An immunodeficiency disease is caused when the immune system of the body fails to carry out its regular function of providing protection from various pathogens such as virus, bacteria, fungus, parasites, and others. These immunodeficiency diseases can be classified into primary and secondary immunodeficiency diseases. Primary immunodeficiency diseases are present in an individual at birth itself and are a result of gene mutations such as deletion of gene, duplication of gene and others. These genetic defects may lead to primary immunodeficiency diseases such as combined immunodeficiency disease, DiGeorge syndrome, hypogammaglobulinemia, Bruton’s disease, and others. Some of the factors responsible for causing secondary immunodeficiency disease include malnutrition, diabetes, radiation, severe burns, and others. Few examples of secondary immunodeficiency diseases include HIV-AIDS, leukemia, viral hepatitis, multiple myeloma, and others. Immunodeficiency diseases can be treated using antibiotic therapy, immunoglobulin therapy, anti-viral drugs, stem cell, and gene therapy.
Global Immunodeficiency Therapeutics Market - Impact of the Coronavirus (COVID-19) Pandemic
Coronavirus (COVID-19) outbreak was first reported on December 31, 2019, in Wuhan, China. The World Health Organization declared COVID-19, as pandemic on March 11, 2020.
According to the Coronavirus (COVID-19) Weekly Epidemiological Update by the World Health Organization, over 186,240,393 cases and 4,027,861 deaths due to Coronavirus disease (COVID-19) were reported till July 11, 2021, across the globe.
Impact of COVID-19 on Demand and Supply of Immunodeficiency Therapeutics
The COVID-19 pandemic and lockdown in various countries across the globe have impacted the financial status of businesses across all sectors. The pharmaceutical sector is one such sector which has been majorly impacted by the pandemic.
The COVID-19 pandemic has impacted the entire supply chain of the pharmaceutical industry mainly due to strict lockdown in several regions. However, the demand for immunodeficiency therapeutics is expected to increase in COVID-19 pandemic owing to association of immunodeficiency diseases with COVID-19. For instance, according to an article published by the Centers for Disease Control and Prevention (CDC) in May 2021, people suffering from immunodeficiency disorders such as HIV-AIDS and primary immunodeficiency disease are more likely to get infected with severe COVID-19 since their immune functions are suppressed due to the immunodeficiency diseases. Thus, impact of the Coronavirus (COVID-19) pandemic is expected to bolster the growth of the global immunodeficiency therapeutics market during the forecast period.
The global immunodeficiency therapeutics market is estimated to be valued at US$ 6,991.1 Mn in 2021, and is expected to exhibit a CAGR of 6.1% over the forecast period (2021-2028).
Immunodeficiency Therapeutics Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2020 | Market Size in 2021: | US$ 6,991.1 Mn |
| Historical Data for: | 2017 to 2020 | Forecast Period: | 2021 to 2028 |
| Forecast Period 2021 to 2028 CAGR: | 6.1% | 2028 Value Projection: | US$ 10,581.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer Inc., CSL Behring, Grifols, S.A., Abbott, ADMA Biologics, Inc., AstraZeneca, Baxter, Bayer AG, Biocon, Bristol-Myers Squibb Company, BDI Pharma, Inc., Bharat Serums and Vaccines Ltd., Eli Lilly and Company, GlaxoSmithKline plc., Lupin Pharmaceuticals Inc., Novartis AG, Octapharma AG, Sanofi, and Takeda Pharmaceutical Company Limited |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Figure 1: Global Immunodeficiency Therapeutics Market Share (%) Analysis, By Therapy Type, 2021
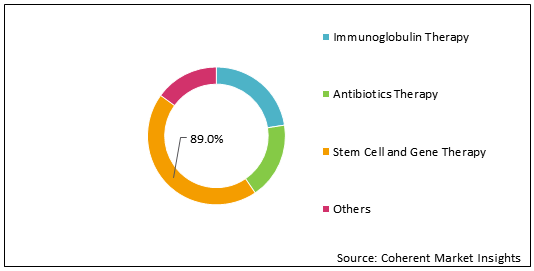
To learn more about this report, Request Free Sample
The rising cases of immunodeficiency diseases is the major factor that is expected to drive the global immunodeficiency therapeutics market growth over the forecast period.
The rising prevalence of immunodeficiency disease is expected to drive growth of global immunodeficiency therapeutics market over the forecast period. For instance, according to an article published by the National Institutes for Health (NIH), U.S. in May 2021, the prevalence of ADA-SCID (Adenosine deaminase-severe combined immunodeficiency syndrome) is estimated to be one in 200,000 to 1,000,000 newborns across the world. Moreover, according to the data published by the World Health Organization (WHO) in November 2020, around 1.5 million new human immunodeficiency virus (HIV) infections were reported globally in 2020 and 37.6 million are reported to be suffering from HIV worldwide in 2020. HIV causes acquired immunodeficiency disorder syndrome (AIDS).
Moreover, increasing number of research and development activities for improving the effectiveness of immunodeficiency therapeutics is expected to aid in the growth of the market over the forecast period. For instance, according to an article published by National Institute of Health (NIH) in May 2021, an investigational research study conducted in in U.S. and U.K. and supported by NIH showed that., 96% of children receiving new experimental lentiviral gene therapy for immunodeficiency diseases reported restored immune function.
The rising advancements of technology in the field of genetics is expected to drive the growth of the global immunodeficiency therapeutics market over the forecast period. For instance, according to an article published in the Genes & Diseases international scientific journal in March 2020, recent technological advancements such as next generation sequencing (NGS) have enabled researchers to explore newer genetic variations related to common variable immunodeficiency (CVID) in order to improve its diagnosis.
Global Immunodeficiency Therapeutics Market – Restraints
However, growth of the immunodeficiency therapeutics market may be hampered, owing to high cost of immunotherapy and side effects of immunodeficiency disease treatment. For instance, according to an article published by ScienceDirect in April 2020, the total cost of immunodeficiency disease treatment is estimated to be US$ 6,500 to US$ 108,463 per patient.
Global Immunodeficiency Therapeutics Market – Regional Analysis
On the basis of region, the global immunodeficiency therapeutics market is segmented into North America, Latin America, Europe, Asia Pacific, Middle East, and Africa.
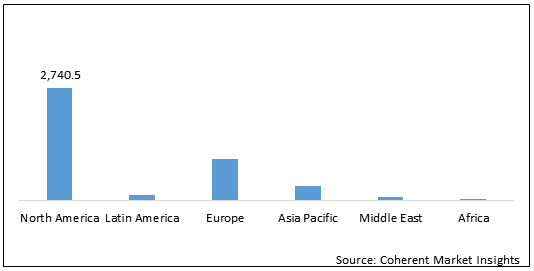
North America is expected to hold dominant position in the global immunodeficiency therapeutics market over the forecast period, owing to rising number of product approvals. For instance, in September 2020, Octapharma USA Inc., a biopharmaceutical company received Medicare Part B approval for its product Cutaquig which is used for treatment of humoral immunodeficiency in adults and children. Medicare is an insurance provider which covers the treatment costs related to the Medicare part B listed diseases. Such approvals from insurance providers is expected to encourage people to opt for immunotherapy and thus drive the growth of global immunodeficiency therapeutics market over the forecast period.
Figure 2: Global Immunodeficiency Therapeutics Market Value (US$ Mn), by Region, 2021
To learn more about this report, Request Free Sample
Global Immunodeficiency Therapeutics Market – Competitive Landscape
Major players operating in the global immunodeficiency therapeutics market include Pfizer Inc., CSL Behring, Grifols, S.A., Abbott, ADMA Biologics, Inc., AstraZeneca, Baxter, Bayer AG, Biocon, Bristol-Myers Squibb Company, BDI Pharma, Inc., Bharat Serums and Vaccines Ltd., Eli Lilly and Company, GlaxoSmithKline plc., Lupin Pharmaceuticals Inc., Novartis AG, Octapharma AG, Sanofi, and Takeda Pharmaceutical Company Limited
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients