The PD-1 and PD-L1 inhibitor market is estimated to be valued at USD 73.87 Bn in 2026 and is expected to reach USD 245.25 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 18.7% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
The market is witnessing positive growth due to increasing demand for immunotherapies for treatment of various cancer types. PD-1 and PD-L1 inhibitors have demonstrated high efficacy in treatment of lung cancer, kidney cancer, melanoma and other cancers. These drugs have better side effect profile compared to conventional chemotherapy. Ongoing clinical trials evaluating efficacy of PD-1 and PD-L1 inhibitors for new cancer indications such as gastric cancer, cervical cancer and pancreatic cancer are also boosting the market. Moreover, approval of combination therapies of PD-1 and PD-L1 inhibitors with other immunotherapies is further driving the market growth. However, high cost of therapy is one of the factors hindering widespread adoption of PD-1 and PD-L1 inhibitors.
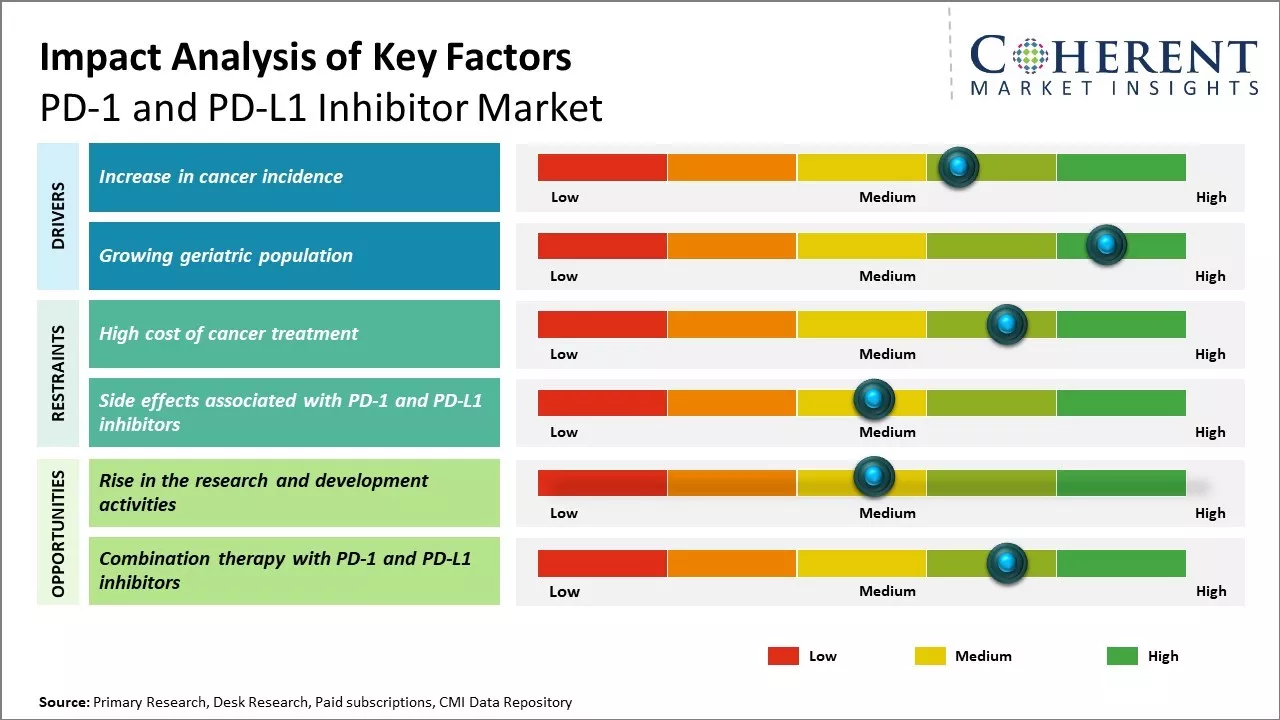
Increase in cancer incidence
The rise in incidence/prevalence of cancer across the globe is expected to boost growth of the global PD-1 and PD-L1 inhibitor market during the forecast period. For instance, PD-1/PD-L1 inhibitors are important immune checkpoint inhibitors (ICIs) used as front-line treatments for several types of cancer. According to the World Health Organization (WHO) February 2022 press release, cancer was one of the leading causes of death worldwide, accounting for around ten million deaths in 2020, or nearly one in six deaths. The most common cancers are breast, lung, colon and rectum and prostate cancers. Thus, there is an increase in demand for PD-1 and PD-L1 inhibitors worldwide.
Get actionable strategies to beat competition: Request Free Sample
Growing geriatric populationGrowing geriatric population around the world is expected to augment growth of the global PD-1 and PD-L1 inhibitor market during the forecast period. For instance, according to the Union for International Cancer Control (UICC), elderly (people aged 65 years and more) are 11 times more likely to develop cancer, compared to younger people. According to the United Nations January 2023 press release, there are over 761 million people aged 65 or older in 2021, a number that is projected to reach 1.6 billion by 2050.

To learn more about this report, Request Free Sample
Market Challenges: High cost of cancer treatmentThe unsustainably high cost of cancer treatment is a major restraint on the growth of the PD-1 and PD-L1 inhibitor market. Cancer treatments have become exorbitantly expensive over the past few decades, to the point where they cause financial distress for many patients and their families . Healthcare systems and insurance companies around the world struggle with providing coverage and reimbursement for such expensive therapies. Many are unable to bear the brunt of the costs, leaving patients in the lurch.
Market Opportunities: Rise in the research and development activities
Rise in research and development activities are expected to offer significant growth opportunities for players in the PD-1 and PD-L1 inhibitor market. For instance, market players are increasingly investing in research and development activities to meet the increasing demand. Researchers at Johns Hopkins Kimmel Cancer Center are leading the way in developing novel immunotherapies called anti-PD-1 and anti-PD-L1 for people with advanced melanomas. The aim of the therapy is not to kill cancer cells directly, but to block a pathway that protects tumor cells from components of the immune system that are capable and ready to fight cancer.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
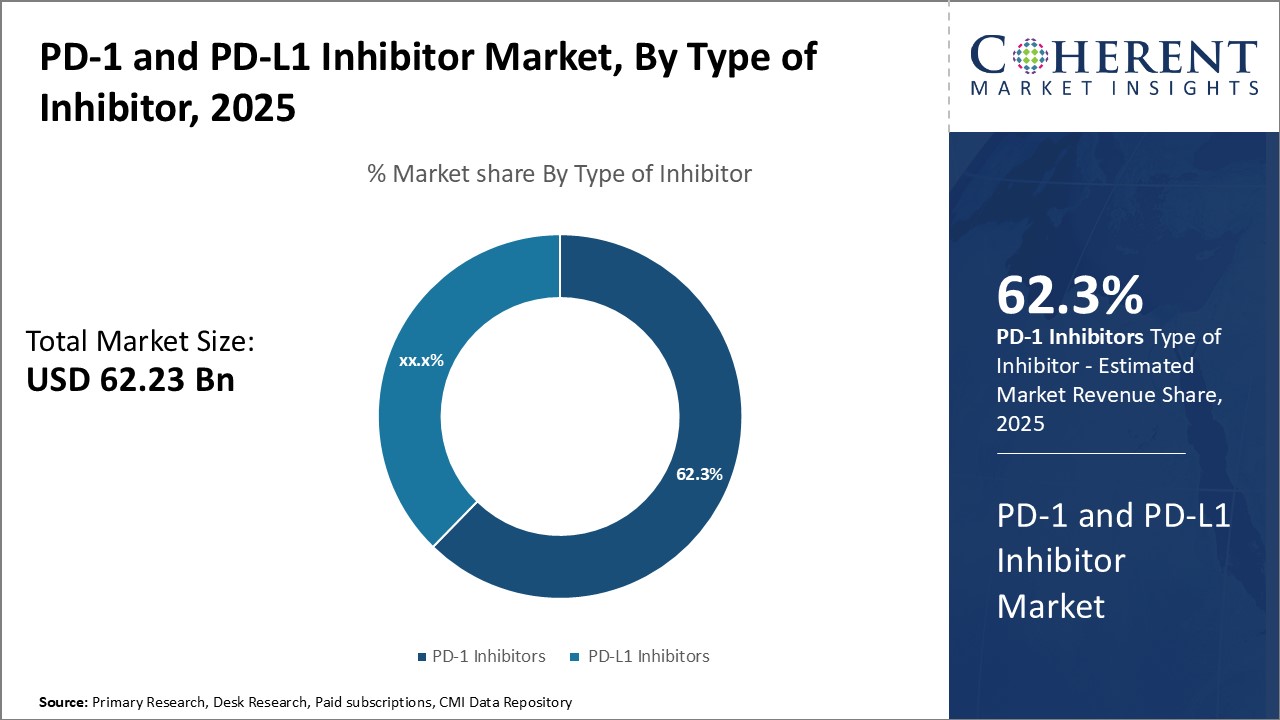
Insights, By Type of Inhibitor: PD-1 Inhibitors holds the highest share of the market owing to wide applicabilityThe type of inhibitor segment includes PD-1 Inhibitors and PD-L1 Inhibitors. PD-1 Inhibitors is anticipated to hold 63.3% of the market share in 2026 due to their versatility in treating various cancer types. By blocking the PD-1 receptor, these inhibitors help unleash the immune system’s attack on cancer cells. This targeted approach offers more precision than conventional treatments while reducing side effects like nausea, hair loss or organ damage from chemotherapy. A key factor driving the demand for PD-1 inhibitors is their approval for treating various cancers beyond their pioneering use in melanoma. FDA approvals for diseases like lung cancer, lymphoma and head & neck cancer have expanded the eligible patient population manifold. Developers are continually exploring new cancer candidates as well, holding promise to enlarge approved use in the future. Furthermore, early success rates from clinical trials have encouraged oncologists to prescribe PD-1 inhibitors as the first line of treatment in multiple settings.
To learn more about this report, Request Free Sample
Insights, By Cancer Type: Growing prevalence of lung cancer across the globe
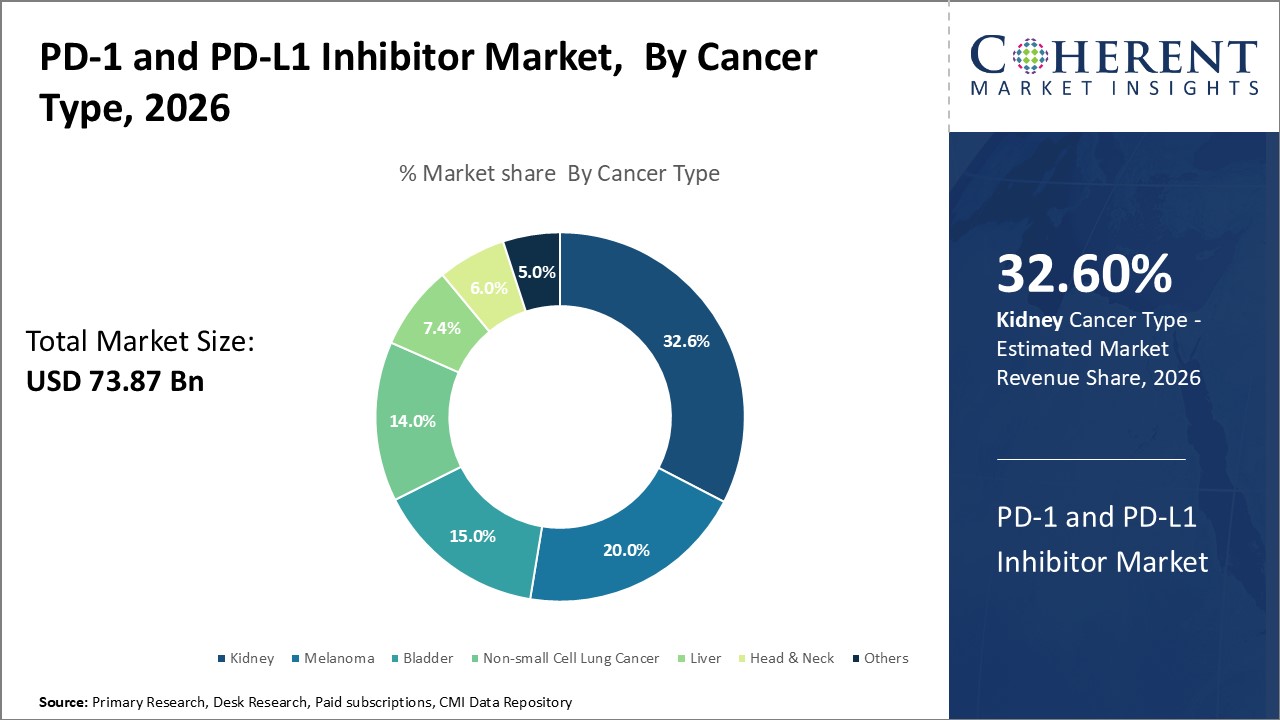
The cancer type segment includes kidney, melanoma, bladder, non-small cell lung cancer, liver, head & neck, and others. Non-small cell lung cancer holds the highest share of the PD-1 and PD-L1 inhibitor market and is projected to hold 32.6% of the market share in 2026. This is primarily because lung cancer remains the leading cause of cancer-related deaths globally with a poor five-year survival rate of less than 20%. Most diagnoses still occur in advanced stages when surgical options are limited. However, immunosuppressive PD-1/PD-L1 inhibitors have shown promising results even in late-stage non-small cell lung cancer patients who failed previous treatments. They work by suppressing mechanisms enabling cancer to hide from immune surveillance. Post-approval studies continue to validate efficacy across histological subtypes as well as in biomarker-selected patient groups. This expands the addressable population for second- and third-line usage of such drugs.
Insights, By Distribution Channel: Convenience and Compliance drives hospital pharmacies growth
The distribution channel segment includes hospital pharmacies, retail pharmacies, online pharmacies. Hospital pharmacies accounts for the highest share of the PD-1 and PD-L1 inhibitor market and is projected to hold 40.7% of the market share in 2026. Hospital visits allow for integrated care where a dedicated oncology team closely monitors therapy progress and addresses any side effects promptly. Patients find this reassuring especially during initial doses when most adverse reactions occur. It also aids compliance as hospital staff can directly supervise administering intravenous medications on schedule. Moreover, hospital pharmacies are better equipped than retail setups to safely handle riskier substances like immunotherapies requiring refrigeration or preparation under sterile conditions. They facilitate direct billing of treatments to insurance, reducing co-pay hassles as well. The growing trend of hospitals providing cancer centers and infusions under one roof enhances this value proposition significantly.
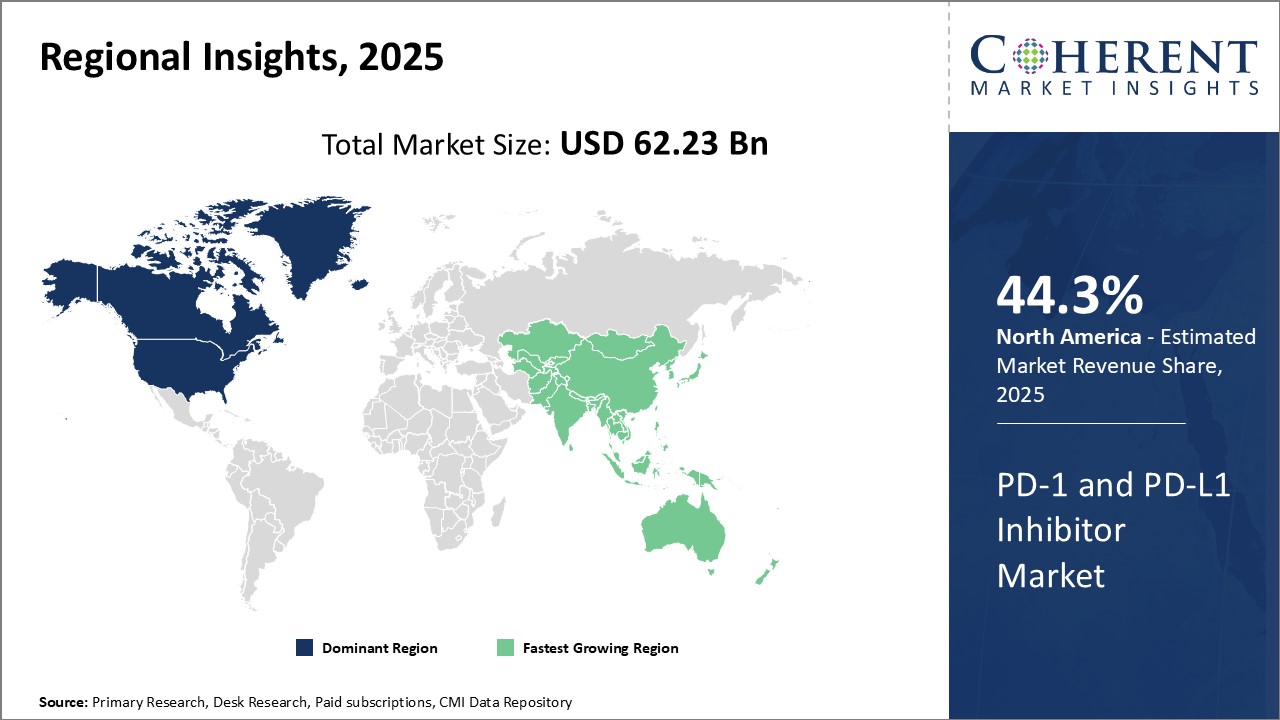
Need a Different Region or Segment? Request Free Sample
North America dominates the global PD-1 and PD-L1 inhibitor market and is expected to continue its stronghold over the forecast period. North America is estimated to hold 44.3% of the market share in 2026 owing to a highly developed healthcare sector and large cancer patient pool in the region. Players in this region have extensively invested in R&D and are constantly launching innovative drug formulations with better efficacy and fewer side effects. This has resulted in rapid adoption of immune checkpoint inhibitors across various cancer indications in the country. Additionally, supportive regulatory guidelines and favorable reimbursement policies have encouraged pharmaceutical manufacturers to seek early market approvals for drug candidates. This has ensured high commercial availability and affordability of approved therapies, driving the regional market. Furthermore, higher per capita healthcare expenditure has empowered cancer patients to opt for premium treatment options like immune checkpoint inhibitors.
The Asia Pacific region has emerged as the fastest growing market for PD-1 and PD-L1 inhibitors globally. Rapid economic development, growing geriatric population susceptible to cancer and rising healthcare investments are some of the key factors propelling the Asia Pacific market. Nations like China, Japan and South Korea have well-established pharmaceutical industries that are investing heavily in innovation and manufacturing of biosimilars. This has made immune checkpoint therapies more affordable for local patients compared to imported brands.
PD-1 and PD-L1 Inhibitor Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 73.87 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 18.7% | 2033 Value Projection: | USD 245.25 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
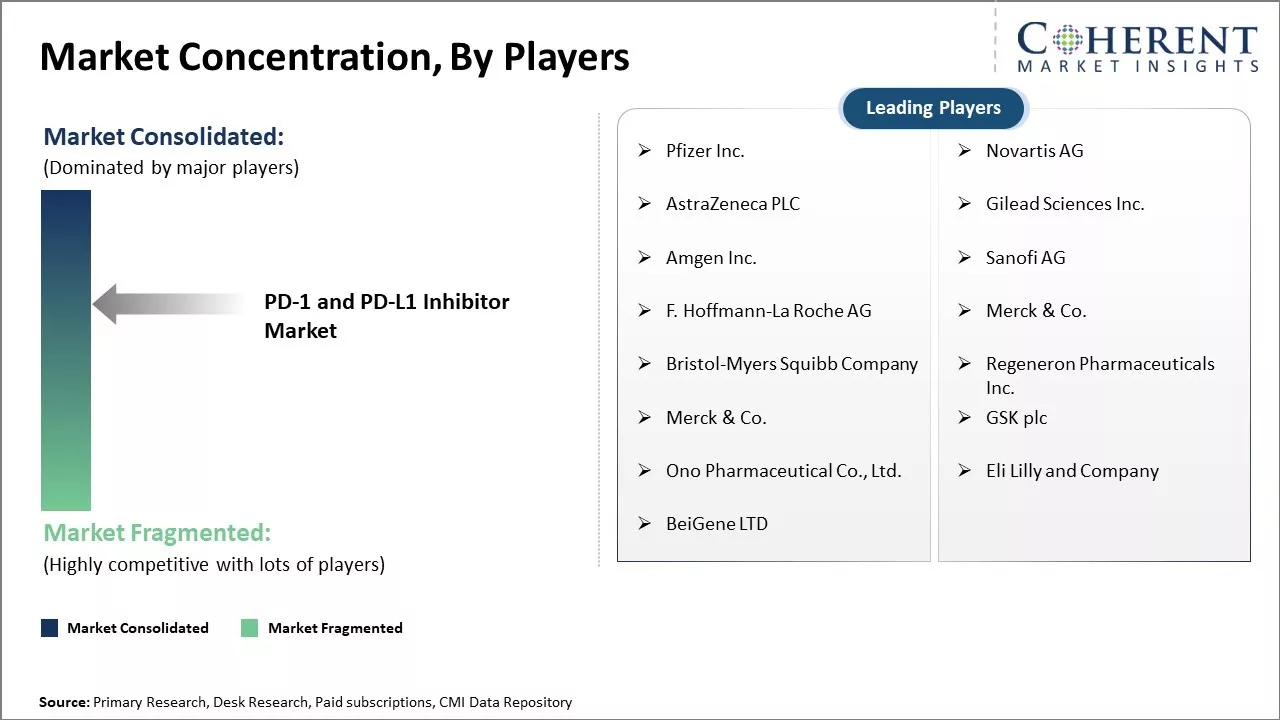
| Companies covered: |
Pfizer Inc., Novartis AG, AstraZeneca PLC, Gilead Sciences Inc., Amgen Inc., Sanofi AG, F. Hoffmann-La Roche AG, Merck & Co., Bristol-Myers Squibb Company, Regeneron Pharmaceuticals Inc., Merck & Co., GSK plc, Ono Pharmaceutical Co., Ltd., Eli Lilly and Company, and BeiGene LTD |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition:The PD-1 and PD-L1 inhibitor market refers to drugs and therapies that target the immune checkpoint receptors PD-1 (programmed cell death protein 1) and PD-L1 (programmed death-ligand 1). These receptors, when activated, help cancer cells avoid detection and destruction by the immune system. PD-1 and PD-L1 inhibitors work by blocking these receptors and thereby helping the immune system recognize and attack cancer cells.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients