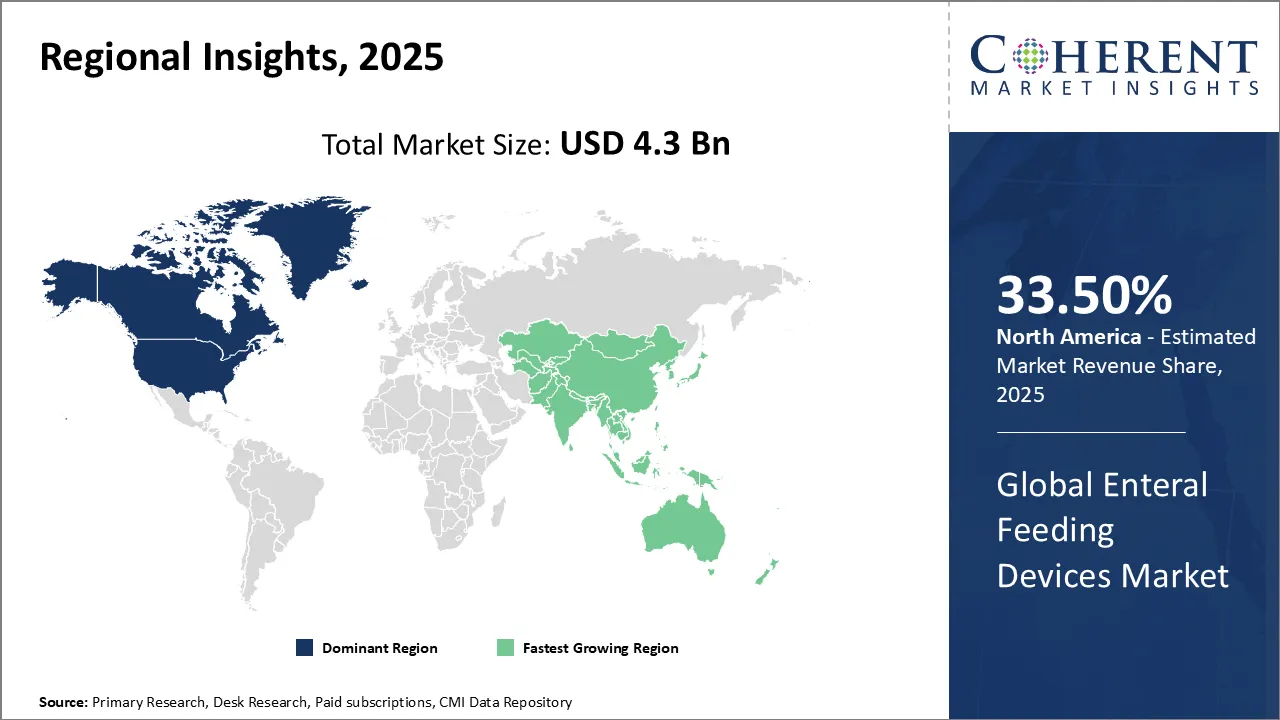
North America dominates the overall enteral feeding device market revenue with an estimated share of 33.5% in 2026. In North America, technological innovation and patient-centered care actively shape the enteral feeding devices market. Healthcare providers increasingly adopt smart feeding pumps and antimicrobial-coated tubes to boost safety and comfort. They also prioritize home-based enteral nutrition, supported by rising awareness and supportive reimbursement policies. The growing number of chronic disease cases and an aging population further drive demand. Manufacturers and healthcare systems collaborate closely to enhance device accessibility and performance, fueling market growth and improving outcomes for patients. For instance, Vonco Products LLC, a leading manufacturer of plastic and packaging solutions, announced that the U.S. Food and Drug Administration (FDA) has cleared EnteraLoc Flow as an over-the-counter (OTC) direct-connect feeding solution for enteral patients.
In Asia Pacific, expanding healthcare infrastructure and growing awareness about nutritional support actively drive the enteral feeding devices market. Hospitals and home care providers increasingly adopt advanced feeding devices to combat malnutrition and chronic diseases. Governments implement initiatives and improve reimbursement policies to increase device accessibility. Technological advancements and local manufacturing lower costs, encouraging broader use. The region’s rising elderly population, along with heightened attention to pediatric and oncology nutrition, also play key roles in boosting market growth.
In the United States, strong healthcare infrastructure and a focus on patient-centered nutrition actively advance the enteral feeding devices market demand. Healthcare providers increasingly implement innovative devices like smart pumps and low-profile tubes to enhance patient comfort and safety. Growing awareness of the benefits of enteral nutrition and supportive reimbursement policies encourage wider adoption in hospitals and home care. Rising chronic disease rates and an aging population further drive demand. Device manufacturers and healthcare professionals collaborate closely to promote market growth and improve patient outcomes. For instance, Cardinal Health (CAH) launched its next-generation Kangaroo OMNI enteral feeding pump in the United States, offering patients more options to support their personalized enteral feeding needs.
In India, expanding healthcare facilities and rising awareness of nutritional therapy actively drive growth in the enteral feeding devices market. Hospitals and clinics increasingly adopt advanced feeding solutions to address malnutrition and chronic diseases. Government support and better insurance coverage make these devices more affordable and accessible. Local manufacturers produce cost-effective options that promote wider usage. The growing elderly population and emphasis on pediatric care further increase demand, while continuous technological advancements improve device safety and efficiency nationwide. For instance, Vygon launched Nutrisafe2, an enteral feeding system designed for neonates, to ensure precise and safe feeding in the NICU. With this launch, the company tackles the critical issue of tubing misconnections—a common risk in neonatal care that has caused adverse events, including fatalities.
The market is witnessing rapid technological progress with innovations like smart feeding pumps that allow precise control of feeding rates and remote monitoring. Antimicrobial-coated tubes and low-profile gastrostomy devices enhance patient safety and comfort. These advancements reduce complications, improve usability, and facilitate integration with digital health platforms, making enteral feeding more efficient and adaptable to diverse patient needs in both clinical and home settings.
Home healthcare is becoming a preferred option due to patient comfort and cost-effectiveness, leading to increased use of portable and easy-to-manage enteral feeding devices. Providers are focusing on user-friendly designs that empower caregivers and patients to manage nutrition independently. Telehealth support and remote monitoring technologies complement home care, enabling timely interventions and improving adherence to feeding protocols outside traditional hospital environments.
The growing preference for home healthcare offers significant opportunities for enteral feeding device manufacturers. By developing portable, easy-to-use, and reliable devices, companies can cater to patients seeking comfortable, long-term nutritional support outside hospitals. Integration with telehealth and remote monitoring tools can enhance user experience and adherence, creating a lucrative market segment focused on home-based enteral nutrition solutions.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4.58 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.6% | 2033 Value Projection: | USD 7.17 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Cook Group, Abbott Laboratories, Cardinal Health, Inc., Boston Scientific Corporation, CONMED Corporation, Amsino International Inc., Applied Medical Technology, Inc., Becton, Dickinson and Company, B. Braun Melsungen AG, Fresenius SE & Co. KGaA, Moog, Inc., Vygon S.A, Dynarex Corporation, and Medela AG |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients