The global peripheral neuropathy treatment market is expected to grow from USD 2,071.9 Mn in 2026 to USD 2,564.7 Mn by 2033, registering a compound annual growth rate (CAGR) of 3.1%. The global peripheral neuropathy treatment market is poised for significant expansion, fueled by the rising prevalence of diabetes.
The International Diabetes Federation's 2025 Atlas estimates that roughly 589 million adults had diabetes in 2024. This number is expected to climb to approximately 853 million by 2050, which translates to one in eight adults – a 46% increase. This surge will considerably broaden the population vulnerable to diabetic peripheral neuropathy, thereby escalating the need for treatment.
(Source: International Diabetes Federation)
Diabetic peripheral neuropathy is projected to hold the market share of 45.2% in 2026, owing to high prevalence and strong causal link with diabetes. According to data from the Centers for Disease Control and Prevention and Clinical Studies, about half of all people with diabetes develop some form of nerve damage, with peripheral neuropathy being the most common type (Source: U.S. Centers for Disease Control and Prevention).
Additionally, the research published by National Library of Medicine, suggests that Diabetic Peripheral Neuropathy (DPN) accounts for nearly 75–90% of neuropathy cases among diabetic patients, and affects 10%–30% of individuals with prediabetes making it the leading indication for treatment. (Source: National Library of Medicine) The combination of this high disease burden, chronic nature, and need for symptom management results in continuous demand for drug-based and device-based treatments, thus ensuring that the treatment segment continues to dominate the market.
To learn more about this report, Request Free Sample
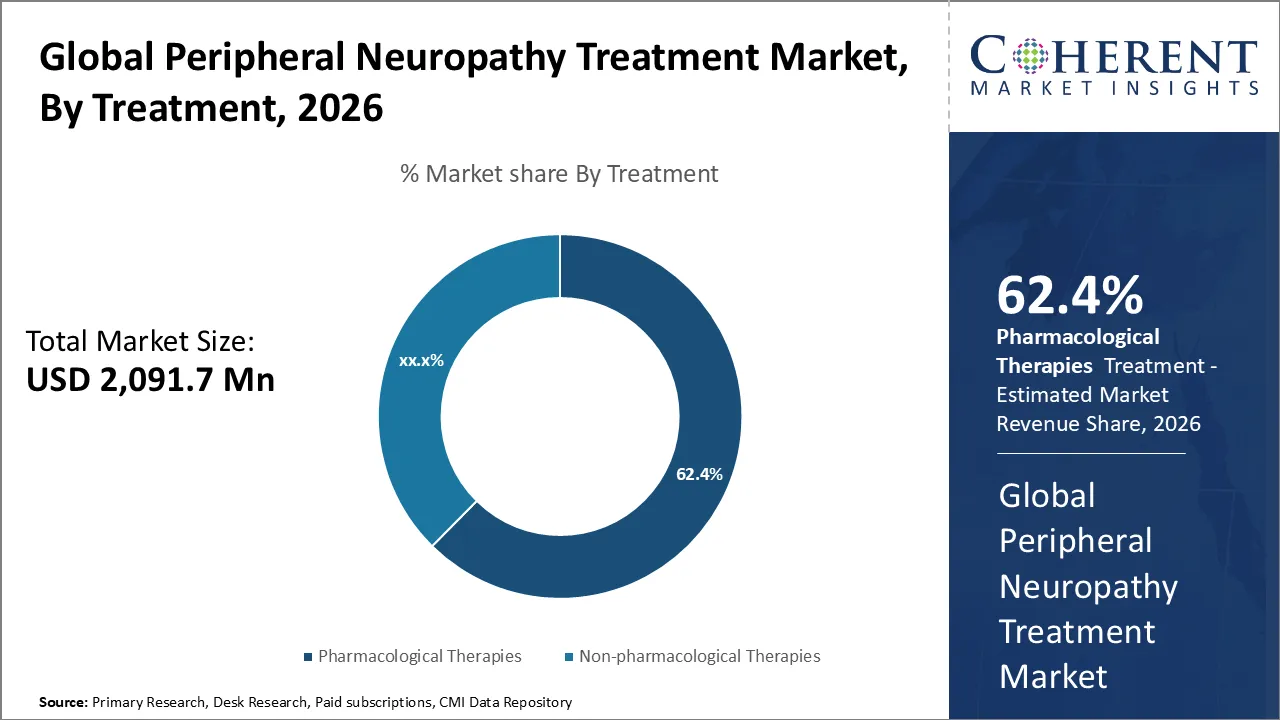
Pharmacological therapies are projected to hold a market share of 68.6% in 2026, due to the existence of evidence-based guidelines (e.g., American Academy of Neurology, National Institute for Health and Care Excellence, World Health Organization), evidence-based recommendation, and high availability. Pharmacologically based clinical studies supported by governments, like the National Institutes of Health (NIH), have found that the first approach to treat neuropathic pain involves the use of medications, specifically antidepressants and anticonvulsants.
A study published by National Library of Medicine in September 2024, showed that antidepressants and anticonvulsants still work effectively as the first approach for neuropathic pain treatment, in addition to evidence proving the effectiveness of gabapentin in managing diabetic neuropathy (Source: National Library of Medicine). Whereas additional evidence from clinical practice demonstrates the effectiveness of gabapentinoids like gabapentin in treating diabetic neuropathy, highlighting the continued reliance on pharmacological therapies globally. (Source: National Library of Medicine)
The hospitals segment is projected to hold a market share of 56.1% in 2026, owing to its capacity to deliver proper diagnosis, treatment, and holistic care for complicated cases of neuropathy. Hospitals are equipped with advanced medical technologies that aid in diagnosing patients through nerve conduction studies (motor nerve conduction velocity (MNCV), electromyography (EMG)) and other means). Moreover, hospitals offer services like neurology, pain clinic therapy, and other advanced forms of treatment. In addition, a high flow of patients for chronic conditions such as diabetes boosts hospital shares in the market.
|
Current Events |
Description and its Impact |
|
Increasing Burden of Chronic Diseases & Aging Population (Macro Shift – 2024–2025) |
|
|
Shift Toward Non-Opioid Therapies (Regulatory + Industry Shift – 2024–2025) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: World Health Organization, U.S. Food and Drug Administration)
To learn more about this report, Request Free Sample
To learn more about this report, Request Free Sample
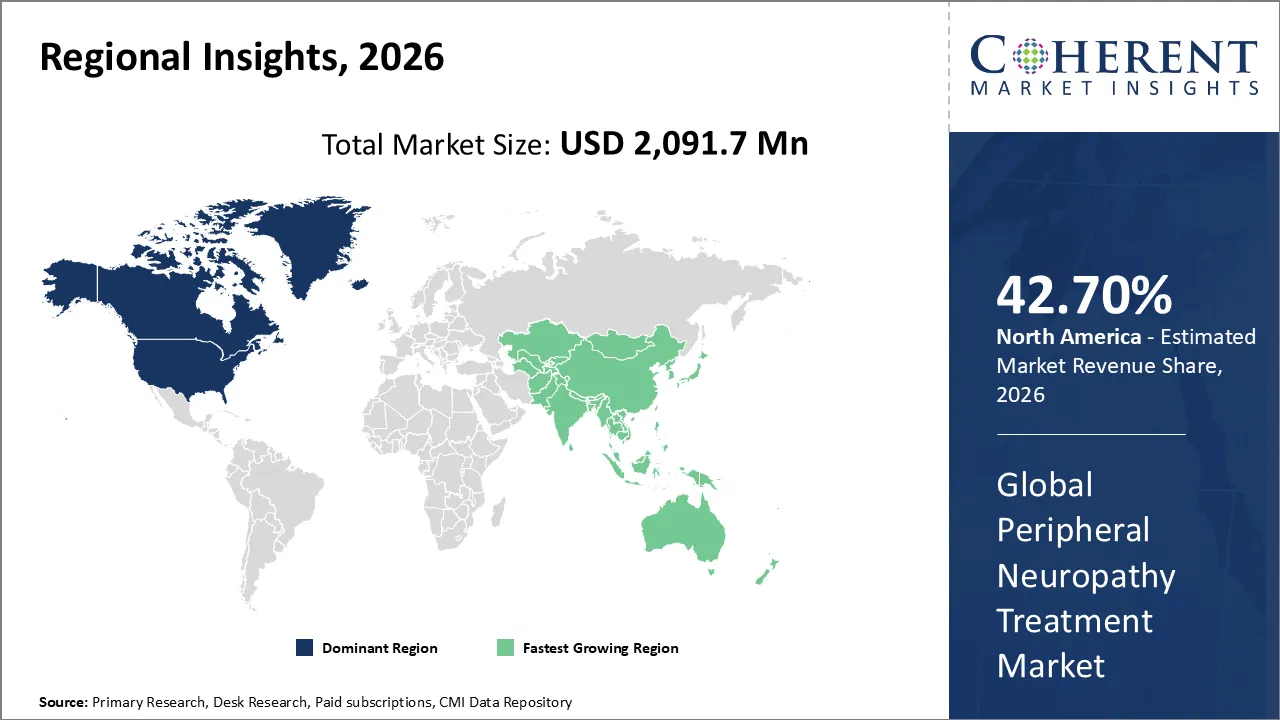
North America leads the global peripheral neuropathy treatment market, accounting for an estimated 41.1% share in 2026, due to the high disease burden and well-developed healthcare system. The Centers for Disease Control and Prevention estimates that more than 40 million individuals in the U.S. are living with diabetes (≈12% of the population), and about half develop nerve damage, with peripheral neuropathy being the most common form. (Source: Centers for Disease Control and Prevention)
Additionally, a study published by National Library of Medicine indicates that around 28% of adults with diabetes in the U.S. already have peripheral neuropathy, reflecting a substantial treatment population (Source: National Library of Medicine). The region's leading position in the peripheral neuropathy treatment market is significantly driven by the high prevalence of the condition, along with easy access to advanced diagnostic tools, reimbursement systems (Medicare, Medicaid, private insurers), and the quick adoption of new treatments.
The Asia Pacific peripheral neuropathy treatment market is expected to exhibit the fastest growth with an estimated contribution of 18.8% share to the global market in 2026, owing to the increasing prevalence of diabetes and related complications, as well as enhanced healthcare facilities in developing nations. The Asia Pacific region constitutes a major portion of the world’s diabetic population, and the number of people suffering from diabetes in the region stands in the hundreds of millions, leading to a vast patient pool susceptible to neuropathy.
The International Diabetes Federation reports that the number of adults with diabetes in South-East Asia grew from 34.9 million in 2000 to 106.9 million in 2024. By 2050, the number is projected to hit 184.5 million, significantly raising the number of individuals vulnerable to diabetic peripheral neuropathy (Source: International Diabetes Federation). Given that a substantial proportion of those with diabetes develop neuropathy, this swift uptick in the disease's incidence, coupled with improved healthcare access and the broader adoption of treatments, is driving robust demand for pharmaceutical interventions across the Asia Pacific region.
The U.S. is at the forefront of developing new innovations and adopting new peripheral neuropathy treatment approaches owing to its rich clinical research environment, quick regulatory approval process, and higher uptake of cutting-edge technologies. The existence of a broad number of drug and device companies in addition to investment in research for neurology (NIH budget reached ~USD 47.4 billion in 2024, with billions allocated toward neuroscience and neurological disorder research, including ~USD 2.8 billion for neurological disorders programs) and clinical trials leads to rapid development of new devices and drugs such as neuromodulation devices and non-opioid pain drugs. Further, the involvement of the U.S. FDA in speeding innovations through fast-track designation ensures that innovations reach the market rapidly, and awareness facilitates quicker uptake. (Source: Springer Nature Limited)
The peripheral neuropathy treatment market is expected to witness positive growth in the U.K. owing to robust healthcare system, high prevalence of the disease, and effective clinical management structures. Based on data provided by the National Health Service of the U.K., about 1 in 10 people aged 55 or over are affected by peripheral neuropathy. (Source: National Health Service)
Furthermore, diabetes is considered as the primary contributor to neuropathy, exhibits a high prevalence; research indicates that approximately 25% of diabetic individuals in the U.K. suffer from painful diabetic peripheral neuropathy, thereby underscoring a significant need for therapeutic interventions (Source: National Library of Medicine). The establishment of structured treatment protocols, elevated diagnostic rates, and broad availability of pharmacological treatments collectively reinforce the U.K.'s status as a well-established and developed market for peripheral neuropathy treatment.
China represents a major growth center for the market for peripheral neuropathy treatment owing to rapid growth in the diabetic patient base in the country coupled with an improved healthcare infrastructure and robust governmental support. Research published in January 2026, by ScienceDirect stated that the prevalence of diabetes in Chinese adults surged from just 2.5% in 1994 to 13.0% in 2021, affecting over 140 million adults and projected to reach 174 million by 2045, thus increasing the risk factor for developing diabetic peripheral neuropathy (Source: ScienceDirect). The government's "Healthy China 2030" plan, coupled with the increasing inclusion of sophisticated therapies in national reimbursement programs (National Reimbursement Drug List (NRDL), Urban Employee Basic Medical Insurance (UEBMI), Urban-Rural Resident Basic Medical Insurance (URRBMI)) is driving further growth.
Germany emerges as the dominant player within the European peripheral neuropathy treatment market owing to the high prevalence of patients, superior health care facilities, and huge spending on health care services (health expenditure reached ~12.7% of GDP, among the highest in Europe, with per capita spending exceeding USD 7,000), in the region. Being the most populated nation in Europe, Germany suffers from a considerable number of cases of Diabetes Mellitus, which is one of the main causative agents of neuropathy, resulting in a steady demand for treatment.
According to the national health research institutes and epidemiology, it is estimated that about 8.5 million individuals in Germany suffer from diabetes, while about 2 million people live with undiagnosed diabetes (Source: Deutsche Zentren der Gesundheitsforschung). Moreover, Germany is well-positioned as far as its reimbursement system and wide acceptance of guideline-based treatments are concerned (backed by the Federal Joint Committee and Robert Koch Institute). Together with the presence of established pharmaceutical firms operating within its territory and efficient distribution channels, Germany emerges as the dominant European market.
The peripheral neuropathy treatment market is experiencing growth in India owing to the fast-growing incidence of diabetes in India and the increasing healthcare services in the country. As per estimates by the World Health Organization, there are 77 million individuals in India suffering from diabetes, out of which 25 million are pre-diabetic, highlighting a significant population of people at risk of developing neuropathy in the country (Source: World Health Organization).
Government-sponsored research suggests that the prevalence of diabetic peripheral neuropathy in India varies tremendously, ranging between 9.6% and 78% of all cases of the disease (Source: Indian Journal of Medical Research). Additionally, with the help of national initiatives like NPCDCS (National Programme for Prevention and Control of Cancer, Diabetes, Cardiovascular Diseases and Stroke), India has been identified as one of the potential growth markets for peripheral neuropathy therapies.
|
Region |
Key Regulations |
Regulatory Overview |
Impact on Market |
|
North America |
U.S. Food and Drug Administration (FDA) – NDA/ANDA pathways, Controlled Substances Act (for certain pain drugs), HIPAA |
Highly structured regulatory framework with stringent clinical trial, safety, efficacy, and labeling requirements; strong oversight on opioid and CNS-active drugs |
Ensures high drug safety and efficacy standards, supporting innovation; however, strict approval timelines and compliance requirements increase development costs |
|
Europe |
European Medicines Agency (EMA), EU Clinical Trials Regulation (CTR), Pharmacovigilance Directive, GDPR |
Centralized and decentralized approval pathways across EU; strong emphasis on drug safety monitoring, real-world evidence, and data protection |
Facilitates access to multiple markets via centralized approvals; however, varying reimbursement policies and compliance complexity can impact commercialization |
|
Asia Pacific |
CDSCO (India), NMPA (China), PMDA (Japan), TGA (Australia) |
Diverse and evolving regulatory landscape with increasing harmonization toward global standards; growing focus on generics and biosimilars |
High growth potential due to faster generic approvals and large patient pool; regulatory variability and evolving policies may create entry challenges |
|
Latin America |
ANVISA (Brazil), COFEPRIS (Mexico), INVIMA (Colombia) |
Developing regulatory systems with improving clinical and manufacturing standards; increasing alignment with international guidelines |
Supports gradual market expansion and generics penetration; however, approval delays and inconsistent regulatory timelines may hinder rapid entry |
|
Middle East & Africa |
Saudi FDA (SFDA), South African Health Products Regulatory Authority (SAHPRA), UAE MOHAP |
Emerging regulatory frameworks with growing focus on quality standards, import regulations, and localization policies |
Creates opportunities for market entry and expansion; however, fragmented regulations and longer approval timelines may pose operational challenges |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The peripheral neuropathy treatment market is poised for considerable expansion, fueled by the emergence of new pipeline medicines and drug combinations that increase efficacy and meet unmet needs in the clinical space. The National Institutes of Health reports that the agency spends more than USD 48 billion every year on medical research, with a significant amount being devoted to neurodegenerative conditions and pain science to discover new treatments. (Source: National Institutes of Health)
Moreover, the U.S. FDA continues to promote the research and development of innovative non-opioid therapies through its regulatory initiatives (Non-Addictive Pain Treatment Development Program, Breakthrough Therapy Designation, Fast Track Designation). This favorable funding scenario and increasing pipeline have led to a fast-tracked development of therapies, opening up numerous growth prospects in the peripheral neuropathy drugs market.

To learn more about this report, Request Free Sample
The peripheral neuropathy treatment market is characterized by intense competition, largely fueled by the presence of generic medications. This has led to considerable price competition worldwide. As numerous therapies are no longer under patent, businesses are vying for an edge through cost-effectiveness, the ability to manufacture at scale, and robust distribution systems. Simultaneously, they are working to broaden their product offerings and make treatments more readily available. Key focus areas include:
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2,071.9 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 3.1% | 2033 Value Projection: | USD 2,564.7 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer Inc., Eli Lilly and Company, Novartis AG, Johnson & Johnson, GlaxoSmithKline plc, Teva Pharmaceutical Industries Ltd., Abbott Laboratories, AstraZeneca PLC, Sanofi, and Merck & Co., Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients