Global Immuno-Oncology Assays Market size is estimated to be valued at USD 6.19 Bn in 2026 and is expected to reach USD 13.35 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 11.6% from 2026 to 2033.
To learn more about this report, Request Free Sample
The global immuno-oncology assays market trend is witnessing a high growth due to rising incidences of cancer globally and increasing adoption of targeted therapies for cancer treatment. Immuno-oncology is an evolving field in oncology that harnesses the power of the immune system to detect and destroy cancer cells. With promising clinical trial results, improved understanding of tumor microenvironment and biomarkers, new product approvals, and growing emphasis on precision medicine, the global immuno-oncology assays market is estimated to witness a healthy growth during the forecast period.
|
Event |
Description and Impact |
|
Technological Advances in Precision Diagnostics |
|
|
Regulatory Environment and Clinical Trial Trends |
|
|
Economic and Industry Investment Activities |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Even though these users are not core customers, IVF clinics sometimes use immuno-oncology assays as a part of their research activities. These assays aid in studying the immune system’s behavior, providing information that may be useful for reproductive treatments and reproductive immunology.
This first group is the most advanced in their use of immuno-oncology assays. Extensive efforts are being made by companies to incorporate these assays into the processes of drug development and more refined techniques in immunotherapy and precision medicine. Their application ranges from preliminary investigations to clinical trials, underscoring the strategic significance of these assays in therapeutic innovation.
University and research institutions play a very important role in advancing immuno-oncology technologies. They are working towards the validation of the assays and the identification of new biomarkers to advance diagnostics as well as rational clinical pathways supported by evidence.
Biobanks perform a fundamental task for the development of an assay by submitting well-characterized biological specimens. Their participation guarantees reliability of the assay from the scientific research and clinical research viewpoint concerning immuno-oncological studies.
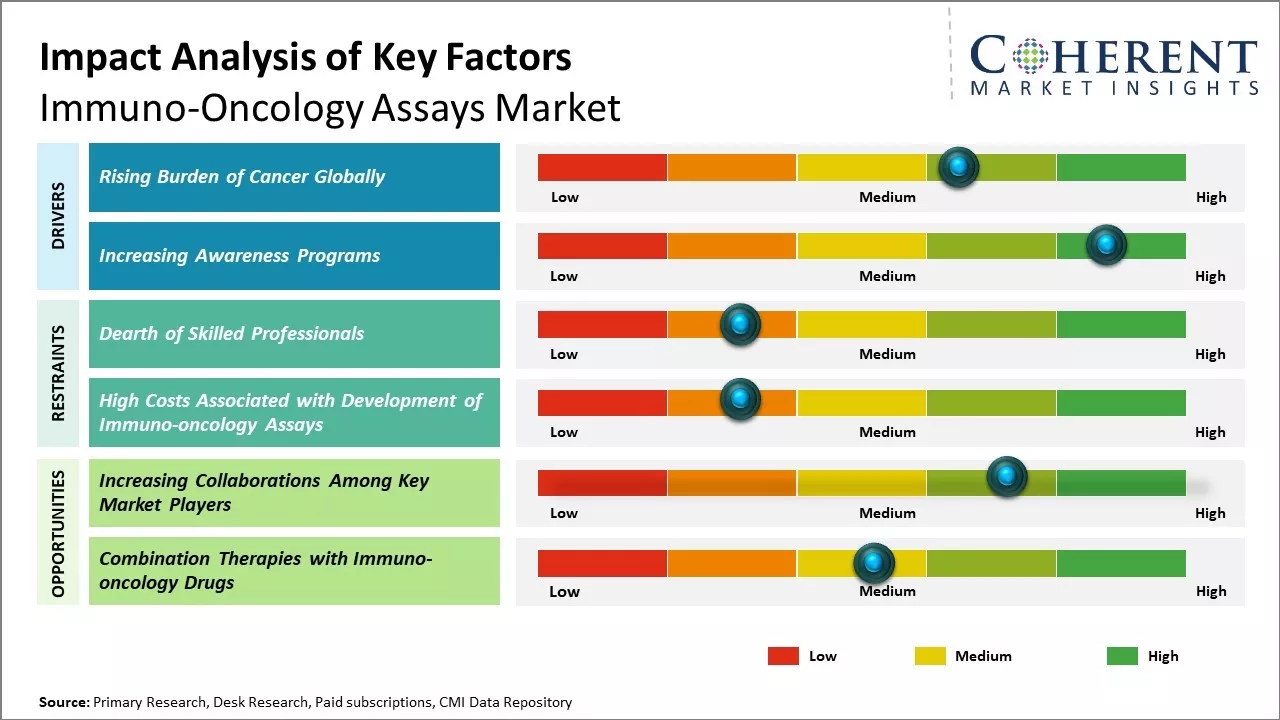
Rise in burden of cancer across the globe is expected to augment growth of the global immuno-oncology assays market over the forecast period. Immuno-oncology (IO) is a class of treatment designed to boost the body's natural immune defense against cancer. For instance, in March 2023, according to the data published by the Centers for Disease Control and Prevention, it was estimated that a total of 32,320 new ovarian cancer cases will be diagnosed globally in the year 2023.
Moreover, according to the World Health Organization (WHO), cancer was a leading cause of death worldwide, accounting for nearly 10 million deaths in 2020, or nearly one in six deaths. The most common cancers were breast, lung, prostate, and colon and rectum cancer. This in turn is expected to increase demand for immuno-oncology assays around the world.
Increasing inorganic activities, such as awareness programs, is expected to drive the market growth over the forecast period. For instance, in June, 2022, the U.S. Department of Health and Human Services and the Centers for Medicare & Medicaid Services (CMS) announced a new model aimed at improving cancer care for Medicare patients and lowering health care costs.
Increasing adoption of inorganic growth strategies such collaborations by the key market players is expected to drive the market growth over the forecast period. For instance, in May, 2021, the University of California, San Francisco (UCSF) in collaboration with the San Francisco Veteran Affairs (VA) Health Care System, the Prostate Cancer Foundation, launched the Drew Foundation Precision Oncology Center of Excellence, providing precision medicine treatments to veterans facing prostate cancer.
Product segment is sub-segmented into consumables, software. The consumables segment is estimated to hold 67.4% of the market share in 2026, owing to the increasing incidences of colorectal cancer.
According to International Agency for Research on Cancer, March 2022, in 2020, almost 2 million cases were diagnosed, for example, alcohol consumption was responsible for more than 160 000 new cases of colorectal cancer in 2020. It is the second most common cause of cancer death, leading to almost 1 million deaths per year.
To learn more about this report, Request Free Sample
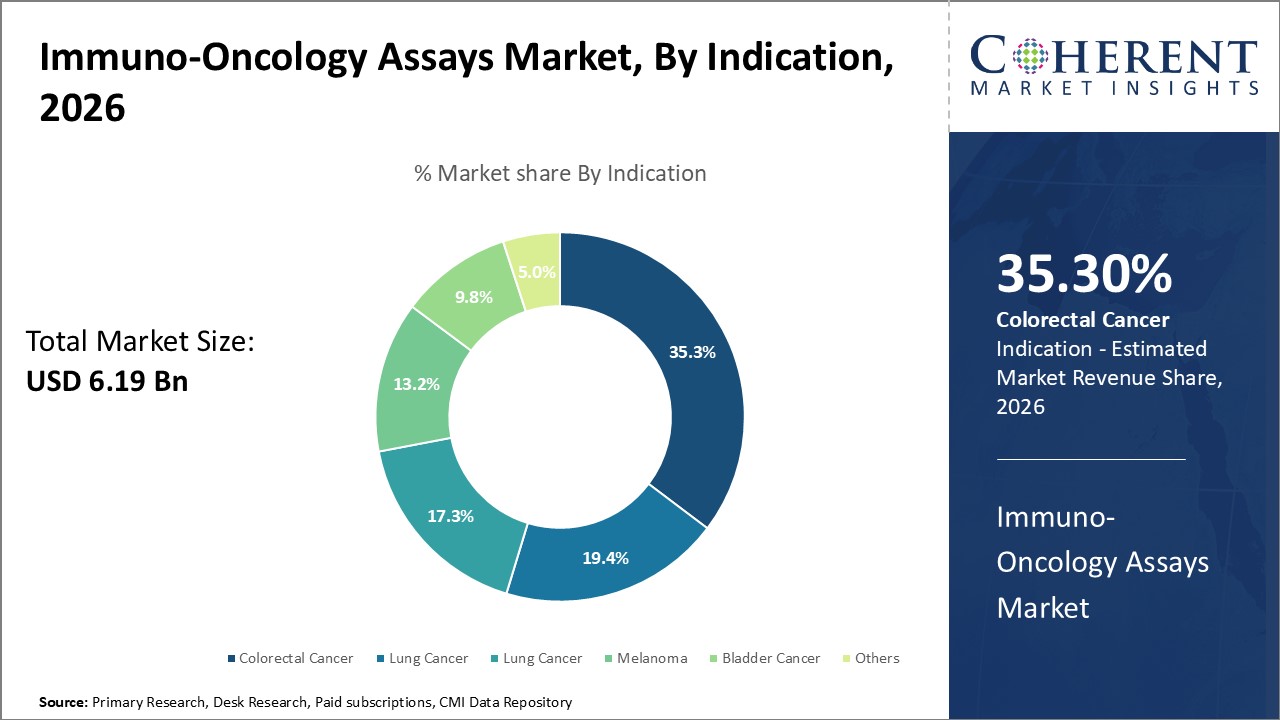
Indication segment is sub-segmented into colorectal cancer, lung cancer, melanoma, bladder cancer, others. The colorectal cancer segment is estimated to hold 35.3% of the market share in 2026 owing to the increasing incidences of colorectal cancer.
The burden of colorectal cancer is the highest in Asia, where more than half of all cases and deaths are recorded. China alone accounts for more than half a million new cases and more than 280 000 deaths per year. Japan records the second highest number of deaths from colorectal cancer, almost 60 000 per year.
Application segment is sub-segmented into clinical diagnostics, and research and development. The research and development segment is estimated to hold 55.3% of the market share in 2026 due to the extensive translational research activities in this field. Pharmaceutical and biotech companies heavily rely on various immunoassay platforms to study immune mechanisms, discover and validate immune-related biomarkers, and conduct preclinical safety and efficacy testing of immunotherapies.
National cancer institutes and academic medical centers too employ immuno-oncology assays while investigating tumor immunobiology, immunomonitoring, and novel treatment strategies. The need to deepen understanding of complex immune-tumor interactions, identify predictive and prognostic biomarkers along with responding patient subsets is driving the demand for customized research applications of these assays.
Additionally, they aid evaluation of immune effects during nonclinical and early clinical phases of drug development. With ongoing efforts to harness immunity against cancer, the R&D application segment will likely maintain its lead position in the coming years backed by significant investments in immuno-oncology innovations.
To learn more about this report, Request Free Sample
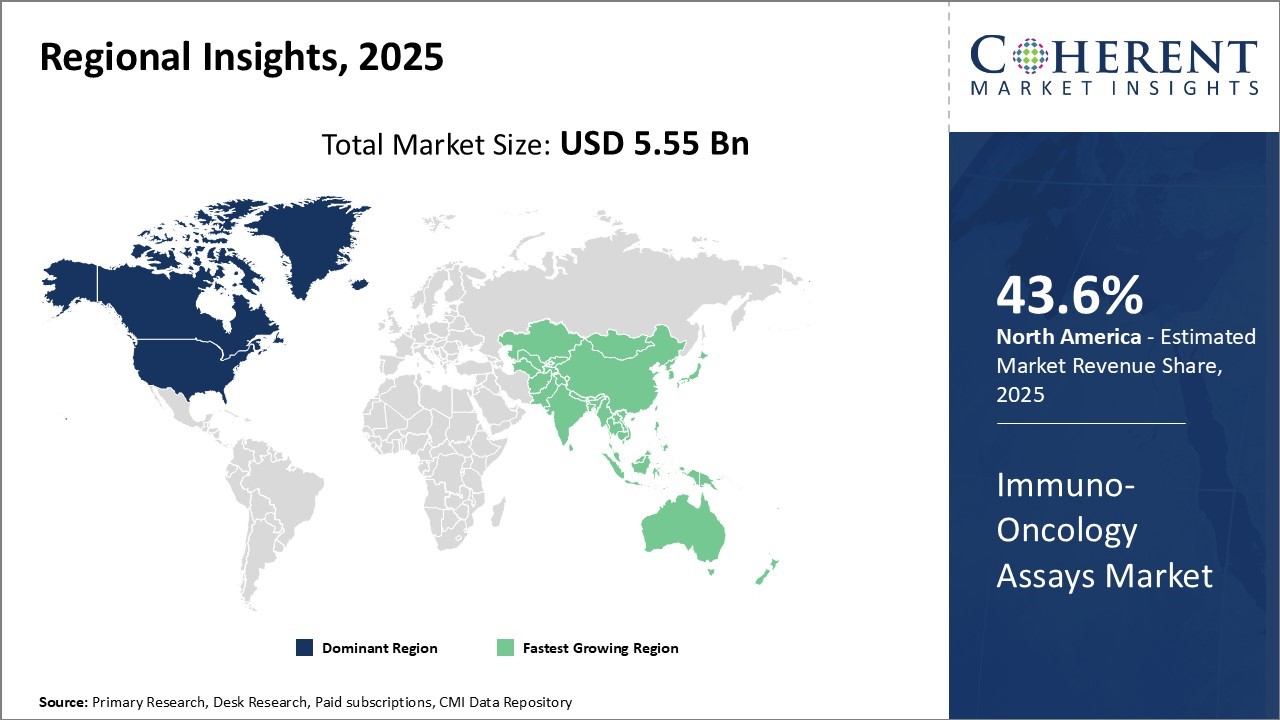
North America is projected to dominate the global Immuno-Oncology Assays Market, accounting for 43.6% of the market share in 2026. This dominance is driven by the increasing incidence of cancer and the rising elderly population in the U.S. In 2024, the American Cancer Society (ACS) estimated that nearly 1.9 million new cancer cases would be diagnosed in the U.S., along with over 600,000 cancer-related deaths.
With growing government initiatives, advancements in cancer research, and strong healthcare infrastructure, the adoption of immuno-oncology assays in the region is accelerating, especially for the diagnosis and treatment of solid tumors and hematologic cancers.
Asia Pacific is expected to experience robust growth in the global Immuno-Oncology Assays Market by 2026, driven by the region’s increasing cancer incidence and a rapidly aging population. India, for instance, is anticipated to see a 12.5% rise in cancer cases within the next five years, with projections indicating that 1.5 million people will be affected by cancer by 2026.
The Indian Council of Medical Research (ICMR) reported in 2024 that cancer cases in the country have been on the rise due to changing lifestyle factors, which is expected to result in higher demand for immuno-oncology assays as part of the nation’s healthcare strategy.
The U.S. continues to lead the Immuno-Oncology Assays Market in North America, contributing significantly to global market share. Recent advancements in immunotherapy and cancer diagnostics have further driven the need for more sophisticated diagnostic tools. In 2024, major healthcare providers and research institutions in the U.S. have ramped up investments in immuno-oncology research.
As the U.S. government implements increased funding for cancer research through the National Cancer Institute (NCI), the integration of advanced immuno-oncology assays into routine clinical practice is gaining momentum.
India is emerging as a significant market in the Asia Pacific region, with the expected increase in cancer cases driving the demand for immuno-oncology assays. The Indian Council of Medical Research (ICMR) forecasted a sharp rise in cancer prevalence by 2026, which is prompting the government to implement nationwide cancer awareness and screening programs.
With a focus on boosting diagnostic accuracy and early cancer detection, India’s healthcare infrastructure is increasingly incorporating immuno-oncology assays, especially in urban centers with improving access to cutting-edge medical technologies.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 6.19 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 11.6% | 2033 Value Projection: | USD 13.35 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Charles River Laboratories, F. Hoffmann-La Roche Ltd, llumina, Inc., Agilent Technologies, Inc., Thermo Fisher Scientific Inc., Merck KGaA, Invivoscribe, Inc., QIAGEN, PerkinElmer Inc., Sartorius AG, Bio-Rad Laboratories, Inc., Bruker Spatial Biology, Inc., Nexelis, Reaction Biology, Crown Bioscience, HTG Molecular Diagnostics, Inc., Axion BioSystems, Inc, and BioTherapeutics LLC |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Immuno-oncology is emerging as a novel approach to cancer treatment through the stimulation of the body's own immune system. It is a formidable weapon against cancer as it has the ability to harness the body's natural defenses against infection. It is one of the most promising areas of research in the world of anti-cancer therapeutics.
Share
Share
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients