The Herceptin Biosimilar market is anticipated to grow at a CAGR of 9.7% with USD 5.27 Bn in 2026 and is expected to reach USD 10.07 Bn in 2033. This strong growth trend is fueled by multiple factors, such as the rising incidence of HER2-positive cancers (1.2 million) especially breast and colorectal cancer and the expanding use of biosimilars, which offer a more cost-effective alternative to original biologic therapies.
Breast cancer is projected to account for the largest share of indication in 2026, representing approximately 70% of the total volume. As HER2-positive breast cancer is common globally and trastuzumab is proven effective, it is broadly used. According to the World Health Organization (WHO), reports about 2.3 million new breast cancer cases each year, making it the most diagnosed cancer among women globally.
The dominance of breast cancer in the Herceptin biosimilar market is further supported by the fact that approximately 15-20% of all breast cancer cases are HER2-positive, showing a major patient population requiring targeted therapy.
To learn more about this report, Request Free Sample
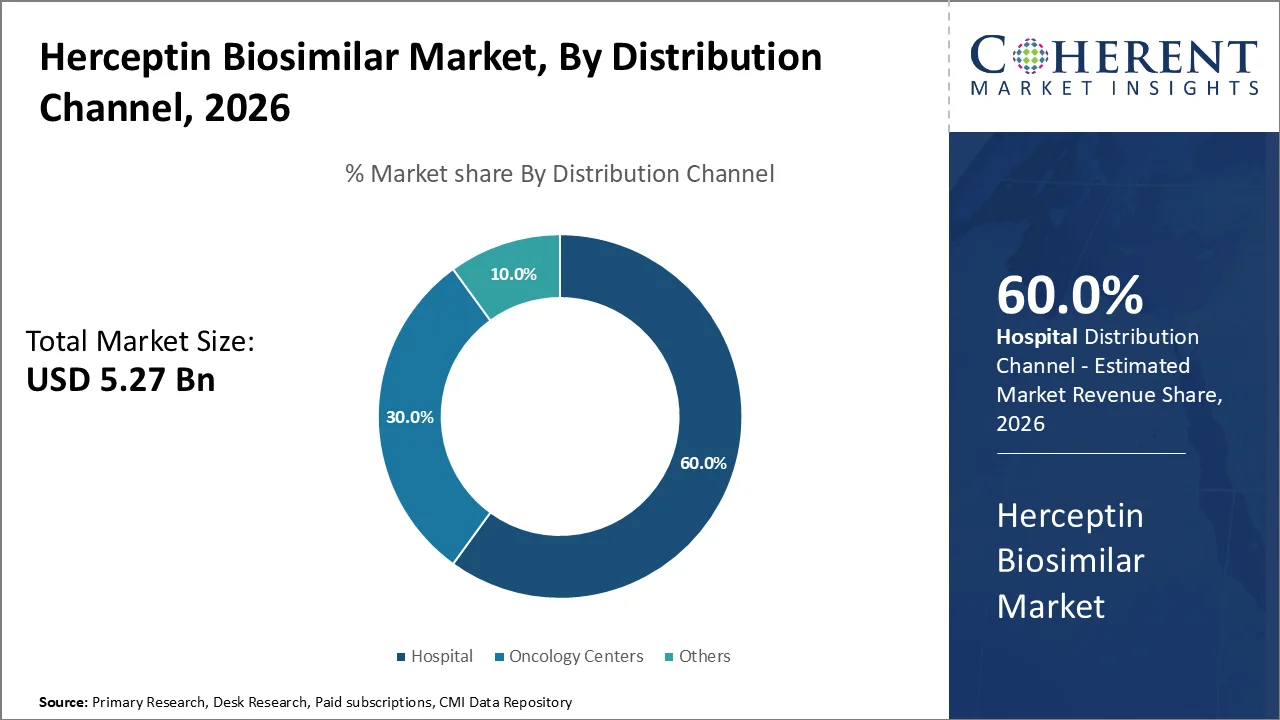
Based on distribution channel, hospital dominates the market, accounting for a significant 60% share in 2026, primarily owing to their established infrastructure, broad patient base, as well as overall treatment capabilities. Hospitals, particularly large tertiary care institutions, serve as primary points of care for cancer patients owing to their multidisciplinary approach, availability of advanced diagnostic tools, as well as access to various specialized therapies.
Tertiary II hospitals accounted for 81.7% of the total budget allocated by the Health Insurance Institute, with 69.3% of this funding directed to the University Medical Centre of Ljubljana, supporting the central role of well-resourced institutions in delivering comprehensive cancer care.
This means hospitals can easily use Herceptin biosimilars to treat HER2-positive breast cancer and other HER2-related tumors. For example, well-known hospitals like the Mayo Clinic and the National Cancer Institute (NCI) use biosimilars like Herceptin because they work as well as the original drug and cost less, helping more patients get the cancer care they need.
Advanced Chinese Hamster Ovary (CHO) cell line development remains the industry standard for trastuzumab biosimilar production, leveraging gene editing and metabolic engineering to optimize productivity and quality. Leading adopters include Celltrion (Herzuma®) with CHO-DG44 lines, Pfizer/Mylan (Ogivri®) using optimized glycosylation CHO cells, and Samsung Bioepis (Ontruzant®) employing proprietary CHO-K1 expression systems.
The adoption of these advanced CHO technologies has majorly improved manufacturing efficiency and product consistency. Trastuzumab biosimilar titers have increased to 3–5 g/L from 1–2 g/L in earlier systems, manufacturing costs have dropped by 30–40%, and critical quality attributes now achieve >95% similarity, ensuring reliable therapeutic performance.
Advanced transient gene expression platforms are increasingly used for rapid clone screening and early-stage process development. Key adopters include Amgen (Kanjinti), which uses ExpiCHO systems for accelerated clone selection, and Biocon/Mylan, which employs HEK293 transient systems for fasten early development.
These systems have majorly shortened development timelines by 6–12 months as well as allow rapid screening of over 1,000 clones compared with 100–200 in traditional methods, enabling faster identification of high-performing candidates.
|
Current Event |
Description and its Impact |
|
FDA Biosimilar Guidance Update (2025) |
|
|
European Commission Price Negotiation Policies (2026) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
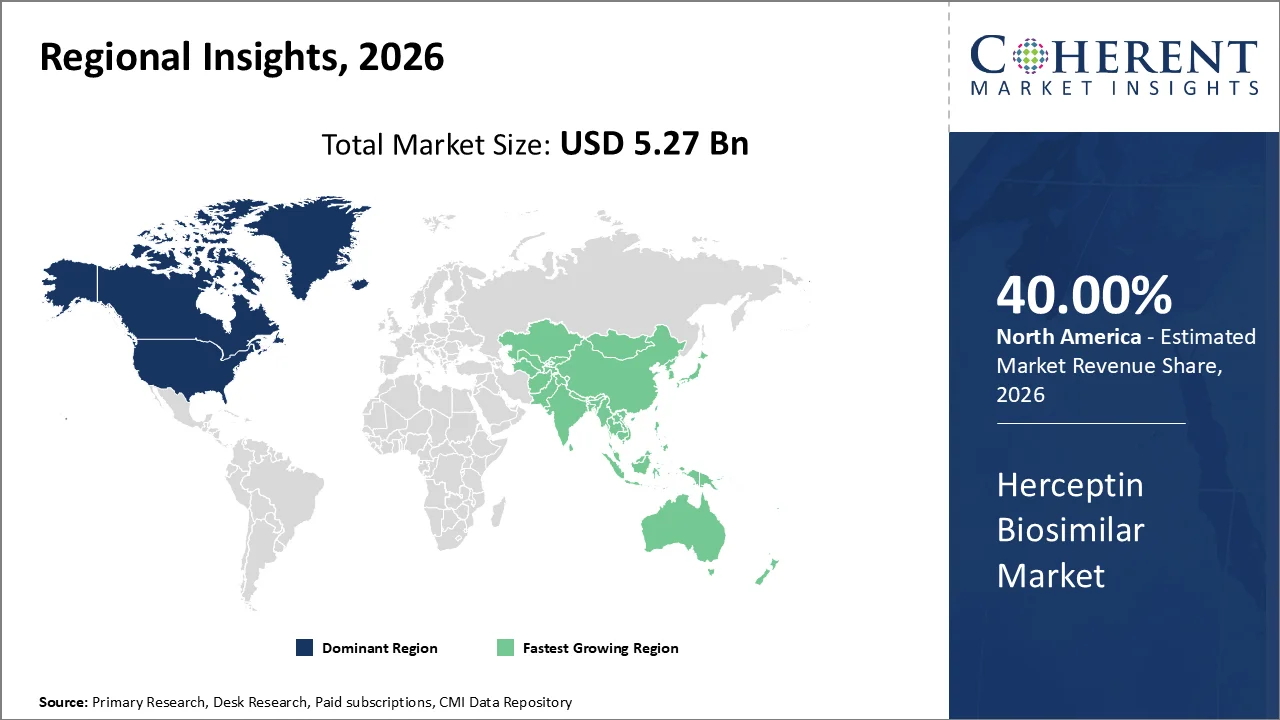
North America account 40% market share in 2026, owing primarily to the presence of a highly developed healthcare infrastructure, stringent regulatory frameworks, and an increasing prevalence of HER2-positive breast cancer which necessitates advanced therapeutic options such as biosimilars. In 2026, it is estimated that there will be 321,910 new cases of female breast cancer. The breast cancer subtype HR+/HER2- is the most common subtype with an age-adjusted rate of 92.9 new cases per 100,000 women, based on 2019–2023 cases.
For example, the U.S. Food and Drug Administration (FDA) has been highly proactive in creating streamlined regulatory pathways for biosimilars, leading to earlier market entry and broader acceptance among oncologists as well as healthcare providers. The FDA approved biosimilars like Ogivri and Herzuma, making cheaper alternatives to the original Herceptin available to more patients.
The Asia-Pacific region is poised to be the fastest-growing region through 2026-2033, expanding at a CAGR of approximately 7.5%. Countries such as India, China, South Korea, Japan, Australia, etc., have shown remarkable government and institutional support in biosimilar development through enhanced regulatory pathways as well as financial incentives. India’s Department of Biotechnology as well as China’s National Medical Products Administration have streamlined biosimilar approval procedures, which facilitates faster market entry for Herceptin biosimilars.
The National Biopharma Mission (NBM), a ₹1,500 crore joint initiative of the DBT and the World Bank. It focuses on accelerating the transition of biopharmaceuticals from discovery research to early-stage development. The mission is a major driver in achieving India’s vision of a USD150 billion biotechnology industry, fostering deep collaboration between industry as well as academia.
The World Health Organization (WHO) highlights the rising incidence of breast cancer in Asia Pacific, which has prompted multiple public health initiatives expanding access to advanced therapies, including biosimilars. Another major driver in Asia Pacific is the escalating number of local pharmaceutical companies investing heavily in biosimilar R&D, such as Biocon in India as well as Celltrion in South Korea.
the U.S. subsegment contributes the highest share in the Herceptin biosimilar market within the region primarily due to its advanced healthcare infrastructure, expansive oncology treatment centers, and well-established regulatory pathways that facilitate the entry and adoption of biosimilars.
The US Food and Drug Administration (FDA) has been proactive in streamlining biosimilar approvals, escalating physician and patient trust in biosimilar products such as Herceptin biosimilars.
Additionally, the U.S. has a large patient base affected by HER2-positive breast cancer, which is a key indication for trastuzumab (Herceptin). The high prevalence of breast cancer in the U.S., as reported by the American Cancer Society, affects approximately 280,000 women annually, making a considerable demand for targeted therapies, including biosimilars.
China contributes the highest share in the Herceptin biosimilar market in Asia Pacific owing to its robust regulatory framework as well as established manufacturing capabilities that have positioned the country as a biosimilar powerhouse. The National Medical Products Administration (NMPA) has streamlined approval processes for biosimilars, making a conducive environment for domestic and international manufacturers.
Shanghai Henlius Biotech's trastuzumab biosimilar, which received NMPA approval, exemplifies China's leadership in this space, demonstrating the country's capability to develop and commercialize complex oncology biosimilars.
The inclusion of biosimilar trastuzumab in China's National Reimbursement Drug List has significantly enhanced patient accessibility, fueling adoption across tier-one as well as tier-two cities. China's vast healthcare infrastructure, comprising over 35,000 hospitals as well as a escalating network of cancer treatment centers, provides extensive distribution channels for Herceptin biosimilars.
Some of the major key players in Herceptin Biosimilar market include, Amgen Inc., AryoGen Biopharma, Biocon Limited, Celltrion Inc., Pfizer Inc., Merck & Co., Inc., Accord Healthcare Ltd, Gedeon Richter Plc, Genor Biopharma Company Ltd, Mabion SA, Mylan N.V, Roche Holding AG and Samsungbioepis Co., Ltd.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 5.27 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.7% | 2033 Value Projection: | USD 10.07 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Amgen Inc., AryoGen Biopharma, Biocon Limited, Celltrion Inc., Pfizer Inc., Merck & Co., Inc., Accord Healthcare Ltd, Gedeon Richter Plc, Genor Biopharma Company Ltd, Mabion SA, Mylan N.V, Roche Holding AG and Samsungbioepis Co,.Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients