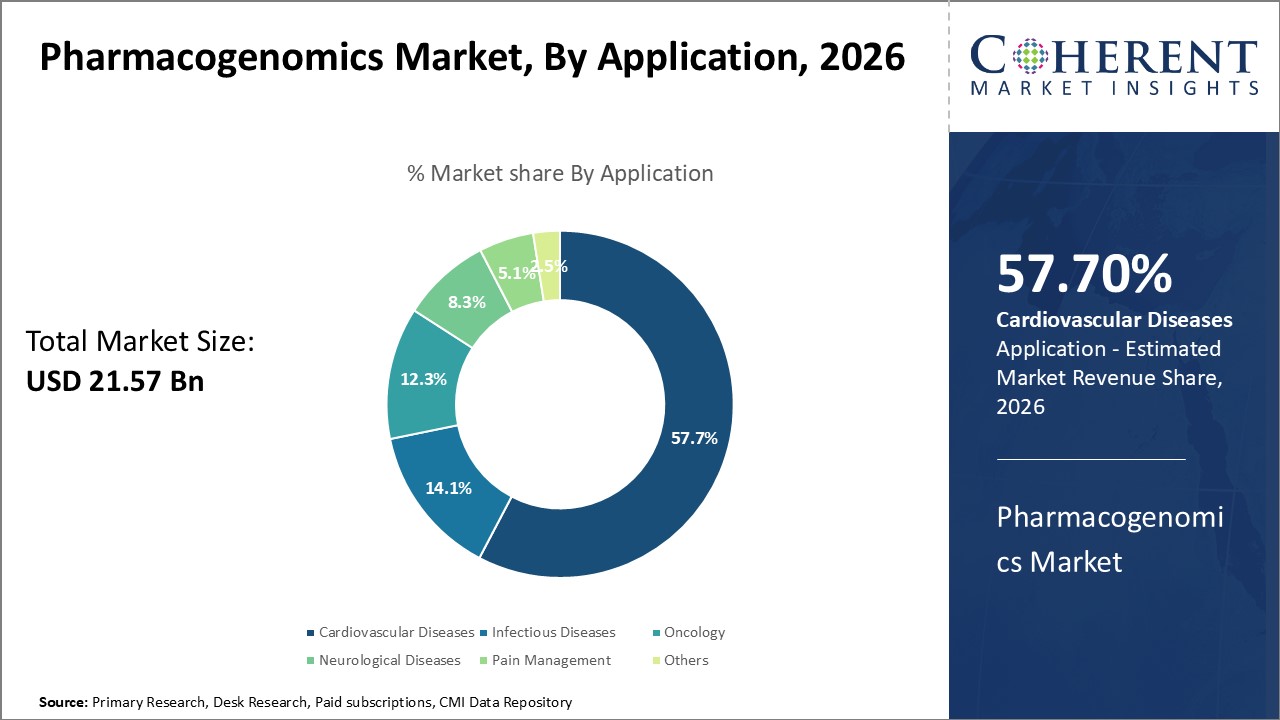
The global pharmacogenomics market size is estimated to be valued at USD 21.57 Bn in 2026 and is expected to reach USD 47.98 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 12.1% from 2026 to 2033.
To learn more about this report, Request Free Sample
The pharmacogenomics market demand is driven by factors such as rising prevalence of chronic diseases, growth in genomics application, and increased R&D investments in precision medicine. The pharmacogenomics market trends in the pharmacogenomics space has seen swift adoption of pharmacogenomic testing. There is a growing shift from trial and error to a more proactive approach that leverages genetic insights to determine the drug, dose, and duration of treatment tailored to individual patients. Vendors are focused on developing comprehensive genetic testing panels that can efficiently guide medication selection. The genomic approach is showing promise in improving outcomes and quality of life for patients with conditions like cancer, cardiovascular diseases, diabetes, and neurological disorders.
|
Event |
Description and Impact |
|
AI-Driven Drug Discovery Revolution |
|
|
Regulatory Framework Evolution for Personalized Medicine |
|
|
Healthcare System Integration and Reimbursement Shifts |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
AI and automation are transforming pharmacogenomics by enabling faster, data-driven personalization in drug development. Advanced AI models now analyze genomic datasets to predict drug responses, optimize clinical trial design, and identify new gene-drug associations. Automation tools streamline genotyping workflows with robotic pipetting and real-time PCR systems—reducing error rates and turnaround times. In clinical labs, machine learning enhances diagnostic accuracy by flagging rare genetic variants linked to drug metabolism, allowing for scalable, cost-efficient delivery of PGx testing across healthcare systems.
Pharmacogenomics market prices are influenced by factors such as test complexity, technology platforms, and regional reimbursement policies, reflecting a value-based pricing model. As of early 2025, single-gene PGx tests ranged between USD 250–450, while comprehensive multi-gene panels cost USD 800–1,500 depending on technology and provider.
For instance, in Q1 2025, U.S. hospital labs averaged USD 1,200/test, while clinics in India offered targeted panels from USD 300. Price drops were observed in South Korea (~USD 550) due to government subsidies. Automation and AI adoption are expected to further lower prices by 10–15% by 2027.
Clinics offering pre‑implantation genetic testing report a growing interest in integrating pharmacogenomic data to support personalized fertility treatments. However, many cite a lack of standardized PGx guidelines in reproductive medicine, making it difficult to confidently interpret results and integrate them into care protocols—hampering broader adoption.
Bio/pharma firms are actively leveraging PGx insights to streamline drug discovery and reduce adverse drug reactions. They value collaborations with biobanks and research institutes for access to genetic databases. However, reimbursement uncertainty and high testing costs remain primary concerns that limit investments in PGx-enabled trials.
Academic labs appreciate the ability to access comprehensive gene panels and next-generation sequencing platforms, enabling impactful research into gene–drug interactions. Yet, many researchers face shortages of skilled professionals and limited infrastructure, hindering scalability and translation into clinical practice.
Large-scale biobanks and research repositories report that PGx collaborations with pharmaceutical companies and academic partners are yielding new biomarkers and genotype–phenotype correlations. These initiatives support novel drug target discovery but are sometimes constrained by regulatory complexity around genetic data governance.
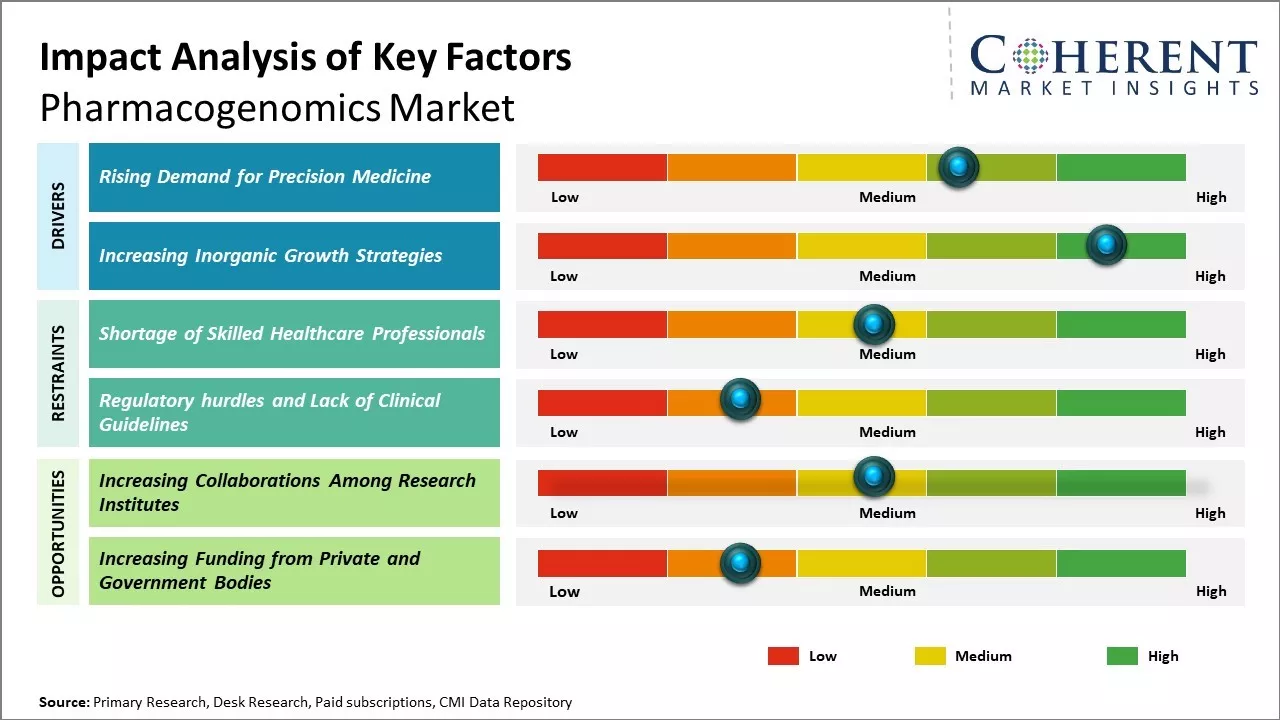
Surge in global incidence of cancer and increasing aging population susceptible to disease are expected to boost the demand of precision medicines, which is expected to drive the market growth over the forecast period. For instance, according to the data published by the American Cancer Society, in 2022, it was stated that over 1.9 million new cancer cases are expected to be diagnosed in the U.S. in 2023. Approximately, 609,820 deaths from cancer are expected in the U.S. in 2023, which is about 1,670 deaths per day.
Increasing inorganic growth strategies by key market players is expected to drive the market growth over the forecast period. For instance, in November 2021, Agena Bioscience, a provider of genetic testing solution, announced a partnership with Manchester University, to expand awareness and access to pharmacogenomics educational resources. The partnership enabled Agena Bioscience to promote and offer its customers, the established MU PGx programs including the Graduate Certification in PGx or Master of Science PGx degree.
Shortage of skilled healthcare professionals in performing pharmacogenomics diagnostic tests and lack of pharmacogenomics information in the electronic systems used by healthcare professionals are expected to hamper the market growth. For instance, an article published in Frontiers, a publisher of peer-reviewed open access scientific journal, in September 2021, healthcare professionals lack adequate training and education in pharmacogenomics and they are concerned about the absence of evidence-based clinical guidelines.
Lack of available resources, such as laboratory equipment, infrastructure requirements for pharmacogenomics, and skilled and highly trained healthcare professionals, also constitutes an important factor for the low integration level of pharmacogenomics.
Increasing adoption of inorganic growth strategies, such as collaborations, are expected to offer lucrative growth opportunities in the market. For instance, in August 2020, the University of California, San Francisco (UCSF), a health science university, expanded its pharmacogenomics efforts by launching a new collaboration with Tampa General Hospital and its pharmacy team that will allow clinicians to tailor medications based on a patient’s genetic makeup.
Plans include developing clinical decision support tools in the electronic health record (EHR) to support clinicians in interpreting results and guide treatment options in the presence of pharmacogenetic data.
The technology segment includes DNA sequencing, microarray, polymerase chain reaction, electrophoresis, mass spectrometry, and others. The DNA sequencing sub-segment is estimated to hold 40.9% of the market share in 2026 owing to its ability to rapidly decipher genetic information.
Next-generation sequencing technologies have accelerated scientific discovery by enabling researchers to sequence entire human genomes more quickly and cost-effectively than previous methods. This has supported the integration of genomic information into healthcare to guide treatment decisions.
DNA sequencing allows clinicians to match patients with therapies most suited to their genetic profile, improving outcomes while reducing trial-and-error prescribing. It also enables monitoring of disease progression and detection of mutations that may develop resistance to therapies.
Rapid DNA analyzers have automated sequencing workflows for clinical laboratories, streamlining pharmacogenomic testing to inform precision prescribing across diverse therapeutic areas. Continued technological advancements promise to make sequencing even faster, cheaper and easier to interpret, supporting wider pharmacogenomic applications.
To learn more about this report, Request Free Sample
The application segment includes cardiovascular diseases, infectious diseases, oncology, neurological diseases, pain management, and others. The oncology sub-segment is estimated to hold 57.7% of the market share in 2026 due to the growing burden of cancer. Pharmacogenomics helps optimize drug efficacy and minimize toxicity in oncology by accounting for patients' genetic variations.
Predictive analytics in pharmacogenomics leverages genetic testing to select therapies targeting cancer’s molecular drivers and identify biomarkers that predict treatment response or resistance. It allows pairing of patients with regimens most suited to their tumor profiles, rather than unguided trials. This precision approach improves outcomes for subgroups as therapies can be switched early in non-responders.
Genomics analysis also aids in identifying patients at high risk of drug-induced toxicity who may require dose reductions. Pharmacogenomics helps overcome challenges from tumor heterogeneity and evolution by tracking genetic changes through disease progression and treatment. It facilitates longitudinal monitoring to detect resistance mutations emerging with targeted therapies. These capabilities are central to realizing the promise of personalized cancer care.
The end user segment includes hospitals & clinics, research institutions and academic institutes, diagnostic laboratories, and others. The hospitals & clinics sub-segment is estimated to hold 33.4% of the market share in 2026 owing to their expanding clinical pharmacogenomics services.
Hospitals are adopting genomic technologies like next-generation sequencing and microarrays to establish precision medicine platforms. This allows embedding pharmacogenomic testing and consulting directly within clinical workflows to guide medication management for patients.
It helps optimize drug regimens from the outset of care. By automating genomic profiling, hospitals also provide seamless access to board-certified pharmacogenomic experts for busy clinicians. This consolidated model of testing and consultation enhances pharmacogenomics uptake by addressing barriers like interdisciplinary collaboration and complex genomic data interpretation challenges.
Expanding clinical decision support tools further aid healthcare professionals in applying test results. With pharmacological implications across therapeutic areas, hospital-based precision medicine services boost pharmacogenomics applications and associated economic outcomes across the care continuum.
To learn more about this report, Request Free Sample
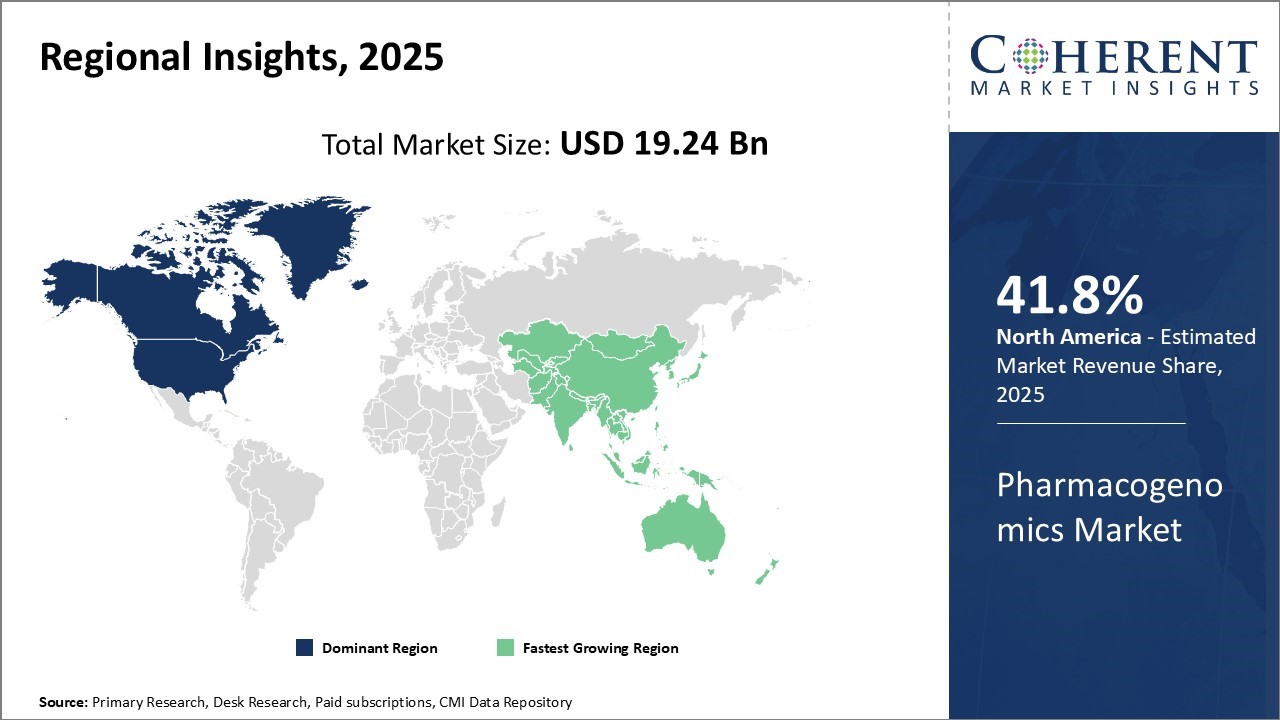
North America remains the dominant region in the global pharmacogenomics market and is estimated to hold 41.8% of the market share in 2026 due to the presence of major pharmaceutical companies and established biotechnology industry in the region. The U.S. is a hub for drug innovation and discovery activities. The presence of leading pharmaceutical companies with strong R&D capabilities allows them to invest heavily in pharmacogenomic research.
Additionally, supportive regulatory environment and increasing adoption of personalized medicine approach have further boosted the market growth in the region.
The Asia Pacific region has emerged as the fastest growing market for pharmacogenomics in recent years. Rapid economic development and growing healthcare investments in countries like China and India have created conducive environment for market growth. Rising patient population and increasing burden of chronic diseases have bolstered the demand.
Furthermore, improving access to healthcare along with increasing disposable incomes have augmented the demand for advanced diagnostic technologies in the region India and China’s low-cost operations are boosting the pharmacogenomics market in clinical trials, attracting global pharmaceutical players to invest in local research and manufacturing. This has provided major impetus to the regional market.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 21.57 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 12.1% | 2033 Value Projection: | USD 47.98 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Thermo Fisher Scientific Inc., F. Hoffmann-La Roche Ltd, Pharmigene, Abbott, Agilent Technologies, Inc., QIAGEN, GE HealthCare, Empire Genomics, Inc., Bio-Rad Laboratories, Inc., Oxford Nanopore Technologies plc., PerkinElmer Inc., Myriad Genetics, Inc., Illumina, Inc., Macrogen, Inc., BGI Genomics, Eurofins Scientific, MapMyGenome, and Invitae Corporation |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Pharmacogenomics is a branch of pharmacology that focuses on using DNA and amino acid sequence data to inform drug development and testing. It involves the study of how an individual's genetic inheritance affects the body's response to drugs, combining traditional pharmaceutical sciences with genetics and genomics to tailor pharmaceutical treatment to an individual. Pharmacogenomics aims to identify the most effective drug for a person, minimize side effects, and find the ideal dosage by using information about a person’s genomic make-up.
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients