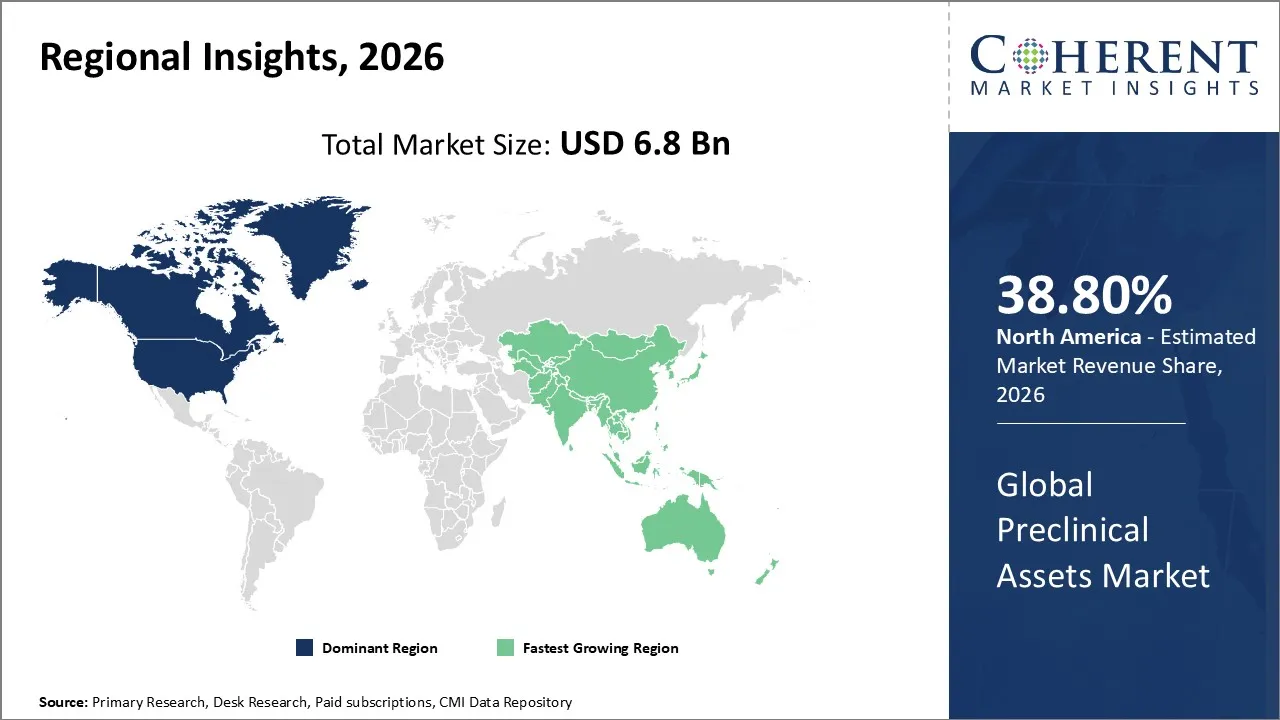
The Preclinical Assets Market is estimated to be valued at USD 6.8 Bn in 2026 and is expected to reach USD 9.2 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 8.6% from 2026 to 2033.
The preclinical assets market provides services, models, and technologies that support early-stage drug development by assessing safety, efficacy, and pharmacokinetics before clinical trials. It covers toxicology testing, patient-derived organoids, animal models, in vitro and in silico platforms, and contract research organization services. Pharmaceutical and biotech companies drive growth through increased R&D investment, adoption of advanced preclinical models, compliance with regulatory standards, and focus on personalized and precision medicine.
|
Current Events |
Description and its impact |
|
Regulatory and Policy Developments in Major Regions |
|
|
Geopolitical Tensions and Trade Policies |
|
|
Technological Advances in Preclinical Research |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Patient Derived Organoid (PDO) Model expected to hold the largest market share of 52.6% in 2026. The preclinical assets market increasingly adopts patient-derived organoid (PDO) models because they closely mimic human tissue architecture and genetic profiles, offering highly predictive insights into drug responses. Biopharmaceutical companies employ PDOs to assess efficacy and safety more effectively, advancing precision medicine efforts. Improvements in 3D culture techniques, biobanking, and integration with omics technologies strengthen their applications, while ethical concerns and the limitations of traditional models prompt researchers to rely on PDOs for personalized and translational studies. For instance, in April 2025, Tempus AI has launched Tempus Loop, a platform that leverages patient data—clinical records, test results, and treatment outcomes—along with patient-derived organoid (PDO) models to improve the identification and validation of cancer drug targets.
Toxicology Testing hold the largest market share of 42.2% in 2026. The preclinical assets market sees increasing demand for toxicology testing as companies aim to ensure drug safety and meet regulatory requirements before clinical trials. Pharmaceutical and biotechnology firms use these tests to detect potential adverse effects early, minimizing late-stage failures. Innovations in in vitro, in silico, and high-throughput screening improve predictive accuracy, while ethical concerns promote alternatives to animal testing. The expanding pipeline of novel therapies and complex biologics also drives the widespread adoption of comprehensive toxicology assessment platforms. For instance, in July 2025, Biocytogen Pharmaceuticals announced an upgrade to its preclinical division, expanding its portfolio of genetically engineered animal models and enhancing its services with efficacy studies, PK/PD analysis, biomarker evaluation, and non-GLP toxicology testing.
Biopharmaceutical Companies acquires the prominent market share of 38.8% in 2026. Biopharmaceutical companies expand the preclinical assets market by growing their R&D pipelines and implementing efficient methods to assess safety and efficacy before clinical trials. They rely more on specialized preclinical platforms and CROs to access advanced models, skilled expertise, and infrastructure, while cutting costs and speeding up development timelines. The development of complex biologics, cell and gene therapies, and personalized medicine further increases demand, as companies utilize predictive preclinical models and comprehensive testing platforms to drive innovation and meet regulatory requirements.
To learn more about this report, Download Free Sample
North America dominates the overall market with an estimated share of 38.80% in 2026. The North American preclinical assets market grows steadily, supported by advanced research infrastructure, a strong presence of pharmaceutical and biotechnology companies, and established contract research organizations. Companies in the region adopt innovative models, such as organoids, in silico platforms, and high-throughput screening, to enhance drug development efficiency. Regulatory backing, a skilled workforce, and collaborations between industry and academic institutions further drive expansion. The increasing focus on precision medicine, complex biologics, and personalized therapies shapes market trends and boosts demand for advanced preclinical assets. For instance, in January 2026, NAMSA, a global provider of medical device testing, clinical research, and regulatory consulting, announced that it has completed the acquisition of Labcorp’s Early Development medical device testing business.
The Asia Pacific preclinical assets market is growing as pharmaceutical and biotechnology companies ramp up R&D activities across the region. Improving research infrastructure, wider adoption of advanced preclinical models, and the expansion of contract research organizations drive this growth. Governments promote innovation through supportive regulations and funding initiatives. Companies increasingly implement organoids, in silico platforms, and high-throughput screening to streamline drug development. The rising focus on emerging therapies, personalized medicine, and strategic collaborations further boosts demand for advanced preclinical assets.
The United States preclinical assets market grows steadily as pharmaceutical and biotechnology companies expand operations and leverage well-established research infrastructure. Companies adopt innovative models, such as patient-derived organoids, in silico simulations, and high-throughput screening, to enhance drug development efficiency. Leading contract research organizations and a skilled scientific workforce enable faster and more reliable preclinical studies. Government regulations and initiatives promote innovation, while increasing focus on complex biologics, precision medicine, and personalized therapies drives greater demand for advanced preclinical assets nationwide.
The India preclinical assets market is growing as pharmaceutical and biotechnology companies ramp up early-stage research and drug development efforts. Companies implement advanced preclinical models, such as organoids, in vitro systems, and in silico platforms, to enhance efficiency and minimize development risks. Expanding research infrastructure, a larger pool of skilled scientists, and the rise of contract research organizations support this growth. Government initiatives and favorable regulations promote innovation, while increasing focus on personalized medicine and novel therapies boosts demand for advanced preclinical assets nationwide.
Companies increasingly use patient-derived organoids, 3D cell cultures, and in silico platforms to replicate human physiology more accurately. These models improve predictive outcomes, reduce late-stage failures, and accelerate drug development. The trend emphasizes integrating high-throughput screening and multi-omics data to enhance translational relevance and support personalized medicine initiatives.
Biopharmaceutical firms are outsourcing preclinical studies to CROs to access specialized expertise, advanced infrastructure, and scalable testing solutions. CROs enable faster execution of toxicology, pharmacology, and bioanalytical studies while reducing costs. Partnerships with CROs also facilitate global research collaboration and access to diverse preclinical assets, driving market growth.
Emerging models such as patient-derived organoids, organ-on-chip systems, 3D cultures, and in silico platforms offer highly predictive insights. There is an opportunity for companies to develop and commercialize these models, integrating automation, AI, and high-throughput screening to improve preclinical efficiency and reduce late-stage failures. Providing reliable, human-relevant platforms can address unmet needs in drug evaluation and precision medicine.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 6.8 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.6% | 2033 Value Projection: | USD 9.2 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Eurofins Scientific, ICON plc, WuXi AppTec, Viroclinics Xplore, Medpace, Inc., Charles River Laboratories., Pharmatest Services, PPD Inc., SGS SA (SGS), Intertek Group plc, Labcorp Drug Development, Laboratory Corporation of America, Inc., Crown Bioscience, Comparative Biosciences, Inc., TCG Lifesciences Private Limited., Shanghai Medicilon Inc., Domainex, Absorption Systems, AmplifyBio, and IQVIA |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients