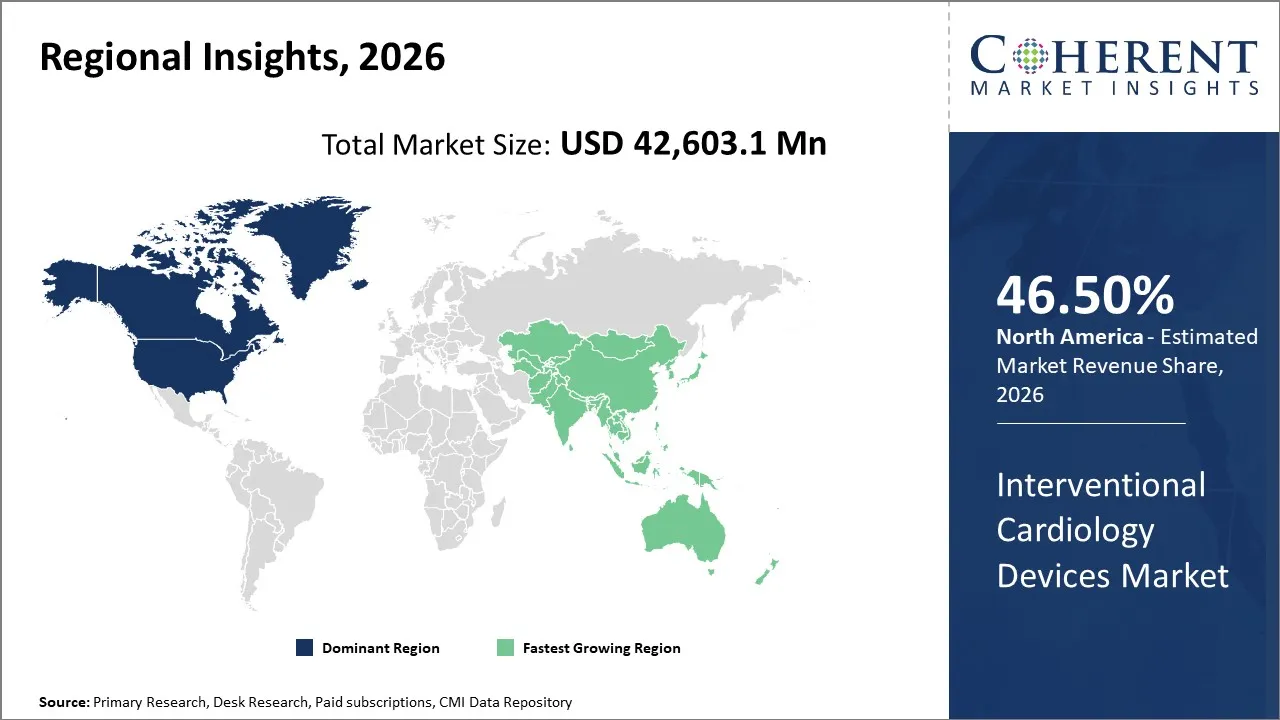
The global interventional cardiology devices market size is expected to reach approximately USD 42,603.1 Mn in 2026 and USD 70,171.1 Mn by 2033, growing at a CAGR of 7.5% throughout the forecast period (2026-2033).
Interventional cardiology devices are medical devices used to diagnose and treat cardiovascular disease and lower risk of complications like excessive bleeding and infection without need for open-heart surgery. It is used to carry out minimal invasive treatment which no large surgical cuts and reduced trauma to the body. Interventional cardiology devices are minimally invasive, catheter-based tools used to diagnose and treat cardiovascular diseases. The interventional cardiology devices market is driven by expanding use of transcatheter therapies in structural heart disease and better clinical outcomes in complex interventions through imaging-guided therapy.
|
Current Event |
Description and its Impact |
|
Increasing Regulator Approval of New Devices |
|
|
The market is moving toward smarter, software-assisted procedures |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Based on device, stents are expected to lead the market, accounting for a revenue share of 44.5% in 2026. Stents are used in almost every angioplasty as it directly restores blood flow and keep arteries open. High prevalence of coronary artery disease (CAD) increases demand for stent implantation. Stents are the standard of care for treating coronary blockages preferred by cardiologists worldwide. Many healthcare systems cover stent procedures encourages higher adoption. Hence, stents segment dominates the market due to its essential role in PCI procedures, wide clinical adoption, high procedure volume and continuous innovation.
By end user, the hospitals segment is slated to account for a prominent market share of 66.5% in 2026. Most procedures like angioplasty and stent placement are primarily performed in hospitals which leads to highest devices utilization. Hospitals are equipped with catheterization labs, advanced imaging systems, ICU and emergency facilities which are essential for complex cardiac interventions. Hospitals are open 24/7 and are capable to handle emergency cases like heart attacks. Hospitals are more likely to be covered under public and private insurance schemes which encourages patients to undergo procedures in hospitals. Hospitals can afford and adopt latest stents and imaging technologies. Hospitals provide surgery, diagnosis and post-operative monitoring that means complete treatment ecosystem in one place which is consider to be major factor driving the growth of the market.
To learn more about this report, Request Free Sample
North America is expected to dominate the global interventional cardiology devices market, accounting for a share of 46.50% in 2026. North America region leads the market due to large patient pool suffering from coronary artery disease and heart attacks, advanced healthcare infrastructure, early adoption of advanced technologies and strong presence of key market players. The favourable reimbursement policies and well-developed insurance systems especially in the U.S. supports the dominance in the global market. Increasing launch and approval of new cardiovascular devices by the key players operating in the North America region has driven the growth of the market in the near future. North America, particularly the U.S., spends heavily on healthcare supports adoption of expensive, high-end devices. Furthermore, availability of highly trained interventional cardiologists ensures success of cardiac surgery and strong research and development activities in the North America has driven the overall market growth.
Asia Pacific is expected to grow at the fastest CAGR in the interventional cardiology devices market during the forecast period. Rural and semi-urban areas in Asia Pacific region which are still underpenetrated have huge growth potential as access improves, which drives the growth of the market. Expansion of multinational companies in Asia-Pacific region leads to competitive pricing, better product availability and technology transfer. Increasing government initiatives by launching public health programs targeting cardiovascular diseases and investments in insurance schemes supports market growth.
The U.S. is the dominant country in the North America interventional cardiology devices market due to strong presence of major market players, favorable reimbursement policies, high healthcare expenditure, and skilled healthcare professionals. The U.S. country has high prevalence of cardiovascular diseases which creates strong and consistent demand for interventional procedures. Furthermore, presence of highly equipped hospitals and advanced catheterized labs enables high procedure volumes.
The China country leads the Asia Pacific interventional cardiology devices market owing to higher incidence of cardiovascular diseases, improving healthcare infrastructure, strong government funding and private sector investment. China is investing in improving healthcare infrastructure and growth of local medical device companies which leads to cost-effective devices and increased market penetration. China has one of the largest populations globally rising cases of coronary artery disease (CAD) and hypertension and diabetes creating massive demand for interventional procedures.
Source: Steriotaxis
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 42,603.1 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.5% | 2033 Value Projection: | USD 70,171.1 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Abbott Laboratories, Terumo Medical Corporation, Boston Scientific Corporation, Cardinal Health, Medtronic, Cook Medical, SINOMED, Biotronik SE & Co. KG, and B. Braun Melsungen AG |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Rising advancements in technology has significantly driven the growth of the market. Innovations such as drug-eluting stents (DES), advanced guidewires and catheters, IVUS and OCT imaging systems have helped in caring out operation smoothly. Technology advancement has improved safety and success rates which increases adoption of latest version interventional devices.
For instance, in February 2026, FUJIFILM Middle East FZE, a subsidiary of FUJIFILM Holdings Corporation, focusing on healthcare, photographic materials, and digital imaging solutions based in UAE, revealed the introduction of its ground-breaking Interventional Cardiology Business Line in the Middle East and Africa. A major turning point in the company's dedication to improving healthcare and democratizing cardiac care throughout the area and beyond will be reached with the official launch, which will occur during the event at the FUJIFILM Middle East booth. FUJIFILM Middle East is working with Innvolution Healthcare, a well-known supplier of top-notch precision diagnostic tools and individualized care for cardiovascular conditions, as part of the project. The Middle East and Africa will have more access to cutting-edge interventional cardiology technologies thanks to this calculated decision, which will equip hospitals and healthcare providers with superior, cutting-edge interventional cardiac solutions.
Increasing approval and launch of new products has driven the market growth significantly. New products (e.g., advanced drug-eluting stents, improved guidewires) offer lower risk of restenosis and complications and better efficacy. Doctors prefer latest version, safer devices which increases the demand for newly launched products. Innovative devices allow treatment of more complex coronary lesions with high-risk and previously untreatable patients which increases the total number of procedures performed. Companies launch products with better flexibility, deliverability, and precision which encourages hospitals to upgrade equipment and fuel the market growth. For instance, in December 2025, Vesalio Inc., a company focused on developing thrombectomy solutions, revealed the enVast, the first and only clot retriever specially approved for mechanical thrombectomy in the cardiac circulation, will soon go on sale in the United States after receiving FDA 510(k) clearance. For patients with a large thrombus burden (LTB), enVast redefines coronary thrombectomy by introducing a tried-and-true method of clot capture and removal. A frequent and difficult finding in patients receiving initial percutaneous coronary intervention (PCI) is LTB. Improving clinical outcomes, limiting myocardial damage, and lowering procedural complications all depend on the timely and efficient restoration of coronary flow.
Reduced surgical trauma is one of the biggest growth drivers. Strong preference for catheter-based interventions over open-heart surgery has driven the growth of the interventional cardiology devices market. Moreover, minimally invasive procedures provide benefits such as lower risk, faster recovery, shorter hospital stay. When compared to traditional surgery, minimally invasive techniques frequently enable patients to recuperate faster and resume their regular activities sooner. These therapies are more appealing from a clinical and operational standpoint since hospitals gain from shorter inpatient stays and improved procedural turnover. In market terms, Shift drives growth by boosting adoption of catheters, stents, balloons, guidewires, and transcatheter devices
AI-powered tools improve precision in angioplasty and real-time decision-making. Technologies like IVUS (intravascular Ultrasound) and OCT (Optical Coherence Tomography) enhances procedural accuracy and patient outcomes. Increasing adoption of advanced imaging technologies and AI integration has made cardiac procedures more accurate, safer, and more efficient. Advanced imaging tools such as 3D mapping, CT-based planning, and real-time fluoroscopic guidance help physicians visualize vessel structure, plaque burden, lesion complexity, and device positioning more clearly. Advanced imaging technologies and AI integration make the complex procedures such as bifurcations, calcified vessels, chronic total occlusions, coronary lesions, and structural heart conditions easier to manage.
Expansion of ambulatory surgical centers (ASCs) has estimated to create significant opportunity for the growth of the market in the near future. Shift toward outpatient procedures due to lower cost and faster discharge creates new demand outside traditional hospitals. For instance, March 2025, according to the data published by the Ambulatory Surgery Center Association, in the US, there are more than 6,500 ambulatory surgery centers with Medicare certification; some estimates place the number of operational ASCs at close to 10,000. With 250 new ASCs debuting in 2023, the number of Medicare-certified ASCs increased by 2.5%. As of early 2026, orthopedics accounted for about 30% of the market, with single-specialty facilities holding around 61% of the market (2025). With 549 and 543 active centers, respectively, Los Angeles and New York have the highest ASC densities as of July 2025.
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients