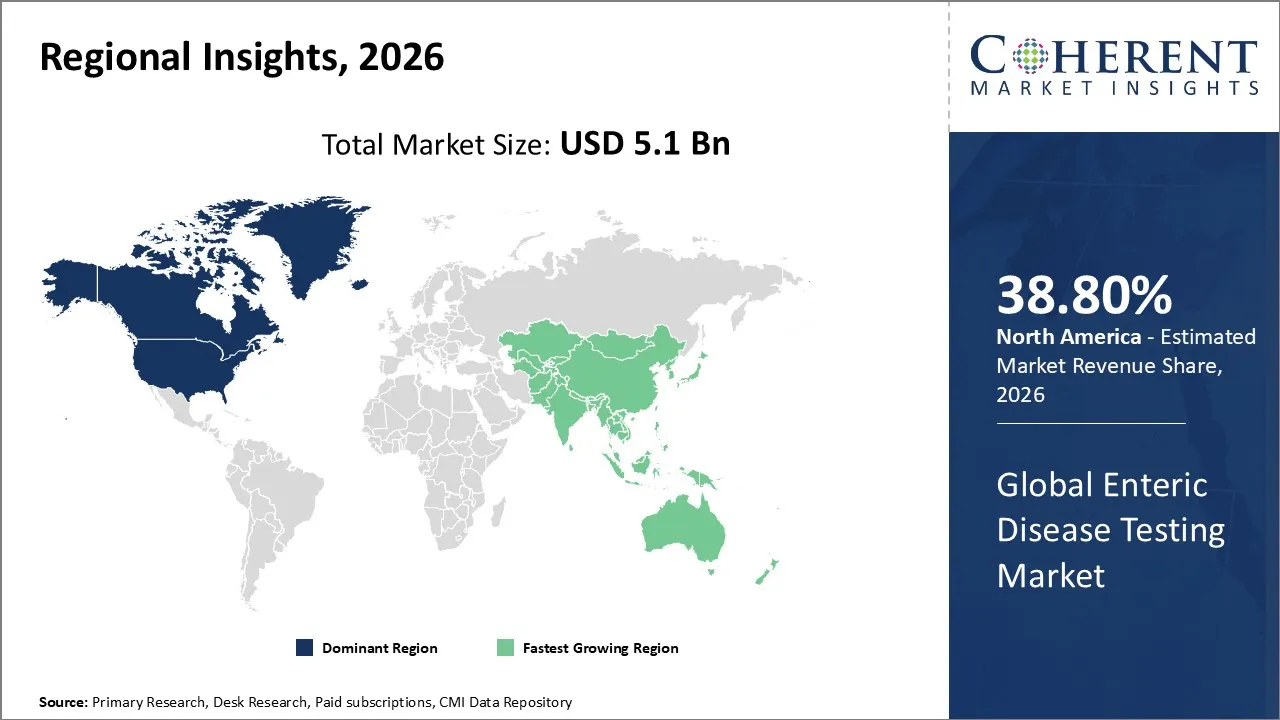
The Enteric Disease Testing Market is estimated to be valued at USD 5.1 Bn in 2026 and is expected to reach USD 7.8 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 4.2% from 2026 to 2033.
The enteric disease testing market delivers diagnostic solutions for gastrointestinal illnesses caused by bacterial, viral, and parasitic pathogens spread through contaminated food, water, or person‑to‑person contact. Rising awareness of early detection, the push for rapid and accurate diagnostics, innovations in molecular testing technologies, and the expansion of public health surveillance programs drive demand. Stringent food safety regulations and the increasing occurrence of enteric infections worldwide further boost the adoption of advanced testing methods in clinical and public health settings.
|
Current Events |
Description and its impact |
|
Global Public Health Initiatives on Enteric Diseases |
|
|
Technological Advancements in Molecular and Point-of-Care Testing |
|
|
Regional Outbreaks and Epidemiological Shifts |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
E.coli Infection hold the largest market share of 31.1% in 2026. Increasing E. coli infections are driving growth in the enteric disease testing market, as contaminated food, water, and poor hygiene practices spread outbreaks. Public health authorities and food safety regulators prioritize early detection, while healthcare providers adopt rapid, reliable diagnostics to manage cases effectively. Advances in molecular assays and multiplex panels improve detection speed and accuracy. Heightened awareness of antimicrobial resistance and the demand for precise pathogen identification motivate wider use of advanced testing methods across clinical, food, and environmental monitoring sectors. For instance, in August 2025, CARB-X awarded USD 3 million to Swiss biotech startup Baxiva AG to advance its multivalent glycoconjugate vaccine targeting major serotypes of invasive extraintestinal pathogenic Escherichia coli.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 38.80% in 2026. In North America, advanced molecular and multiplex diagnostics are transforming the enteric disease testing market by enabling faster and more accurate pathogen detection. Hospitals and laboratories increasingly adopt rapid and point‑of‑care tests to support wider disease surveillance and manage outbreaks effectively. Strong healthcare infrastructure, proactive public health programs, and strict food and water safety regulations drive routine testing. Leading diagnostic manufacturers innovate continuously, and growing investments in new technologies further accelerate the market’s growth and adoption of sophisticated testing solutions. For instance, NIAID, part of the NIH, launched an early-stage trial of CRS3123, an oral antibiotic that targets C. difficile while preserving healthy gut bacteria.
In the Asia Pacific, high rates of food‑ and water‑borne infections from poor sanitation and hygiene are driving demand for enteric disease diagnostics. Governments are enhancing disease surveillance and raising public awareness, while healthcare systems expand infrastructure to support wider testing. Highly populated countries are upgrading laboratory capabilities and adopting advanced molecular and rapid testing technologies to improve detection. Heightened focus on food safety and increased access to healthcare further encourage the use of enteric disease testing throughout the region.
In the United States, healthcare facilities are driving the enteric disease testing market by adopting advanced molecular and rapid testing technologies for faster, more accurate detection of gastrointestinal pathogens. Hospitals and laboratories increasingly implement PCR and multiplex platforms to guide clinical decisions and respond to outbreaks. Public health agencies, including the CDC and FDA, actively promote routine testing and early detection through robust surveillance programs. Rising awareness of foodborne illnesses and continuous investment in diagnostic innovations further propel market growth. For instance, in January 2025, Eli Lilly announced FDA approval of Omvoh for adults with moderate to severe Crohn’s disease, adding to its existing U.S. approval for ulcerative colitis.
In Japan, healthcare providers are driving the enteric disease testing market by adopting rapid and accurate diagnostics to detect food‑ and water‑borne infections such as norovirus and gastroenteritis. They increasingly use molecular techniques, including PCR and DNA‑based testing, to improve pathogen identification and accelerate response times. Expansion of point‑of‑care testing, along with collaborations between government agencies and private diagnostic firms, enhances access and innovation. Public health investments and digital health integration further promote widespread adoption of enteric disease testing across the country.
The market increasingly favors molecular techniques, such as PCR and next-generation sequencing, for detecting enteric pathogens. These methods provide faster, more accurate results than traditional culture-based tests, enabling healthcare providers to manage patients more effectively. Laboratories adopt these technologies to enhance sensitivity, reduce false negatives, and support outbreak tracking. Continuous innovation in molecular platforms also allows simultaneous identification of multiple pathogens, meeting the growing demand for comprehensive diagnostic solutions in clinical and public health settings.
Multiplex and syndromic testing panels are gaining prominence as they allow simultaneous detection of several gastrointestinal pathogens from a single sample. This trend reduces the need for multiple tests, shortens diagnosis time, and lowers operational burdens for laboratories. Healthcare providers appreciate these panels for improving workflow efficiency, enabling rapid decision-making, and supporting epidemiological monitoring. The shift toward integrated testing solutions reflects the need for more efficient and cost-effective approaches to managing enteric disease outbreaks
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 5.1 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.2% | 2033 Value Projection: | USD 7.8 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Alere, Inc, Becton Dickinson & Company, Biomerica, Inc., bioMerieux, Bio-Rad, Cepheid, Coris BioConcept, DiaSorin, Meridian Bioscience, Inc, Quest Diagnostics, and Trinity Biotech |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients