Dermatology Diagnostic Devices and Therapeutics Market Size and Forecast – 2026 – 2033
The Global Dermatology Diagnostic Devices and Therapeutics Market size is estimated to be valued at USD 11.5 billion in 2026 and is expected to reach USD 19.8 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 8.2% from 2026 to 2033.
Global Dermatology Diagnostic Devices And Therapeutics Market Overview
The dermatology diagnostic devices and therapeutics market is driven by rising prevalence of skin disorders such as acne, psoriasis, and skin cancer, along with growing awareness of early diagnosis and treatment. Technological advancements, including AI-based imaging and non-invasive diagnostic tools, are improving accuracy and adoption rates. Increasing demand for aesthetic procedures and minimally invasive treatments further fuels market growth. Expanding healthcare infrastructure and higher healthcare spending, especially in emerging economies, also contribute. Additionally, aging populations and lifestyle-related skin conditions are boosting demand, while ongoing research and development activities are introducing innovative therapies and devices, accelerating overall market expansion.
Key Takeaways
Early detection devices dominate the Dermatology Diagnostic Devices and Therapeutics market, holding the largest market share due to high demand for melanoma diagnosis and ongoing technological innovation.
Therapeutic laser devices are the fastest-growing subsegment, fueled by increasing use in cosmetic dermatology and treatment of chronic skin conditions.
North America accounts for over 38% of the overall market share, supported by advanced healthcare infrastructure and reimbursement policies that encourage adoption of diagnostic and therapeutic solutions.
Asia Pacific is the fastest-growing region with a CAGR exceeding 10%, driven by rising healthcare expenditure, expanding facilities, and growing awareness of skin health in countries like India and China.
Dermatology Diagnostic Devices and Therapeutics Market Segmentation Analysis
To learn more about this report, Request Free Sample
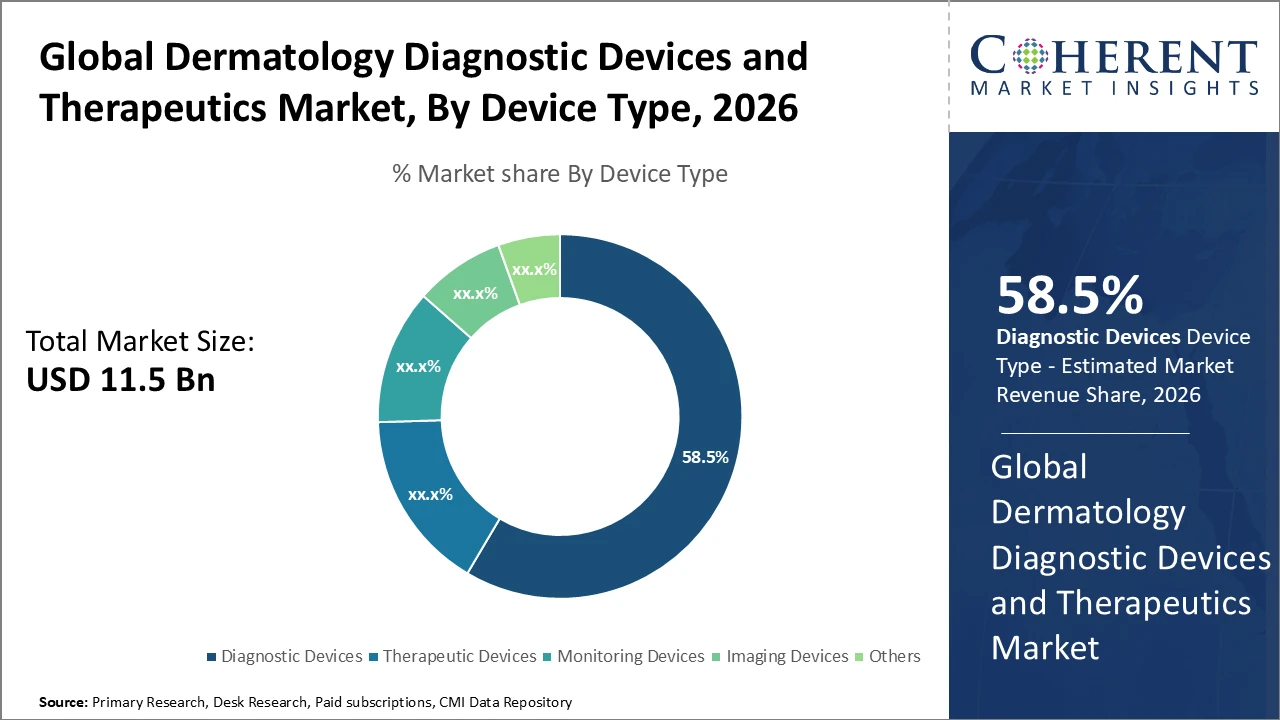
Dermatology Diagnostic Devices and Therapeutics Market Insights, By Device Type
Diagnostic Devices dominate the market with a share of 58.5% due to their essential role in early detection of skin diseases, particularly melanoma and other cancers. Integration of advanced technologies, such as AI-powered dermatoscopes and confocal microscopes, improves accuracy and speed, driving revenue growth. Therapeutic Devices are the fastest-growing subsegment, fueled by innovations in laser and light-based treatments for acne, psoriasis, and skin rejuvenation, with increasing use in clinical and aesthetic settings. Monitoring Devices, including wearable skin sensors, are gaining popularity for chronic condition management, while Imaging Devices enhance visualization and procedural precision. The Others category includes auxiliary devices offering specialized innovations.
Dermatology Diagnostic Devices and Therapeutics Market Insights, By Application
Skin Cancer Diagnosis holds the largest market share, driven by the rising incidence of melanoma and non-melanoma skin cancers globally and the expansion of screening programs. This segment fuels demand for precise diagnostics, promoting the development of AI-assisted and imaging-based technologies. Acne Treatment is the fastest-growing segment, supported by increasing numbers of teenage and adult patients seeking laser and light-based therapies. Psoriasis Management and Eczema Therapy remain steady contributors, utilizing advanced biologics and targeted therapeutic devices to enhance outcomes.
Dermatology Diagnostic Devices and Therapeutics Market Insights, By End User
Hospitals and dermatology clinics dominate the industry, driven by the presence of advanced diagnostic equipment and comprehensive therapeutic services. Rising outpatient dermatology procedures and increased skin cancer screenings further boost demand in these settings. Ambulatory surgical centers are the fastest-growing segment, reflecting patient and insurer preference for minimally invasive, day-care treatments. Homecare settings are emerging, with wearable monitoring devices enabling remote management of chronic skin conditions. Research institutes contribute to innovation, supporting the development of pipeline products and future market expansion. The Others category includes specialized clinics and academic centers fostering niche therapies, further diversifying the market landscape.
Dermatology Diagnostic Devices and Therapeutics Market Trends
Artificial intelligence-assisted diagnostics are increasingly driving market trends, improving accuracy in skin cancer detection, highlighted by the FDA approval of AI-powered dermatoscopes in 2026.
The rise of teledermatology in 2026 is expanding access to dermatology services in rural and underserved urban areas through remote consultations.
Wearable devices that track skin hydration and UV exposure are transforming personalized care, with sales in Asia Pacific increasing by 12% in 2025.
These innovations collectively enhance diagnostic precision, patient engagement, and preventive skin health management, shaping the future of the dermatology diagnostic and therapeutics market.
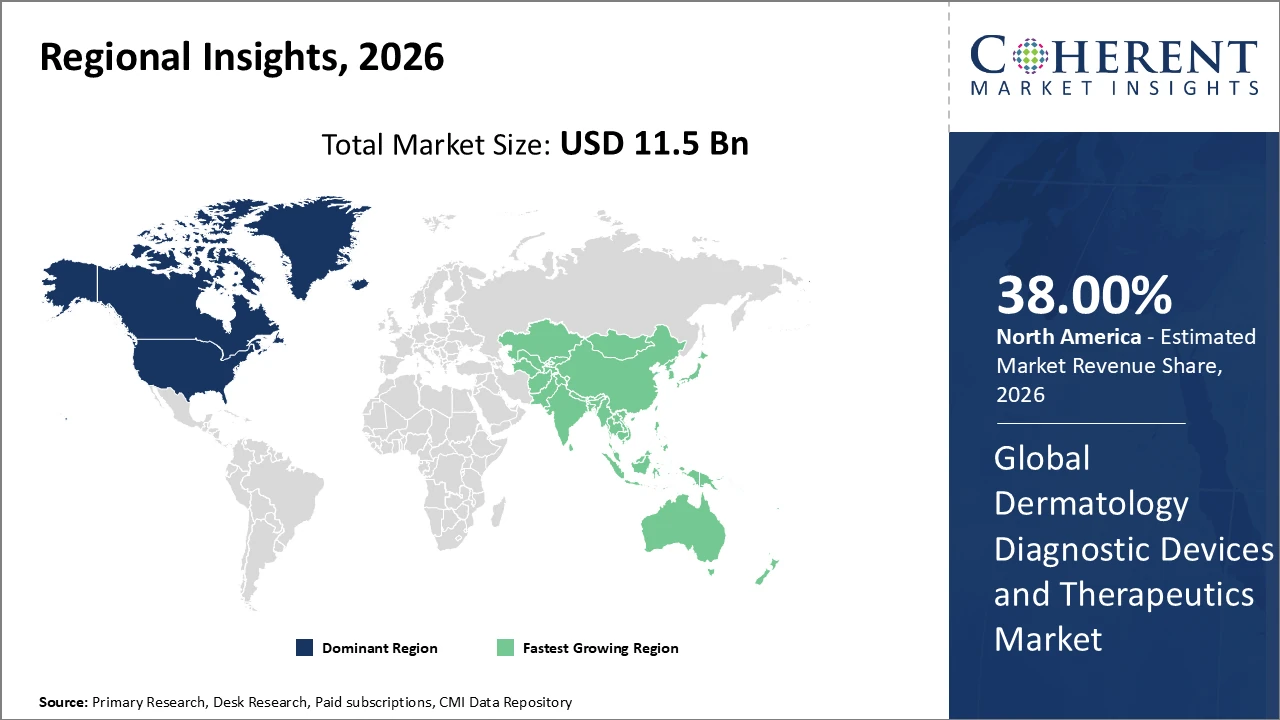
Dermatology Diagnostic Devices and Therapeutics Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Dermatology Diagnostic Devices and Therapeutics Market Analysis and Trends
In North America, the Dermatology Diagnostic Devices and Therapeutics market is dominated by a well-established healthcare infrastructure, favorable reimbursement policies, and the strong presence of major players such as Johnson & Johnson and Hologic Inc. The region accounts for over 38% of the global market share, reflecting high adoption of advanced diagnostic and therapeutic technologies. Extensive public awareness campaigns on skin health and early detection of skin diseases further support market growth. Additionally, ongoing innovations in AI-assisted diagnostics, laser treatments, and minimally invasive procedures are reinforcing North America’s leadership, making it a critical hub for technological advancement and market revenue in the dermatology sector.
Asia Pacific Dermatology Diagnostic Devices and Therapeutics Market Analysis and Trends
Asia Pacific is the fastest-growing region in the Dermatology Diagnostic Devices and Therapeutics market, with a CAGR exceeding 10%, driven by increasing healthcare access in countries like India and China, rising disposable incomes, and government programs promoting skin health awareness. Expanding urbanization and a growing middle class are fueling demand for advanced diagnostic and therapeutic solutions. Both local companies and multinational corporations are investing in regional manufacturing facilities, distribution networks, and sales channels to capitalize on this growth. These combined factors—economic development, healthcare expansion, and corporate investment—are accelerating market adoption and making Asia Pacific a key driver of global market growth.
Dermatology Diagnostic Devices and Therapeutics Market Outlook for Key Countries
USA Dermatology Diagnostic Devices and Therapeutics Market Analysis and Trends
The USA is a major contributor to the Dermatology Diagnostic Devices and Therapeutics market due to the high prevalence of skin disorders, advanced R&D infrastructure, and a supportive regulatory environment. Dermatology clinics recorded a 22% increase in adoption of minimally invasive therapeutic devices between 2024 and 2026, reflecting growing demand for advanced treatments. Public health initiatives emphasizing skin cancer screenings have further boosted the need for diagnostic devices. Leading companies such as Lumenis and Cynosure have strengthened their market presence through technological innovation and strategic penetration, reinforcing the U.S. as a key revenue hub in the global dermatology market.
Germany Dermatology Diagnostic Devices and Therapeutics Market Analysis and Trends
Germany’s Dermatology Diagnostic Devices and Therapeutics market is driven by advanced healthcare infrastructure, high public awareness of skin health, and strong regulatory standards. Hospitals and specialized dermatology clinics dominate demand, supported by routine skin cancer screenings and increasing use of minimally invasive therapeutic devices. Technological innovation, including AI-assisted diagnostics, laser treatments, and imaging systems, enhances early detection and treatment outcomes. Government initiatives promoting preventive healthcare and reimbursement policies further stimulate adoption. Research institutes and local manufacturers contribute to product development and innovation. Overall, Germany represents a mature, technology-driven market emphasizing precision diagnostics, effective therapies, and sustained investment in dermatological care.
Analyst Opinion
The surge in AI-powered diagnostic tools is a key supply-side driver, with 2025 data showing a 27% increase in adoption of AI-integrated dermatoscopes in North American dermatology centers, enhancing melanoma detection and driving a 15% rise in device sales compared to 2024.
On the demand side, rising chronic skin conditions like psoriasis and eczema fuel the need for therapeutic devices, with European dermatology clinics reporting a 22% increase in therapeutic laser device usage in 2026, reflecting patient preference for non-pharmacological treatments.
Micro-indicators show a 14% increase in outpatient dermatology procedures in Asia Pacific in 2026, supported by improved healthcare infrastructure and expanded insurance coverage, enabling higher device utilization.
Nano-size indicators highlight price elasticity in emerging markets, with a 7% year-over-year reduction in device pricing in India and Southeast Asia through 2026, improving market penetration and expanding access in rural areas.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 11.5 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.2% | 2033 Value Projection: | USD 19.8 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Johnson & Johnson, Lumenis Ltd., Alma Lasers, Hologic Inc., Cutera Inc., Sciton Inc., MolecuLight Inc., Cynosure, Medtronic plc | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Dermatology Diagnostic Devices and Therapeutics Market Growth Factors
The rising global incidence of skin cancers, particularly melanoma, is a major growth driver for the Dermatology Diagnostic Devices and Therapeutics market, with WHO reporting a 20% increase in new cases in 2026 compared to 2024. Innovations in non-invasive diagnostic imaging reduce patient discomfort and procedural time, encouraging wider adoption. Government initiatives promoting early skin cancer screening in North America and Europe have further accelerated market penetration. Additionally, growing consumer interest in aesthetic procedures has boosted the therapeutic devices segment, with cosmetic dermatology procedures increasing by 18% in 2026 across Asia Pacific, reflecting expanding demand for both medical and elective skin treatments.
Dermatology Diagnostic Devices and Therapeutics Market Development
In October 2025, Cynosure Lutronic, a global leader in laser and energy-based aesthetic devices, launched eCO2 3D, a versatile fractional CO2 laser. Built on the eCO2 Plus platform, it enhances CO2 laser resurfacing with improved power, precision, and control in a single advanced system.
Key Players
Leading Companies of the Market
Johnson & Johnson
Lumenis Ltd.
Alma Lasers
Hologic Inc.
Cutera Inc.
Cynosure
Sciton Inc.
Medtronic plc
MolecuLight Inc.
Competitive strategies in the Dermatology Diagnostic Devices and Therapeutics market are heavily centered on technological innovation and strategic collaborations. In 2025, Lumenis Ltd. expanded its product portfolio with a new fractional laser system, driving a 12% increase in revenue. Simultaneously, Johnson & Johnson enhanced its market presence through acquisitions of AI-powered diagnostic startups, which accelerated the development of its product pipeline by 2026. These approaches demonstrate how leading companies leverage both innovation and partnerships to maintain competitive advantage, introduce advanced solutions, and capture growing market opportunities in the rapidly evolving dermatology diagnostics and therapeutics sector.
Dermatology Diagnostic Devices and Therapeutics Market Future Outlook
The future outlook for the Dermatology Diagnostic Devices and Therapeutics market is highly promising, driven by increasing prevalence of skin disorders, rising awareness of early detection, and expanding demand for aesthetic procedures. Integration of AI, machine learning, and teledermatology is expected to enhance diagnostic accuracy and accessibility. Minimally invasive therapeutic technologies, including laser and light-based systems, will continue to gain adoption across clinical and cosmetic settings. Emerging markets in Asia Pacific and the Middle East are projected to witness rapid growth due to improving healthcare infrastructure and rising disposable incomes. Overall, innovation, regulatory support, and consumer demand will fuel sustained market expansion.
Dermatology Diagnostic Devices and Therapeutics Market Historical Analysis
The historical analysis of the Dermatology Diagnostic Devices and Therapeutics market shows steady growth driven by rising skin disease prevalence and increasing demand for early detection solutions. From the early 2010s, North America and Europe led market adoption due to advanced healthcare infrastructure, strong regulatory frameworks, and public awareness campaigns on skin cancer and dermatological health. Diagnostic devices, particularly dermatoscopes and imaging systems, dominated the market, while therapeutic lasers and light-based treatments gained traction in cosmetic and clinical applications. Over time, technological advancements, including AI integration and minimally invasive procedures, have improved diagnostic precision, treatment outcomes, and patient convenience, shaping the current market landscape.
Sources
Primary Research Interviews:
Dermatologists, R&D managers, and clinical engineers from companies developing diagnostic devices such as dermatoscopes, imaging systems, and biopsy instruments.
Technical specialists and project managers from firms producing therapeutic devices, including lasers, light-based systems, and wearable monitoring solutions.
Industry experts from equipment providers specializing in AI integration, teledermatology platforms, and minimally invasive dermatology devices.
Magazines:
Dermatology Times – Updates on clinical practice, device innovations, and market trends.
Skin Inc. – Coverage of aesthetic dermatology, laser treatments, and patient care advancements.
MedTech Outlook – Insights on medical device technologies, diagnostics, and therapeutics.
Journals:
Journal of the American Academy of Dermatology – Research on clinical diagnostics, therapies, and treatment outcomes.
Dermatologic Surgery – Studies on minimally invasive procedures, laser treatments, and procedural innovations.
International Journal of Dermatology – Insights on global dermatology trends, device efficacy, and emerging therapies.
Newspapers:
The Wall Street Journal – Corporate activities, mergers, and investments in dermatology technology companies.
Financial Times – Market trends, regulatory developments, and industrial growth impacting dermatology devices.
Reuters – News on medical device adoption, clinical trials, and market performance.
Bloomberg – Company strategies, revenue trends, and global dermatology market growth.
Associations:
American Academy of Dermatology (AAD) – Guidelines, research, and market insights on dermatology practices and devices.
European Academy of Dermatology and Venereology (EADV) – Updates on European dermatology markets, innovations, and regulatory standards.
International Society for Dermatologic Surgery (ISDS) – Insights on surgical and therapeutic device advancements and global trends.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients