The vagus nerve stimulators market is estimated to be valued at USD 734.8 Mn in 2026 and is expected to reach USD 1,441.1 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 10.1% from 2026 to 2033.
To learn more about this report, Download Free Sample
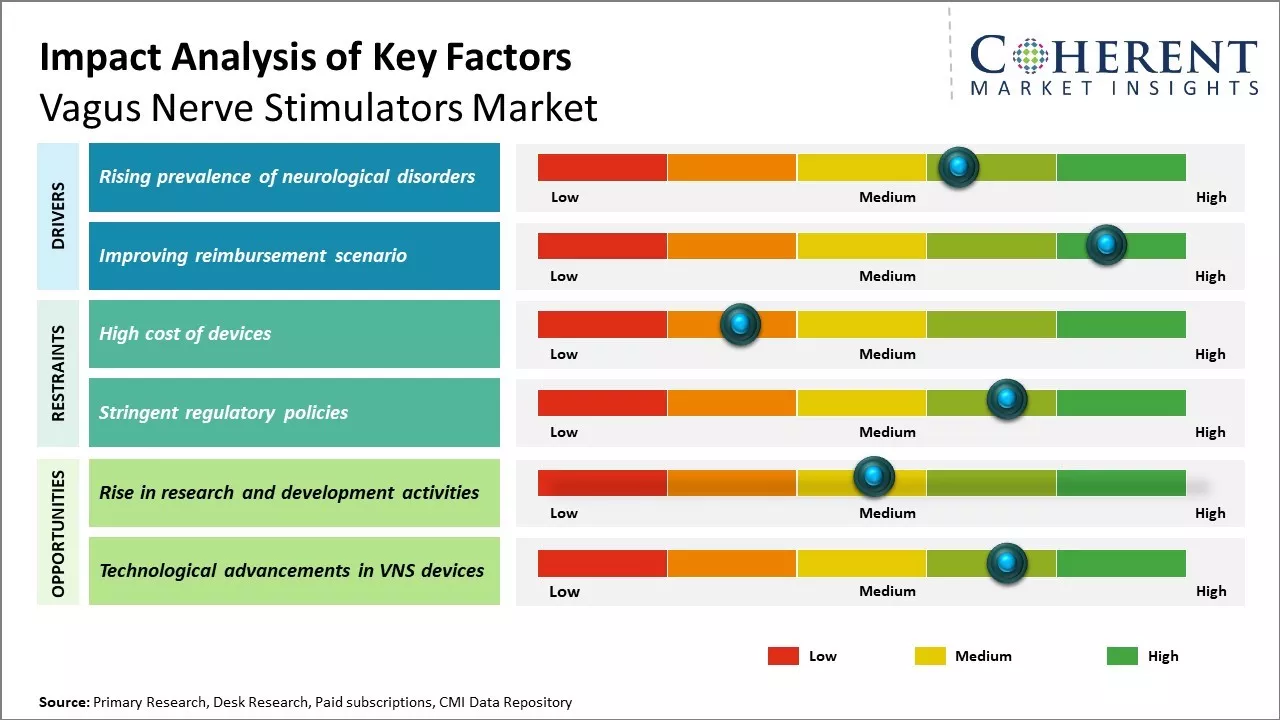
The market is expected to witness positive growth over the forecast period, owing to the increasing prevalence of neurological disorders worldwide. Vagus nerve stimulation therapy has emerged as an effective treatment option for epilepsy and treatment-resistant depression. Further, ongoing technological advancements to develop innovative vagus nerve stimulator devices is also expected to support the market growth.
However, the cost of devices and required surgical implantation limits growth potential.

To learn more about this report, Download Free Sample
|
Current Events |
Description and its impact |
|
Regulatory and Reimbursement Shifts |
|
|
Strategic Corporate Activities |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The global demographic shift toward an aging population has emerged as a pivotal driver for the vagus nerve stimulators market, particularly as neurological disorders become increasingly prevalent among elderly individuals. The physiological changes associated with aging—including reduced neurotransmitter production, compromised blood-brain barrier function, and accumulated oxidative stress—create an environment where traditional pharmaceutical interventions often prove insufficient.
For instance, according to a 2024 World Health Organization report, the number of people aged 60 and above is projected to increase from 1 billion in 2020 to 1.4 billion by 2030, with neurological conditions affecting approximately 15% of this demographic.
The product type segment includes implantable devices and external devices. Implantable devices segment is anticipated to hold 67.3% of the market share in 2026. Implantable devices offer longer-lasting treatment with minimal invasiveness compared to external devices.
Advancements such as miniaturization of components and improved battery lives have increased patient comfort and compliance. Further, latest generation implantable devices allow wireless programming and data collection via external accessories like pads and wq1ands, eliminating the need for replacement surgery. This has boosted patient acceptance of implantable vagus nerve stimulators. Ongoing R&D focusing on programmable and customized stimulation parameters tailored for individual needs also support the growth of this segment.
To learn more about this report, Download Free Sample
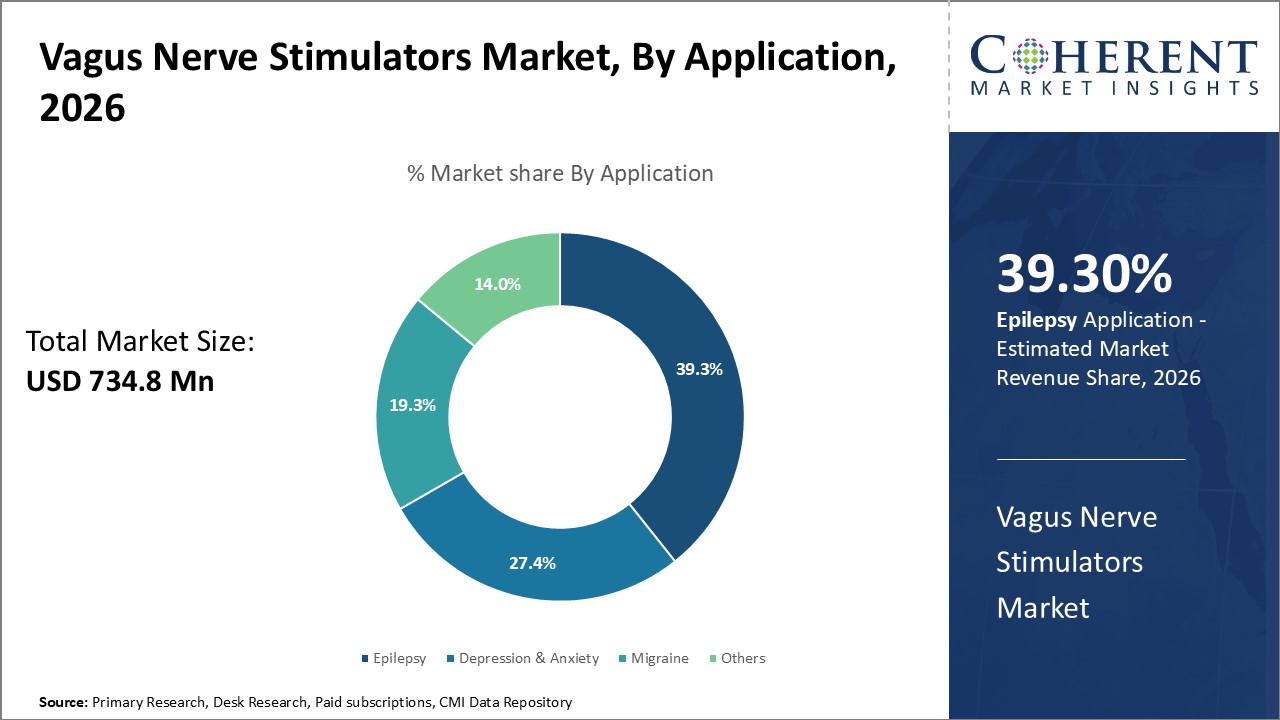
The application segment includes epilepsy, depression & anxiety, migraine, and others. Epilepsy contributes the highest share of the vagus nerve stimulators market and is projected to hold 39.3% of the market share in 2026.
Vagus nerve stimulation has been approved as an adjunctive therapy for epilepsy, especially for cases where seizure control cannot be achieved through medications. Clinical evidence showcasing the neurological benefits of vagus nerve stimulation in reducing seizure frequency and severity has led to increasing adoption among epileptic patients. Various mechanisms of action hypothesized indicate vagus nerve stimulation modulates central nervous system neurotransmitters released during seizure onset.
The material type segment includes ceramics, metallics, and polymerics. Metallics contributes the highest share of the vagus nerve stimulators market and is projected to hold 42.7% of the market share in 2026.
Metallic alloys like cobalt-chromium or titanium provide crucial attributes of corrosion resistance, biocompatibility, and mechanical strength required for implanted medical devices. They withstand harsh in-vivo environments without degradation or adverse tissue reactions over long implant durations. The electrically conductive property of metals enables precise stimulation delivery.
Artificial Intelligence (AI) is revolutionizing numerous medical device sectors, and the vagus nerve stimulators (VNS) market is no exception. By integrating AI technologies, companies are enhancing device functionality, improving patient outcomes, and streamlining operations.
To learn more about this report, Download Free Sample
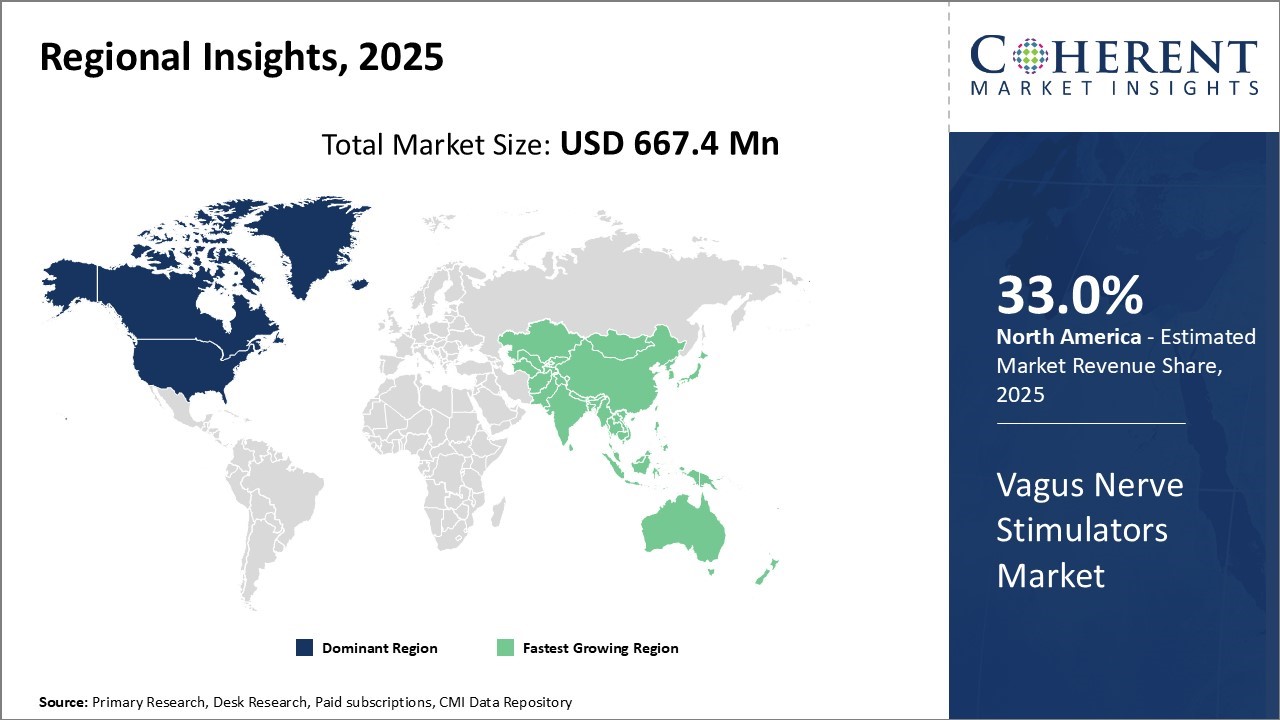
North America has established itself as the dominant region in the vagus nerve stimulators market and is projected to hold 33.0% of the market share in 2026. The presence of leading manufacturers such as LivaNova and electroCore in the U.S. has enabled the early adoption of vagus nerve stimulation devices in the region. Favorable reimbursement policies for conditions including epilepsy and depression have further increased access to these therapies.
Asia Pacific has emerged as the fastest growing regional market for vagus nerve stimulators. Countries such as China and Japan have witnessed a rapid increase in the incidence of neurological and psychiatric disorders in recent years, due to aging populations and changing lifestyles. This has spurred government initiatives to boost accessibility and affordability of effective treatment solutions. The low infrastructure and labor costs have also encouraged global players to establish manufacturing bases in Asia Pacific to serve both local and export markets.
For instance, the Japan Epilepsy Society reported that approximately 1 million people in Japan suffer from epilepsy, with nearly 30% being drug-resistant cases, creating a substantial demand for alternative treatment modalities like vagus nerve stimulation.
The United States leads the global vagus nerve stimulators market, driven by advanced healthcare infrastructure, high patient awareness, and substantial investment in medical technology. The presence of key manufacturers like LivaNova and electroCore has accelerated device adoption for treating conditions such as epilepsy, depression, and chronic pain. Furthermore, favorable reimbursement policies and extensive clinical research in neurological disorders enhance the market's growth prospects in the U.S.
In May 2026, MicroTransponder, a U.S.-based company, reported positive long-term results for stroke patients who received the company’s paired vagus nerve stimulation (VNS) therapy.
Germany stands out as a dominant market in Europe due to its well-established healthcare system and strong focus on medical innovation. German companies actively participate in the development and distribution of vagus nerve stimulation devices, supported by robust insurance frameworks that improve patient access. Research institutions and hospitals in Germany are also engaged in pioneering clinical trials, particularly targeting treatment-resistant depression and epilepsy.
Japan’s vagus nerve stimulators market benefits from a rapidly aging population with increasing prevalence of neurological disorders. The country has made significant strides in integrating advanced medical devices into standard care, supported by government initiatives aimed at improving neurological health. Japanese healthcare providers are adopting vagus nerve stimulation as a key therapy option, while manufacturers focus on creating devices tailored to the unique needs of the local population.
China is emerging as a fast-growing market for vagus nerve stimulators due to expanding healthcare access and rising awareness of neurological treatments. The country’s growing middle class and increasing incidence of neurological diseases drive demand for innovative therapies. Government policies encouraging medical device innovation and approval processes are accelerating market penetration, while local manufacturers are entering the space alongside global companies to meet diverse patient needs.
|
Category |
Price Range |
|
Implantable Cybernetic Systems (e.g., LivaNova VNS Therapy), Surgically implanted neurostimulators with generators and electrodes |
USD 15,000–USD 30,000 (device) Manufacturing complexity, battery life, brand premiums |
|
Non-Invasive Stimulators (e.g., gammaCore by electroCore) Transcutaneous devices (FDA-approved for migraines, depression) |
USD 3,000–USD 7,000 (device) Portability, clinical efficacy, competing therapies |
|
Surgical Implantation Hospital/clinician fees for implantation surgery |
USD 20,000–USD 40,000 Region, healthcare system, surgeon expertise |
|
Battery Replacement Costs for replacing implantable device batteries (every 5–10 years) |
USD 5,000–USD 12,000 Device longevity, regional service fees |
|
Programming/Management Tools | Clinician-facing software/hardware for device calibration and monitoring |
USD 2,000–USD 5,000/year Subscription models, software upgrades |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 734.8 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.1% | 2033 Value Projection: | USD 1,441.1 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
LivaNova PLC, electroCore Inc., SetPoint Medical Corporation, Beijing PINS Medical Co. Ltd., Parasym Ltd., Nervana LLC, tVNS Technologies GmbH, BioControl Medical, NeurAxis Inc., Soterix Medical Inc., MicroTransponder Inc., Boston Scientific Corporation, Cyberonics Inc., EnteroMedics Inc., ReShape Lifesciences Inc., and Cerbomed GmbH |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients