Vaccine Contract Manufacturing Market Size and Forecast – 2026 – 2033
The global vaccine contract manufacturing market is valued at around USD 3.0 billion in 2026 and is forecast to reach approximately USD 6.3 billion by 2033, representing a compound annual growth rate (CAGR) of about 8.45 % from 2026 to 2033.
Global Vaccine Contract Manufacturing Market Overview
Vaccine contract manufacturing refers to outsourcing vaccine development, production, formulation, filling, packaging, and quality testing to specialized third-party manufacturers. Pharmaceutical and biotechnology companies use contract development and manufacturing organizations (CDMOs) to reduce capital investment, accelerate time-to-market, and access advanced technologies. Services include viral and bacterial vaccine production, cell culture, recombinant vaccines, mRNA platforms, and sterile fill-finish operations. Growing demand for immunization programs, pandemic preparedness, and biologics innovation drives market expansion. Regulatory compliance, cold-chain capabilities, and scalable manufacturing infrastructure are critical factors. The sector continues evolving with technological advancements, strategic partnerships, and increased global vaccine production capacity.
Key Takeaways
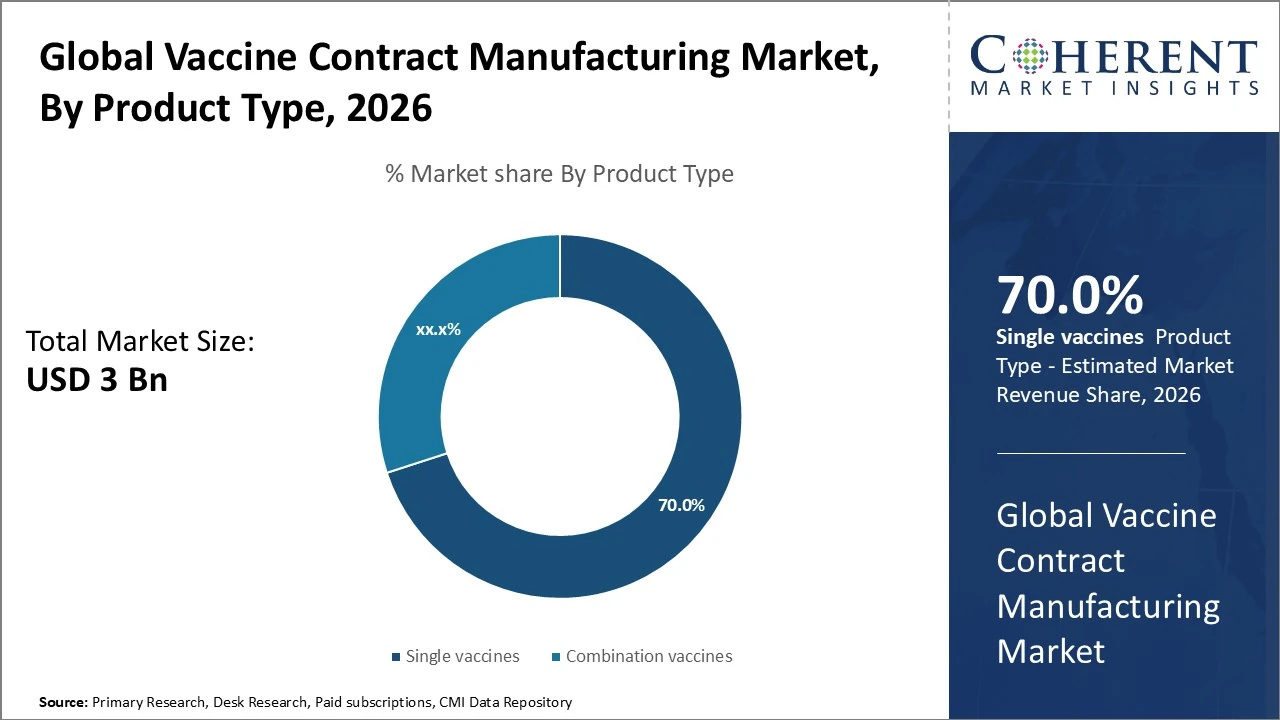
In the global vaccine contract manufacturing market, single vaccines dominate, accounting for an estimated ~70 % share of total revenue.
Viral-based technologies dominate with an estimated ~65 % share.
In the vaccine contract manufacturing market, Pharmaceutical & biotechnology companies are the largest end users, capturing a majority share of around 63 %.
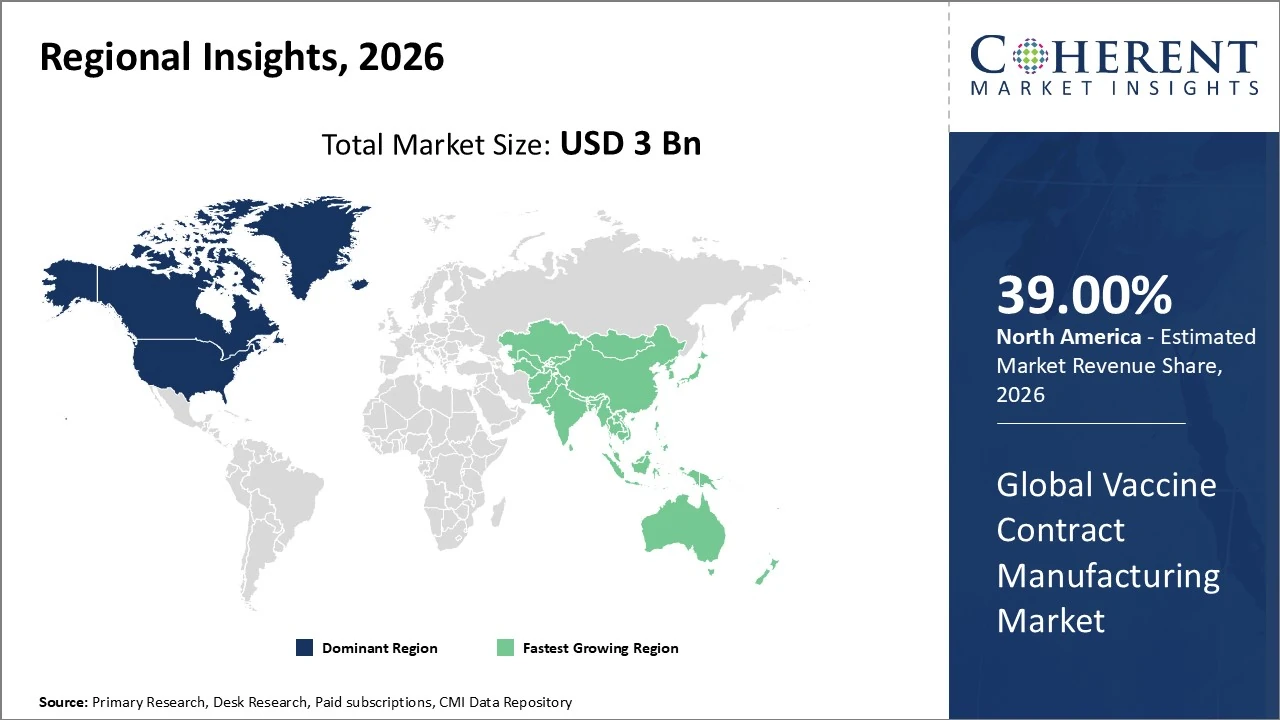
The North America vaccine contract manufacturing market leads globally, holding about 39 % of total market revenue in 2026.
The Asia Pacific vaccine contract manufacturing market is rapidly expanding, holding about 26% of the global market share.
The U.S. leads North America with ~88 % regional share.
The Germany vaccine contract manufacturing market is a major European hub, projected to grow from around USD 443 million in 2026 to about USD 619 million by 2033.
Vaccine Contract Manufacturing Market Segmentation Analysis
To learn more about this report, Request Free Sample
Vaccine Contract Manufacturing Market Insights, By Product Type
In the global vaccine contract manufacturing market, Single vaccines dominate, accounting for an estimated 70 % share of total revenue due to widespread demand for monovalent immunizations like influenza, polio and hepatitis vaccines. Combination vaccines hold the remaining ~30 % share, driven by multi-disease formulations that improve immunization efficiency despite higher production complexity. Single products remain preferred for targeted programs and easier regulatory pathways, while combination products are growing for pediatric schedules and emerging markets. Outsourcing to CDMOs continues rising across both segments as biopharma partners seek scalable, compliant manufacturing capacity.
Vaccine Contract Manufacturing Market Insights, By Technology
In the vaccine contract manufacturing market, Viral-based technologies dominate with an estimated 65 % share, driven by high demand influenza, and other viral vaccines and extensive outsourcing for complex bioprocessing. mRNA technology accounts for a significant portion of remaining share, reflecting rapid adoption and strong growth in novel platforms. Subunit/protein-based technologies hold a moderate share as firms expand recombinant approaches for targeted immunization. Inactivated and live attenuated vaccine technologies together make up smaller individual shares but remain essential for routine immunization programs. Overall, diversified technology portfolios are key for CDMOs to meet broad client needs.
Vaccine Contract Manufacturing Market Insights, By End-User
In the vaccine contract manufacturing market, Pharmaceutical & biotechnology companies are the largest end users, capturing a majority share of around 63 %, as they outsource both clinical- and commercial-scale production to CMOs to save costs and speed time-to-market. Government agencies and public health bodies hold the next significant share (~26 %), driven by pandemic preparedness contracts and national vaccination programs. Research institutions and academia comprise a smaller share (~7 %), focusing on early-phase clinical batches and specialized research production. Other end users contribute the remaining portion, reflecting diverse outsourcing needs across sectors.
Vaccine Contract Manufacturing Market Trends
Growing adoption of mRNA, recombinant, and cell-based technologies is increasing demand for specialized CDMO capabilities and flexible manufacturing facilities.
Biopharma companies are forming multi-year manufacturing partnerships to secure scalable capacity and ensure supply chain stability.
Manufacturers are investing in new facilities across North America, Europe, and Asia-Pacific to strengthen pandemic preparedness and reduce geographic supply risks.
Vaccine Contract Manufacturing Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Vaccine Contract Manufacturing Market Analysis and Trends
The North America vaccine contract manufacturing market leads globally, holding about 39 % of total market revenue in 2026, driven by strong outsourcing demand, advanced biomanufacturing infrastructure, and high R&D investment in mRNA and recombinant platforms. The United States contributes roughly 88 % of the region’s share, supported by major pharmaceutical partnerships and public funding for vaccine production capacity. Trends include expansion of modular, single-use facilities, increased government support for domestic manufacturing, and strategic alliances to meet large-scale vaccine needs, reinforcing North America’s dominant position.
Asia Pacific Vaccine Contract Manufacturing Market Analysis and Trends
The Asia Pacific vaccine contract manufacturing market is rapidly expanding, holding about 26% of the global market share in 2026 as a key outsourcing hub driven by cost-effective production, growing healthcare demand, and supportive government policies. Markets in China and India together contribute roughly 75 % of regional production volume, with China leading in capacity and India growing fastest. The region is forecast to register strong growth (CAGR ~8.5 % through 2033), propelled by infrastructure investments, regulatory improvements, and increasing global biopharma partnerships.
Vaccine Contract Manufacturing Market Outlook for Key Countries
USA Vaccine Contract Manufacturing Market Analysis and Trends
The United States vaccine contract manufacturing market is driven by increasing outsourcing of complex vaccine production and strong domestic biopharma demand. Fill-finishing operations dominate due to regulatory complexity and specialized needs. The U.S. leads North America with 88 % regional share in contract manufacturing, supported by robust immunization programs and advanced infrastructure. Trends include heightened pandemic preparedness spending, rising public-private collaborations, and expansion of mRNA, viral vector, and recombinant vaccine pipelines.
Germany Vaccine Contract Manufacturing Market Analysis and Trends
The Germany vaccine contract manufacturing market is a major European hub led by strong CDMO capabilities, advanced bioprocessing infrastructure, and significant R&D investment. Attenuated vaccines are the largest segment in 2026, while DNA vaccines show rapid growth. Key trends include expansion of mRNA and viral vaccine production capacity, increased government pandemic preparedness funding, and strategic partnerships with global pharma firms to support scalable manufacturing and supply resilience.
Analyst Opinion
Analysts highlight that pharmaceutical and biotech companies increasingly rely on CDMOs to reduce costs, speed up time-to-market, and handle complex vaccine platforms.
mRNA, viral vector, and recombinant technologies are key growth drivers, with analysts noting higher demand for specialized facilities and technical expertise.
Long-term agreements between vaccine developers and CMOs are expected to stabilize supply chains and mitigate production risks.
Expansion in Asia-Pacific and North America is seen as essential for global supply security and cost efficiency.
Analysts emphasize that stringent regulations and quality standards remain the top priority for market players to maintain credibility and secure government contracts.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 3.0 Billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.45% | 2033 Value Projection: | USD 6.3 Billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Lonza Group AG, Catalent, Inc., Thermo Fisher Scientific Inc., Samsung Biologics Co., Ltd. and WuXi Biologics | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Vaccine Contract Manufacturing Market Growth Factors
The vaccine contract manufacturing market is driven by several key growth factors. Rising global immunization programs and increasing demand for vaccines, including mRNA, viral vector, and recombinant types, are fueling outsourcing to CDMOs. High development costs and complex production processes encourage pharmaceutical and biotech companies to rely on specialized manufacturers. Pandemic preparedness and government funding further support capacity expansion. Technological advancements, such as single-use systems and scalable bioreactors, enhance efficiency and reduce production timelines. Strategic partnerships and long-term agreements between developers and contract manufacturers ensure supply stability, while expanding markets in Asia-Pacific and Latin America provide additional growth opportunities.
Vaccine Contract Manufacturing Market Development
The vaccine contract manufacturing market has developed rapidly over the past decade, driven by rising global vaccine demand and the need for specialized production capabilities. Pharmaceutical and biotechnology companies increasingly outsource clinical- and commercial-scale manufacturing to CDMOs to reduce capital expenditure and accelerate time-to-market. Technological advancements, including mRNA platforms, viral vector vaccines, and recombinant production systems, have expanded service offerings. Single-use bioreactors, modular facilities, and automation improve efficiency and scalability. Regional expansion, particularly in North America and Asia-Pacific, supports global supply chains. Strategic collaborations, mergers, and acquisitions among CDMOs and biopharma firms further strengthen capabilities and market reach.
Key Players
Leading Companies of the Market
Lonza Group AG
Catalent, Inc.
Thermo Fisher Scientific Inc.,
Samsung Biologics Co.,
WuXi Biologics
Key global players in the vaccine contract manufacturing market include Lonza Group AG, Catalent, Inc., Thermo Fisher Scientific Inc., Samsung Biologics Co., Ltd. and WuXi Biologics, known for broad CDMO services and scalable capacity.
Vaccine Contract Manufacturing Market Future Outlook
The future outlook of the vaccine contract manufacturing market is highly promising, with sustained growth expected through 2033. Rising global immunization programs, emerging infectious diseases, and expanding mRNA, viral vector, and recombinant vaccine pipelines will drive demand for outsourced manufacturing. CDMOs are investing in scalable, single-use, and automated facilities to enhance efficiency and meet rapid production needs. Strategic partnerships and long-term contracts between biopharma companies and CMOs will continue to secure supply chains. Regional growth is projected in Asia-Pacific, North America, and Europe, supported by government funding and pandemic preparedness initiatives, positioning the market for robust expansion.
Vaccine Contract Manufacturing Market Historical Analysis
The vaccine contract manufacturing market has experienced steady growth over the past decade, driven by increasing global demand for vaccines and the rising complexity of production. Historically, pharmaceutical and biotechnology companies outsourced manufacturing to CDMOs to reduce capital costs and leverage specialized expertise in viral, recombinant, and inactivated vaccines. Early growth was fueled by routine immunization programs, while the COVID‑19 pandemic significantly accelerated demand for mRNA and viral vector vaccine production. Investments in modular facilities, single-use bioreactors, and advanced quality control systems improved scalability and efficiency. Regional expansion in North America, Europe, and Asia-Pacific established a robust global manufacturing network, laying the foundation for future growth.
Sources
Primary Research Interviews:
Pharmaceutical and Biotechnology Companies
Contract Development and Manufacturing Organizations
Regulatory Authorities and Health Agencies
Industry Experts and Consultants
Databases:
PubMed / MEDLINE
ClinicalTrials.gov
USPTO / WIPO / EPO Databases
Journals:
Vaccine
Human Vaccines & Immunotherapeutics
Journal of Biotechnology
Biotechnology and Bioengineering
Newspapers:
The Wall Street Journal
Financial Times
The Guardian
The New York Times
Associations:
International Society for Pharmaceutical Engineering (ISPE)
Parenteral Drug Association (PDA)
BIO (Biotechnology Innovation Organization)
International Federation of Pharmaceutical Manufacturers & Associations (IFPMA)
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients