The pyrazinamide market size is anticipated to grow at a CAGR of 10.9% with USD 7.53 Bn in 2026 and is expected to reach USD 17.23 Bn in 2033. The market is growing with the rising global tuberculosis (TB) incidence and increased use of first‑line anti‑TB drugs. In 2023, the United States reported 9,633 TB cases, a 15.6% increase from 2022, with an incidence rate of 2.9 cases per 100,000 persons, highlighting ongoing TB challenges and demand for effective treatments.
On the basis of patient type, the adult segment accounts for the largest pyrazinamide market share of 70.10% in 2026. The segment’s growth is driven by the higher incidence of active tuberculosis (TB) disease in adults, who represent the majority of global TB cases.
According to the World Health Organization’s Global Tuberculosis Report 2024 data, the adults aged more than 15 years accounted for an estimated 88% of total TB incidence in 2023 in comparison with 12% in children aged 0-14 years. This age distribution demonstrates the greater disease prevalence amongst adults. They are more likely to develop active disease and also require full course combination therapy like pyrazinamide.
In the United States, provisional CDC data for 2025 indicated that total tuberculosis (TB) case counts remained high, with 10,260 cases reported, the majority of which occurred in adults. This highlights the ongoing clinical need for TB treatment regimens specifically targeting adult populations.
To learn more about this report, Request Free Sample
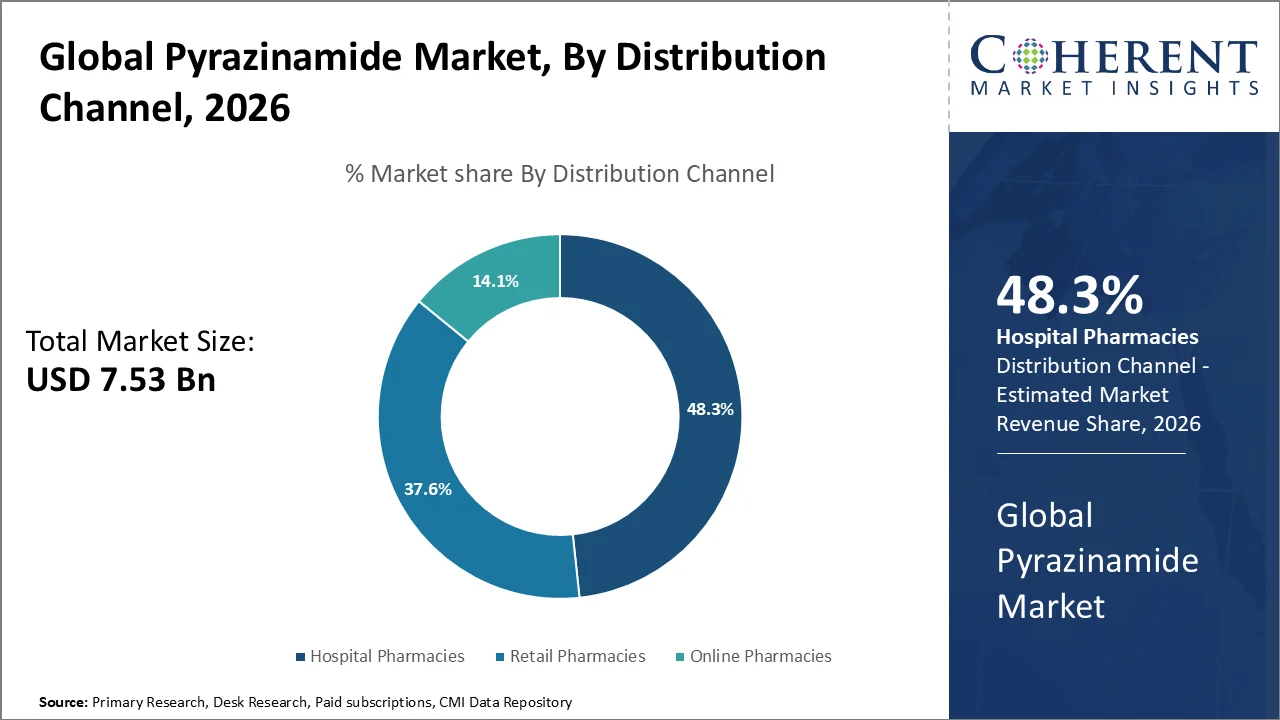
By distribution channel, the hospital pharmacies segment accounts for the largest pyrazinamide market share of 48.30%in 2026. The hospital pharmacies are the main source for dispensing pyrazinamide to patients diagnosed with tuberculosis.
The segment’s growth is owing to the increasing global burden of tuberculosis, particularly in low- and middle-income countries where hospital pharmacies are the primary point of access for essential medicines. The rise in hospital admissions due to severe cases of multi-drug-resistant tuberculosis (MDR-TB) as well as the greater availability of affordable medications is also contributing to the segment’s growth.
According to the World Health Organization (WHO) Global Tuberculosis Report 2025, nearly 10.7 million people across the globe were diagnosed with tuberculosis (TB) in 2024. This marks a substantial increase in drug-resistant cases. In addition, the Centers for Disease Control and Prevention (CDC) reported in 2023 that there were 9,633 documented cases of active tuberculosis in the United States, with a considerable number of patients receiving treatment in hospital pharmacies. This trend has led to a heightened demand for pyrazinamide in hospital pharmacies.
The segment’s leadership is also linked to the government healthcare programs aimed at combating TB, like the U.S. National TB Program, which allocates funds for the procurement of essential TB medicines including Pyrazinamide.
The demand for TB-related therapies such as pyrazinamide is increasing in the United States owing to a surge in reported TB disease cases in 2023. The CDC documented 9,633 cases of TB in the U.S. during this year, thereby resulting in an incidence rate of 2.9 per 100,000 persons. This is the highest case count since 2013 and the highest incidence rate since 2016, with both figures increasing about 15% in comparison with 2022.
This resurgence is widely reflected across reporting areas. California, Texas, New York, and Florida together accounted for over half of all TB cases, thus underscoring regional burdens driving clinical demand. Pyrazinamide, a key first‑line anti‑TB agent alongside isoniazid and rifampicin, remains central to U.S. treatment regimens recommended by CDC and the U.S. Preventive Services Task Force, particularly in combination therapy for drug‑susceptible TB.
The procurement and use of pyrazinamide are directly influenced by public health priorities and funding aimed at TB control and elimination as healthcare systems respond to the rising incidence and intensified screening efforts.
A considerable advancement in the pyrazinamide market is the improved understanding of molecular resistance mechanisms. According to U.S. government surveillance, tuberculosis (TB) case counts in the United States reached 9,633 in 2023, with an incidence rate of 2.9 per 100,000 people, thereby indicating an increase compared to 2022. Although still rare, drug-resistant TB cases were reported, with 589 cases (8.5%) showing resistance to at least isoniazid and 100 cases (1.4%) classified as multidrug-resistant (MDR TB) in 2023.
The effectiveness of pyrazinamide is closely linked to the pncA gene, which encodes the enzyme required to activate the drug. The mutations in this gene are the primary mechanism of pyrazinamide resistance in Mycobacterium tuberculosis. This molecular insight is pivoting treatment strategies. The clinicians are progressively using comprehensive susceptibility testing (including genetic pncA analysis) to tailor regimens and mitigate resistance risk. Together with CDC data showing MDR TB remains largely uncommon, these advances support optimized use of pyrazinamide in combination therapies that shorten and strengthen treatment courses for drug‑susceptible and some resistant TB cases in the U.S. regime.
|
Current Event |
Description and its Impact |
|
WHO Global Tuberculosis Report 2025 - Pyrazinamide in Treatment Regimens |
|
|
Increase in Multidrug-Resistant TB Treatment with Shortened Regimens |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
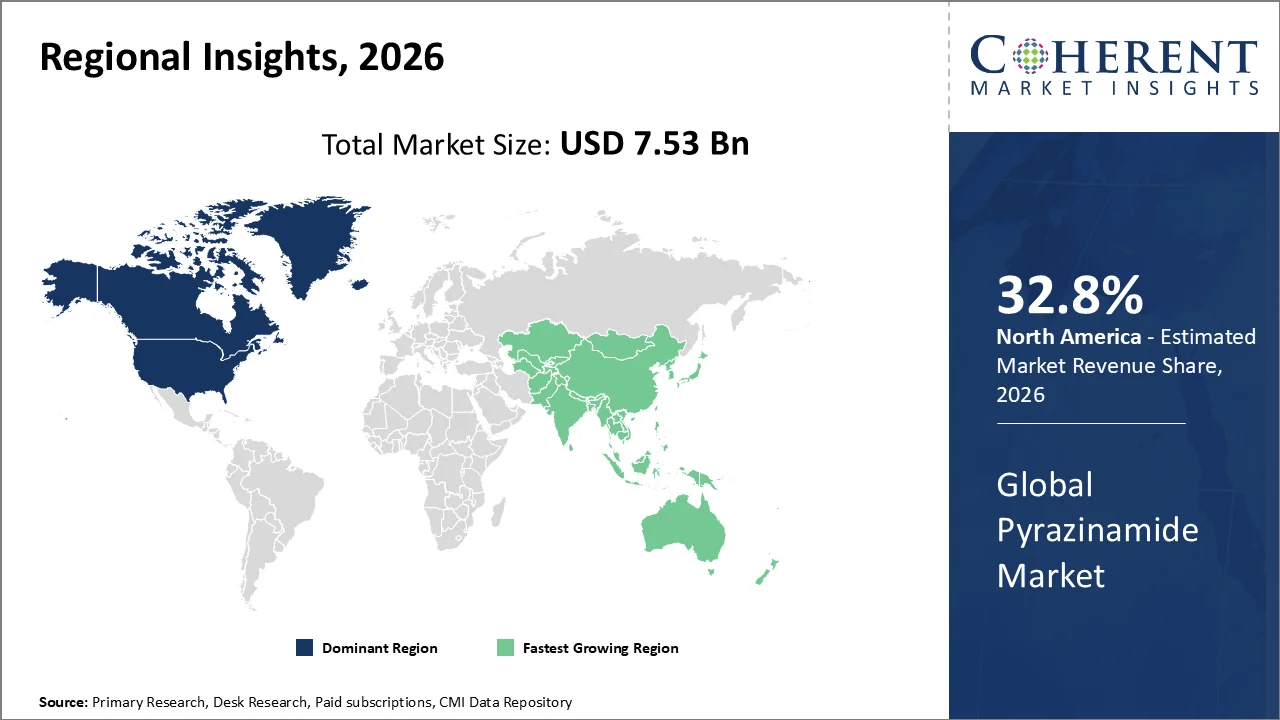
The North America region accounts for 32.8% of the market in 2026. The region’s growth is backed by ongoing burden of tuberculosis (TB) and the public health strategies implemented in both the U.S. and Canada to manage and treat TB infections. In the U.S., Pyrazinamide remains a core first‑line medication in combination therapy. According to the CDC, thus emphasizing the continuous treatment needs for the disease.
The public health investment plays a crucial role in TB management in the U.S. The federal and state programs are prioritizing expanded diagnosis as well as treatment access. The US FDA regularly checks and approves TB treatment plans, including those with pyrazinamide.
In Canada, the Public Health Agency of Canada (PHAC) reported 2,508 new TB cases in 2024, with an incidence rate of about 6.1 per 100,000 people. The TB control strategy in Canada emphasizes on treatment availability as well as enhanced monitoring, thereby promoting the use of recommended first‑line drugs like pyrazinamide. The continuing surveillance efforts as well as government health initiatives are placing a greater emphasis on high‑risk populations, which sustains the demand for anti‑TB therapies, including pyrazinamide.
The persistent TB incidence, health priorities, and government-supported programs in both the U.S. as well as Canada sustain North America’s leadership in the pyrazinamide industry, offering strong growth prospects and sustained demand for treatment.
The Asia Pacific region is poised to be as the fastest-growing region through 2026-2033, driven by ongoing national tuberculosis (TB) control programs, high TB incidence, and increasing adoption of standard first‑line treatment regimens that include pyrazinamide alongside rifampicin and isoniazid. According to the World Health Organization (WHO), in 2024 an estimated 10.7, thereby underscoring the scale of demand for essential tuberculosis drugs like pyrazinamide.
In India, which contributes substantially to global TB burden, the government’s National TB Elimination Programme (NTEP) continues to employ pyrazinamide as part of the standard 6‑month antibiotic regimen of isoniazid, rifampicin, pyrazinamide, and ethambutol for drug‑susceptible TB, thus emphasizing its role in national treatment protocols. China, similarly bearing a large share of TB cases, maintains pyrazinamide use within public health treatment strategies backed by expanded diagnostic and treatment infrastructure, thereby reflecting sustained public sector procurement of first‑line TB medicines.
In addition, the region has witnessed key developments in the tuberculosis therapeutics landscape. In May 2024, Moreover, the leading generic manufacturers in India continue to supply pyrazinamide-based fixed-dose combinations, thus strengthening the region's position as a key supplier for essential TB drugs worldwide.
The United States Pyrazinamide Market is witnessing steady growth. The market is being driven by ongoing U.S. tuberculosis (TB) treatment practices, where pyrazinamide continues to be a key component of first‑line therapy in combination with isoniazid, rifampin, and ethambutol, as recommended by the CDC in standard TB regimens (HRZE) for drug‑susceptible TB patients. In 2023, 82.6, underscoring its clinical importance in treatment plans.
Market interest is also affected by shifts in TB incidence. According to the CDC’s TB Surveillance Report, the United States reported 9,633 TB cases in 2023 with an incidence rate of 2.9 per 100,000 persons, a rise in case counts for the third consecutive year, indicating sustained need for anti‑TB drugs like pyrazinamide.
China’s Pyrazinamide market is closely tied to the country’s national priorities for tuberculosis (TB) control, treatment access and public health policy. Pyrazinamide is a key first‑line antibiotic in standard TB therapy alongside isoniazid and rifampicin, recommended for effective shorter regimens and improved cure rates. China is one of the top global TB burden countries, with national policy placing a greater emphasis on expanding preventive treatment as well as systematic screening. These include increasing Tuberculosis Preventive Treatment (TPT) coverage targets to 60% by 2025 and 80% by 2030 for close contacts of TB cases. These efforts are supported by guidance from the China CDC and national plans.
The government surveillance systems in China (NNIDRS) provide continual reporting of TB incidence and treatment outcomes via the Chinese Center for Disease Control and Prevention, thereby supporting policy adjustments and treatment strategies which include first‑line drugs like pyrazinamide. In recent industry developments, major pharmaceutical firms like in October 2022, Lupin Limited have collaborated with global health initiatives like the Clinton Health Access Initiative to enhance availability and reduce the burden of TB medications, reflecting partnership momentum in anti‑TB therapeutics.
Some of the major key players in Pyrazinamide are Pure Chemistry Scientific, LGM Pharma, TCI, HBCChem, Alfa Chemistry, Toronto Research Chemicals, Acros Organics, AlliChem, Waterstone Technology, City Chemical, EDQM, J & K SCIENTIFIC, Meryer (Shanghai) Chemical Technology, Energy Chemical, and Beijing Ouhe Technology.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 7.53 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.9% | 2033 Value Projection: | USD 17.23 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pure Chemistry Scientific, LGM Pharma, TCI, HBCChem, Alfa Chemistry, Toronto Research Chemicals, Acros Organics, AlliChem, Waterstone Technology, City Chemical, EDQM, J & K SCIENTIFIC, Meryer (Shanghai) Chemical Technology, Energy Chemical, and Beijing Ouhe Technology. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients