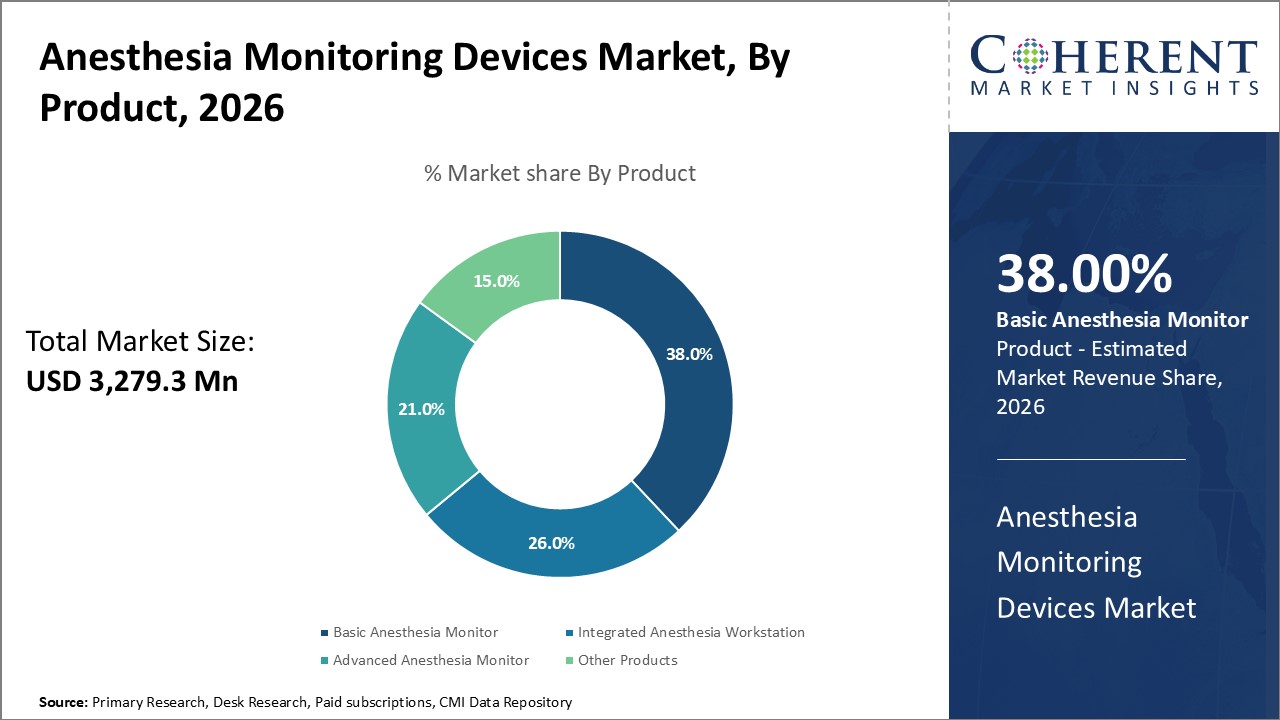
Anesthesia Monitoring Devices Market is estimated to be valued at USD 3,279.3 Mn in 2026 and is expected to reach USD 6,765.6 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 10.9% from 2026 to 2033.
An increasing number of surgical procedures, a higher prevalence of chronic diseases, and a strong focus on patient safety are driving the Anesthesia Monitoring Devices Market growth. Medical professionals are adopting advanced monitoring technologies, including integrated systems and real-time data analytics, to improve clinical decision-making. Hospitals and surgical centers continue to lead demand due to their need for precise, continuous monitoring. In emerging regions, expanding healthcare infrastructure and ongoing innovation from leading medical device companies further boost market expansion.
|
Current Events |
Description and its impact |
|
Technological Advances |
|
|
Regulatory and Policy Developments |
|
|
Healthcare Trends and Demographic Shifts |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Hospitals drive the demand for anesthesia monitoring devices by performing a large number of surgeries and managing complex medical cases within advanced healthcare settings. They prioritize investments in modern monitoring equipment to enhance patient safety and meet strict regulatory standards. Medical teams rely on these systems in well-equipped operating rooms to ensure precision during procedures. As central providers of emergency and specialized care, hospitals consistently use anesthesia monitoring devices to support better surgical outcomes and uphold high levels of clinical care. For instance, in December 2024, The Hal family in San Diego, USA, donated a state-of-the-art anesthesia machine and its operating equipment, valued at Shs 200 million, to Mbarara Regional Referral Hospital and Mbarara University of Science and Technology (MUST).
To learn more about this report, Request Free Sample
Healthcare providers are increasingly choosing basic anesthesia monitors for their cost-effectiveness, simplicity, and vital role in everyday surgical procedures. These monitors track essential vital signs, making them highly useful in smaller clinics and resource-constrained environments. Larger hospitals also rely on them as dependable backup systems during standard operations. Efforts to expand surgical services in developing regions further drive the adoption of these devices, as governments and organizations prioritize basic monitoring tools to strengthen essential medical infrastructure and improve patient care access.
Draeger released the Perseus A500 anesthesia workstation, marking one of their largest development projects ever. This innovative device delivers unprecedented configurability, high-performance ventilation, enhanced ergonomics, and automation to streamline the operating room workflow.
To learn more about this report, Request Free Sample
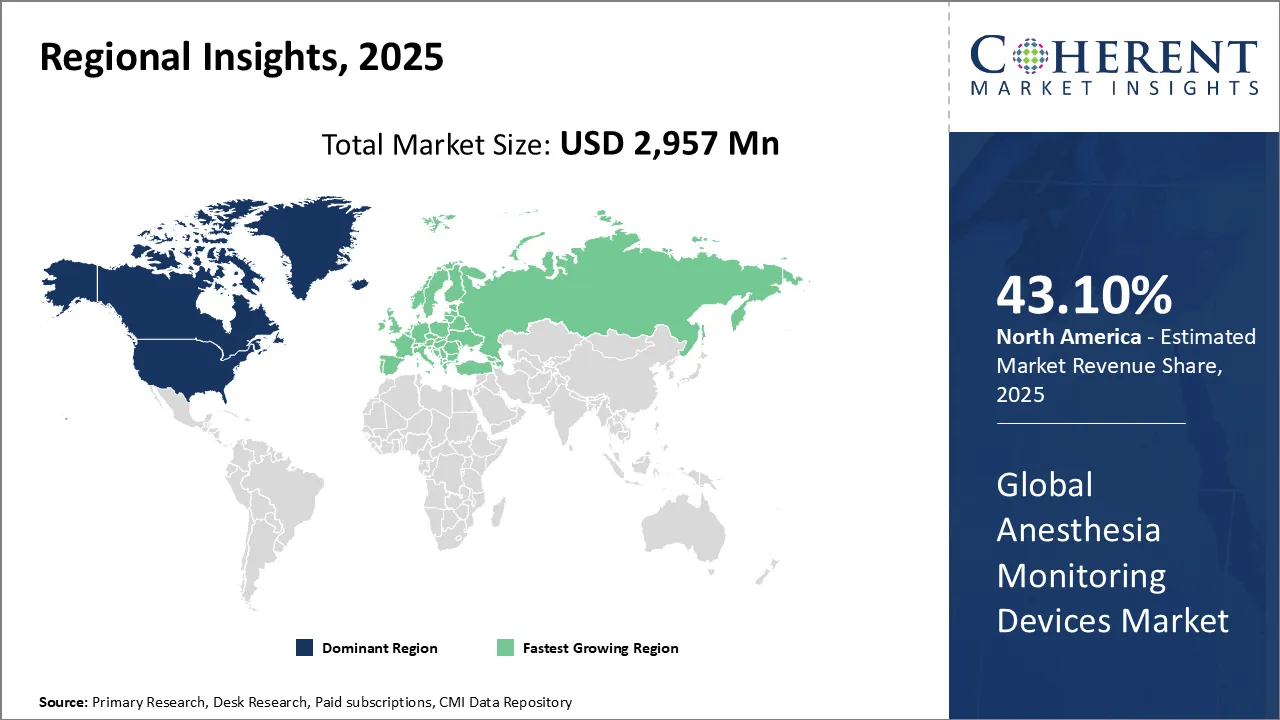
North America dominates the overall market with an estimated share of 43.1% in 2026. Advanced healthcare infrastructure, rapid adoption of innovative technologies, and a high volume of surgical procedures are driving growth in North America’s anesthesia monitoring devices market. Hospitals and surgical centers actively implement smart monitoring systems to improve patient safety and optimize workflows. Regulatory support and the presence of major medical device manufacturers further fuel market expansion. Additionally, the growing preference for outpatient surgeries increases the need for compact and efficient monitoring solutions across different healthcare settings.
Technological advancements and strict patient safety regulations are shaping the evolution of Europe’s anesthesia monitoring devices market. Healthcare providers actively adopt integrated monitoring solutions to enhance surgical accuracy and outcomes. Well-established healthcare infrastructure and increasing demand for minimally invasive surgeries support this growth. Governments promote healthcare modernization and invest in medical technology, accelerating market expansion. Additionally, rising awareness of perioperative care and the growth of outpatient services increase the demand for reliable, advanced anesthesia monitoring devices throughout Europe.
In May 2026, Philips introduced the VeriSight Pro 3D Intracardiac Echocardiography (ICE) catheter in Europe. This advanced catheter gives physicians real-time 3D imaging from inside the heart, enhancing clarity during complex procedures and often removing the need for general anesthesia.
Sweden’s anesthesia monitoring devices market continues to grow steadily as hospitals increase the use of advanced surgical procedures and prioritize patient safety. Companies are driving innovation through AI integration, wireless technology, and precision monitoring tools. Swedish firm Senzime is leading this transformation with its next-generation TetraGraph systems. Healthcare providers are also adopting more multiparameter and portable monitoring systems to improve efficiency in operating rooms. These developments show Sweden’s active efforts to modernize its healthcare system with cutting-edge technology. For instance, in October 2024, in Sweden, Senzime unveiled its next-generation TetraGraph® system at ANESTHESIOLOGY, developed with global anesthesiologists to redefine TOF monitoring through advanced algorithms and precision technology.
Improving healthcare infrastructure and a rise in surgical procedures are driving growth in India’s anesthesia monitoring devices market. Hospitals and diagnostic centers are increasingly adopting advanced monitoring technologies to improve patient safety and surgical accuracy. Government initiatives aimed at expanding healthcare access and upgrading medical facilities actively support market expansion. Additionally, growing awareness of anesthesia risks and the growth of private healthcare providers boost demand. Both urban hospitals and rural healthcare settings actively seek cost-effective, reliable monitoring solutions across India. For instance, in April 2026, Drager, a global leader in medical and safety technology, has launched the Atlan® A100 anesthesia workstation in India. This innovative system is designed to enhance patient care and streamline workflows in the operating room (OR).
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 3,279.3 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.9% | 2033 Value Projection: | USD 6,765.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Koninklijke Philips NV, Dragerwerk AG & Co. KgaA, Schiller AG, Infinium Medical, Masimo Corporation, Nihon Kohden Corporation, GE Healthcare, Medtronic PLC, and Mindray Medical International Limited, among others |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The expansion of outpatient and ambulatory surgical centers is reshaping the anesthesia monitoring market. ASCs require compact, portable, and user-friendly devices to accommodate faster patient turnover and limited space. This shift encourages manufacturers to design lightweight monitors with wireless capabilities and intuitive interfaces, optimizing efficiency in diverse clinical environments. Increasing preference for minimally invasive and day surgeries fuels this trend, driving demand for reliable anesthesia monitoring in non-traditional settings.
Emerging markets present significant growth opportunities as improving healthcare infrastructure and increased surgical demand drive the need for anesthesia monitoring devices. Many regions are focusing on upgrading medical facilities and enhancing patient safety protocols. Affordable, reliable, and easy-to-use anesthesia monitors tailored to these markets can capture a large customer base. Collaborations with local healthcare providers and governments can further facilitate product adoption and long-term growth in these underserved regions.
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients