The Premature Ejaculation Treatment Market is anticipated to grow at a CAGR of 8.9% with USD 3.2 Bn in 2026 and is expected to reach USD 5.7 Bn in 2033. The clinical landscape of premature ejaculation (PE) continues to evolve with updated expert guidance and research. For instance, in March 2026, Progrès en Urologie published current international recommendations emphasizing patient assessment, psychological impact, and first‑line use of lidocaine/prilocaine topical sprays or on‑demand dapoxetine, combined with sex therapy as best practice. Additionally, in March 2026, at the European Association of Urology Annual Congress, research presented on a smartphone app intervention highlighted digital behavioural therapy as an emerging supportive treatment for PE management. Such developments reflect integration of evidence‑based pharmacologic and digital therapeutic advances accepted by clinical authorities.
Source: Science Direct; EAU
Selective Serotonin Reuptake Inhibitors (SSRIs) is projected to account for the largest share of cognitive systems spending in 2026, representing approximately 42.0% of the total volume. Selective Serotonin Reuptake Inhibitors dominate because they have the strongest clinical evidence for efficacy in delaying ejaculation and improving ejaculatory control, which drives physician preference and prescription rates. A bibliometric analysis published in Journal of Pharmacology and Pharmacotherapeutics (online first July 21, 2025; issue June 2026) highlights sustained research and scholarly focus on SSRIs in PE management, reflecting their central role in treatment protocols. Furthermore, a randomized clinical trial published December 1, 2025 demonstrated significant improvements in intravaginal ejaculatory latency time (IELT) with SSRI regimens like dapoxetine compared to other treatments, supporting their clinical utility. This robust and evolving research base reinforces SSRIs as the preferred pharmacological option in clinical practice and market utilization.
To learn more about this report, Request Free Sample
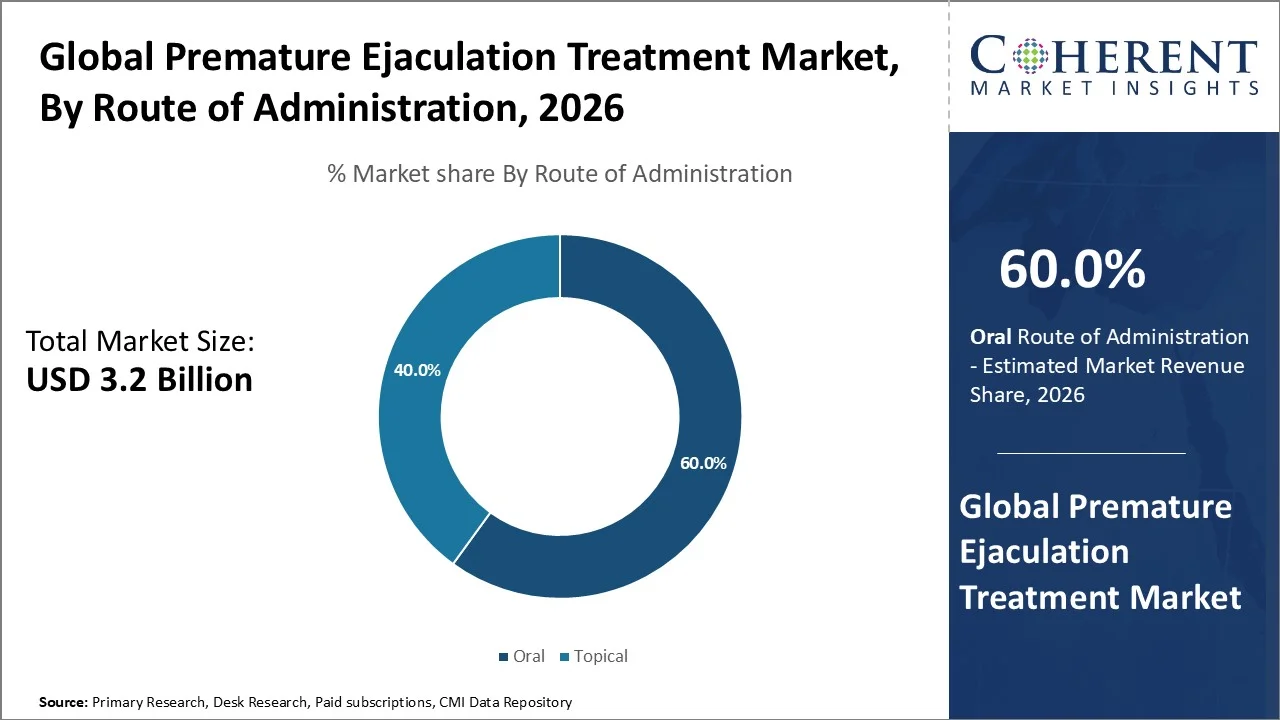
Based on route of administration, oral dominate the market, accounting for a significant 60.0% share in 2026. The oral route dominates the premature ejaculation treatment market because oral pharmacological agents especially SSRIs like dapoxetine are the most clinically studied and widely accepted interventions for PE, offering systemic modulation of ejaculatory control. Dapoxetine, the first **oral “on‑demand” SSRI originally approved for PE, is rapidly absorbed and taken ~1–3 hours before activity, significantly prolonging intravaginal ejaculatory latency time (IELT) versus placebo in large randomized controlled trials involving ~4,800 men (improved stopwatch‑measured IELT in phase III studies reported in multicentre publications). Oral SSRIs like paroxetine, sertraline and citalopram have also shown significant IELT increases compared with placebo, demonstrating broad evidence for oral systemic treatments over behavioral or topical routes. These oral options are preferred due to ease of administration, well‑documented efficacy and physician prescribing patterns.
Additionally, oral medications such as selective serotonin reuptake inhibitors (SSRIs) including dapoxetine (commonly used on demand) and off‑label SSRIs like paroxetine or sertraline are widely prescribed because they can be taken without needing specialized application techniques, fit easily into daily or pre‑sex routines, and have established safety profiles from multiple clinical studies. Although dapoxetine is not yet approved by the US FDA as of May 19, 2023 (per Medscape) and in the US many drugs are used off‑label for PE, it remains a preferred choice where approved due to rapid absorption and ease of dosing.
Hospitals account for the largest share of 40.0% in 2026 because it provides comprehensive, clinician‑led assessment and management of premature ejaculation (PE), integrating urology, sexual medicine, and behavioral therapy with access to advanced diagnostics and prescription pharmaceuticals. Hospitals also facilitate multidisciplinary care pathways, ensuring accurate diagnosis, patient education, and monitored use of both oral SSRIs and topical treatments. In February 2026, the World Health Organization’s consolidated operational handbook on sexually transmitted infections and associated sexual dysfunctions emphasized the central role of hospital and clinical service delivery platforms in effective sexual health care and treatment continuity. (who.int, published 11 February 2026) This structured clinical environment increases patient trust and drives higher PE treatment uptake through hospital pharmacies and specialist referral networks.
A World Health Organization consolidated operational handbook on sexually transmitted infections published 11 February 2026 underscores the importance of integrating sexual health and related dysfunction services, including diagnosis and treatment, within hospital and clinical platforms to ensure high‑quality care. Additionally, the U.S. CDC FY 2026 Congressional Justification published 3 April 2026 highlights substantial federal investment in sexual health services infrastructure, supporting robust clinical service networks where hospitals often lead in delivering care and therapeutic interventions for sexual health conditions.
Rising awareness of sexual health has driven the premature ejaculation treatment market growth over the forecast period. Increasing awareness of sexual health including conditions like premature ejaculation (PE), one of the most common male sexual dysfunctions affecting up to ~38% of men according to clinical practice guidelines updated in 2025 reduces stigma and encourages men to seek professional help, boosting demand for diagnosis and treatment options. Enhanced public knowledge, promoted through campaigns such as Sexual Health Awareness initiatives and broader sexual health education, helps normalize conversations around sexual well‑being and encourages earlier intervention, thereby driving growth in the premature ejaculation treatment market.
For instance, in August 2025, The Birds and Bees Talk – a large sexual health literacy campaign by Durex reached over 4 million young people and 2,000+ schools in India, improving STI awareness by ~20 % among participants through school‑based education and dialogue. Additionally, in 2026, UNESCO reported its flagship programme Our rights, Our lives, Our future reached tens of millions of learners and expanded digital and school‑based sexuality education, with ~85 % of countries having supportive sexuality education policies (not quality across all elements).
Rising prevalence of premature ejaculation has driven the global premature ejaculation treatment market growth over the forecast period. The high and variable prevalence of premature ejaculation (PE) significantly fuels demand for treatments. A systematic review published on 06 August 2025 in The Journal of Sexual Medicine analysing 79 studies from 33 countries reported mean overall PE prevalence at ~14.2 % (with certain subtypes showing up to ~43.9 %) among adult men, reflecting common sexual dysfunction cases worldwide. Additionally, studies indicate lifetime complaint‑based prevalence above 20 % across adult populations (Frontiers in Psychiatry, published 05 January 2026), underscoring widespread occurrence and psychological impact of PE. Such high prevalence increases diagnosis rates and treatment seeking, thereby expanding the market for clinical and pharmacological interventions for premature ejaculation.
Innovation in drug formulations and delivery technologies is expanding treatment options and improving patient adherence in the premature ejaculation (PE) treatment market. Newly developed rapid‑onset oral SSRIs and site‑specific topical agents enhance intravaginal ejaculatory latency time with fewer side effects, encouraging clinician adoption and patient uptake. A peer‑reviewed study published in BMC Urology (December 2025) showed that optimized oral SRIs and advanced topical sprays significantly improved outcomes versus traditional therapies, supporting broader clinical use. Additionally, USFDA guidance updated March 2026 emphasizes streamlined pathways for evaluating novel sexual health therapeutics, boosting R&D investment. Hospitals remain the dominant distribution channel because they integrate specialist diagnosis, multidisciplinary sexual health services, and monitored treatment regimens critical for complex cases and combination therapies. For instance, in February 2026, the World Health Organization’s operational handbook highlights clinical service platforms, including hospital and sexual health clinics, as central to effective management of sexual dysfunctions, reinforcing hospital leadership in therapy distribution.
|
Current Event |
Description and its Impact |
|
USFDA Modernizing Regulatory Framework for Sexual Health Therapies |
|
|
CDC Expands Sexual Health Funding to Include Dysfunction Screening (FY 2026) |
|
|
European Medicines Agency (EMA) Promotes Adaptive Clinical Trials for Sexual Health Drugs |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Digital health platforms and telemedicine services are increasingly used for the remote management of premature ejaculation (PE). In March 2026, research presented at the European Association of Urology Annual Congress showed that smartphone app‑based behavioral therapy significantly improved ejaculatory control outcomes, highlighting the trend toward technology‑enabled treatment and patient engagement outside traditional clinic settings.
Source: European Association of Urology (EAU26); European Medicine Journal
Pharmaceutical innovation is focusing on rapid‑onset oral SSRIs and site‑specific topical sprays to improve efficacy and reduce side effects. A randomized clinical trial published December 2025 in BMC Urology demonstrated that optimized oral formulations (e.g., dapoxetine regimens) significantly increased intravaginal ejaculatory latency time compared with older therapies, representing a key formulation advancement in the PE treatment landscape.
In March 2026, the U.S. Food and Drug Administration (USFDA) issued updated guidance easing evaluation pathways for novel sexual health drugs, including PE therapies, by clarifying requirements for advanced formulations and adaptive trials. This regulatory shift encourages industry innovation and reduces time to market for next‑generation treatments.
Emerging research is exploring biomarkers for ejaculatory control and serotonergic response, enabling more personalized treatment plans. A Frontiers in Endocrinology review published April 2026 highlights ongoing investigations into genetic, hormonal, and neurotransmitter markers that can tailor therapy selection and improve outcomes for PE patients.
To learn more about this report, Request Free Sample
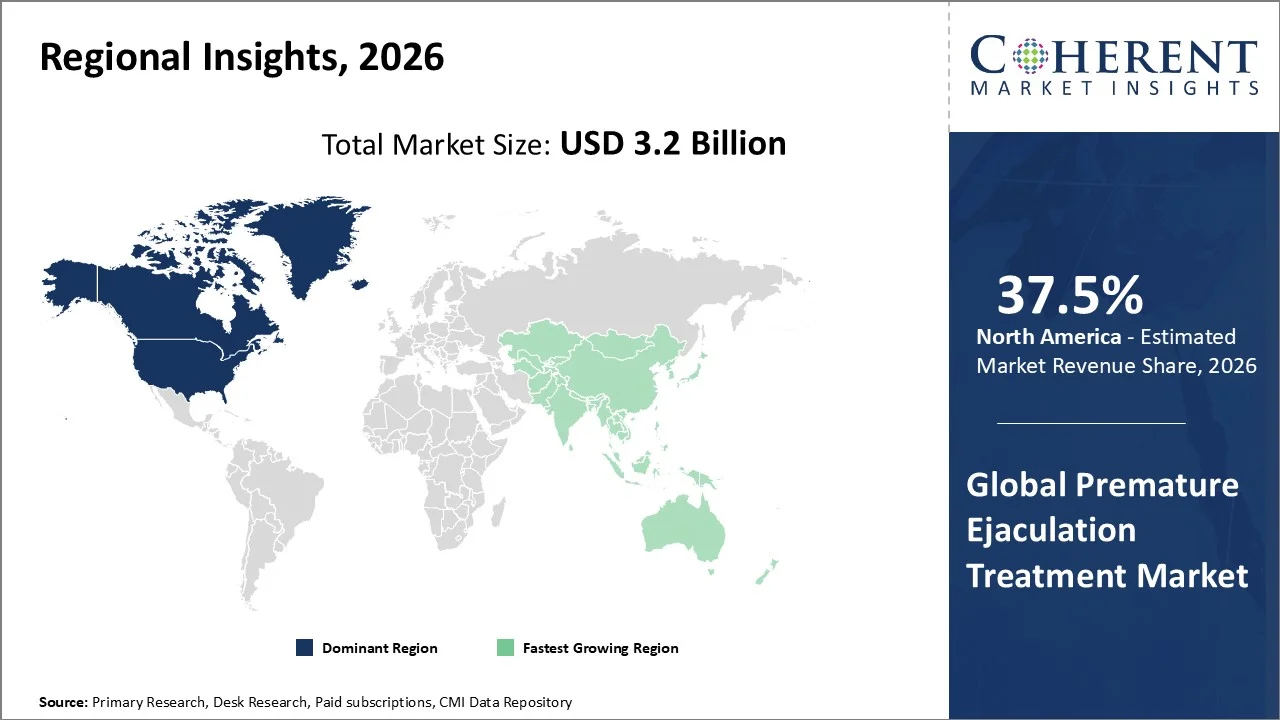
North America account 37.5% market share in 2026, due to a high prevalence of the condition, advanced healthcare infrastructure, and broad access to treatment options. Epidemiological data indicate that 20–30 % of adult men in the U.S. report premature ejaculation, making PE one of the most common male sexual dysfunctions, which drives treatment demand. North America’s high healthcare expenditure and awareness about sexual health encourage men to seek medical help and access therapies, including oral SSRIs and topical treatments. The region also benefits from strong pharmaceutical R&D and major industry players headquartered in the U.S. and Canada, facilitating innovation and product availability. Additionally, telemedicine and online pharmacy expansion in 2026 enhances treatment accessibility and patient discretion, further sustaining regional market leadership.
Additionally, a comprehensive systematic review published on 05 October 2025 found average PE prevalence around 14.2 % worldwide across 79 studies, with the condition commonly observed in adult male populations, highlighting substantial unmet needs that clinicians and patients seek to address. Additionally, premature ejaculation affects roughly 30 % of men aged 18–59 in U.S. clinical epidemiology data (StatPearls report update 2026), reinforcing high regional demand for treatment options. North America’s advanced healthcare infrastructure, widespread sexual health awareness, and extensive research output including recent U.S. and Canadian-based clinical studies on ejaculatory dysfunction published in Sexual Medicine in April 2026 further support strong uptake of diagnosis and therapies, fueling market growth.
The Asia-Pacific region is poised to be as the fastest-growing region through 2026-2033, owing to key manufacturers are expanding access to pharmacological treatments for premature ejaculation through strategic approvals and product launches, aligning with growing demand and regulatory progression. For example, Guangdong Tainkang Pharmaceutical’s dapoxetine hydrochloride tablets (Durative) received drug registration approval in Indonesia in March 2026 from the Indonesian FDA, enabling localized availability of this oral SSRI‑based PE treatment, and marking one of the first domestic PE drug approvals in that market. Menarini Group’s Priligy (dapoxetine) already a leading SSRI option for PE continues distribution in several Asia‑Pacific territories, underpinning the pharmaceutical segment’s depth of offerings. Additionally, ongoing Asia‑Pacific clinical development, such as the dapoxetine flexible‑dose study updated on 28 January 2026, underscores investment in localized evidence for efficacy and tolerability. These authentic regulatory approvals and continued R&D support the region’s rapid uptake of established and emerging PE treatments.
The U.S. remains dominant in the North America premature ejaculation treatment market due to several key factors. First, the U.S. healthcare system is one of the largest globally, with total national health expenditures reaching approximately USD 5.3 trillion in 2024, accounting for about 18 % of U.S. GDP one of the highest shares worldwide which supports broad access to diagnosis and care. This high level of spending enables advanced clinics, extensive specialist networks, and strong sexual health awareness, prompting more men to seek professional help for PE compared with other North American countries. Additionally, substantial pharmaceutical R&D activity and clinical development are underway: for example, the oral on‑demand candidate KH‑001 is in Phase 2 trials targeting lifelong premature ejaculation, illustrating ongoing innovation aimed at addressing unmet needs. Finally, widespread use of off‑label SSRIs and topical anesthetics as standard PE therapies in clinical practice in the U.S. further fuels market demand and treatment uptake.
China’s dominance in the Asia‑Pacific premature ejaculation treatment market is supported by high clinical engagement and documented prevalence, alongside growing physician emphasis on evidence‑based PE care. A nationwide survey of 1,000 Chinese physicians published on 26 March 2026 showed oral dapoxetine was the preferred first‑line therapy (used by 59.7 % of clinicians) along with psychotherapy in PE management, reflecting strong clinical uptake of approved treatments in China. Additionally, China’s large male population with significant PE complaints has long been established in research; earlier regional data showed ~25.8 % PE prevalence in one population sample, underscoring substantial demand for treatment. Together, strong clinical practice focus, patient burden, and established real‑world treatment evidence in China drive its regional leadership in the market.
Source: Lifescience.net; Science Direct
Some of the major key players in Premature Ejaculation Treatment Market are Regent Pacific Group Limited, Menarini Group, Furiex Pharmaceuticals, Inc., Absorption Pharmaceuticals, LLC, Ampio Pharmaceuticals, Inc., Aytu BioScience, NeuroHealing Pharmaceuticals Inc., and Dong-A Pharmaceutical Co., Ltd. Others
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 3.2 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.9% | 2033 Value Projection: | USD 5.7 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Regent Pacific Group Limited, Menarini Group, Furiex Pharmaceuticals, Inc., Absorption Pharmaceuticals, LLC, Ampio Pharmaceuticals, Inc., Aytu BioScience, NeuroHealing Pharmaceuticals Inc., and Dong-A Pharmaceutical Co., Ltd. |
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients