Short Bowel Syndrome Market Size and Forecast – 2026 – 2033
The global Short Bowel Syndrome market is projected to grow from around USD 2.5 billion in 2026 to about USD 7.9 billion by 2033, expanding at a 19.3% CAGR. This reflects strong growth driven by rising gastrointestinal disorders and advanced therapies.
Global Short Bowel Syndrome Market Overview
Short Bowel Syndrome (SBS) is a rare but serious malabsorption disorder caused by the surgical removal, congenital absence, or disease-related loss of a significant portion of the small intestine. With reduced intestinal length, the body cannot adequately absorb nutrients, fluids, and electrolytes, leading to diarrhea, dehydration, weight loss, and malnutrition. SBS commonly results from conditions such as Crohn’s disease, mesenteric ischemia, trauma, or congenital defects. Management includes specialized diets, vitamin and mineral supplementation, anti-diarrheal medications, parenteral nutrition, and in severe cases, intestinal transplantation. Advances in therapies, including GLP-2 analogs, have improved intestinal adaptation and long-term patient outcomes.
Key Takeaways
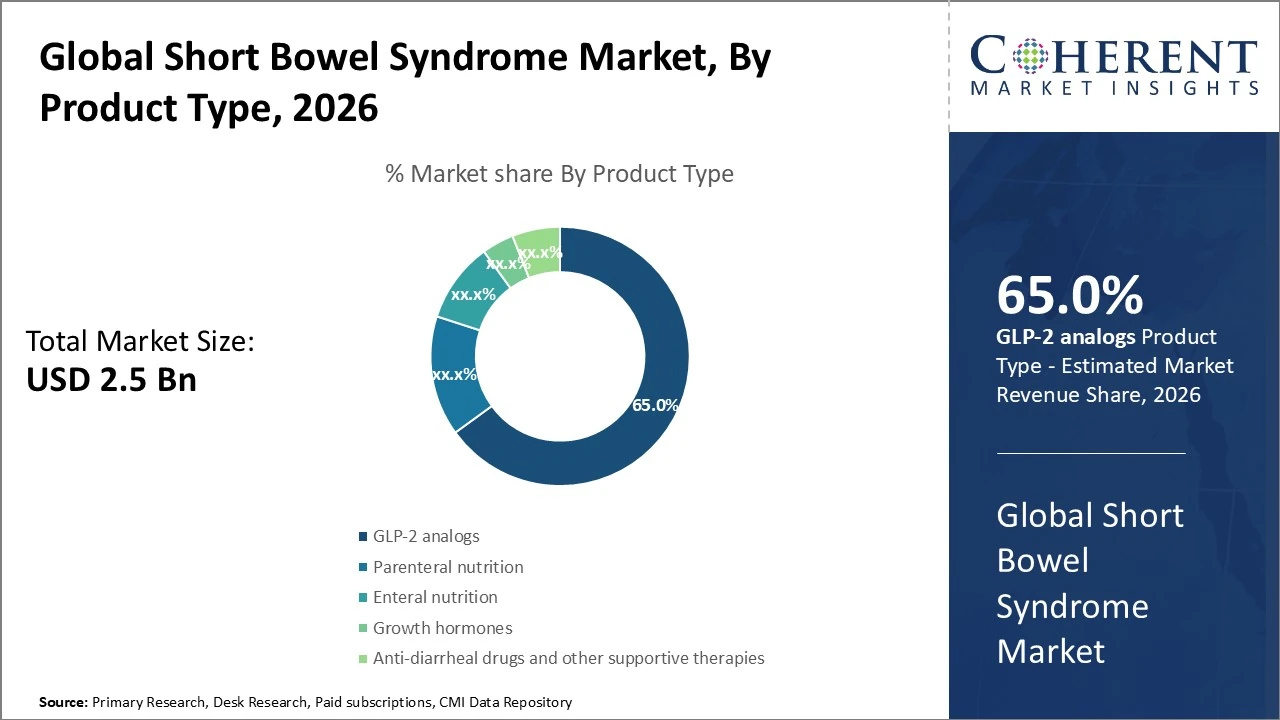
The Short Bowel Syndrome (SBS) market, by product type, is led by GLP-2 analogs, accounting for approximately 65% of total revenue.
The Short Bowel Syndrome market, by end-user, is led by hospitals, which account for about ~55% of total market share.
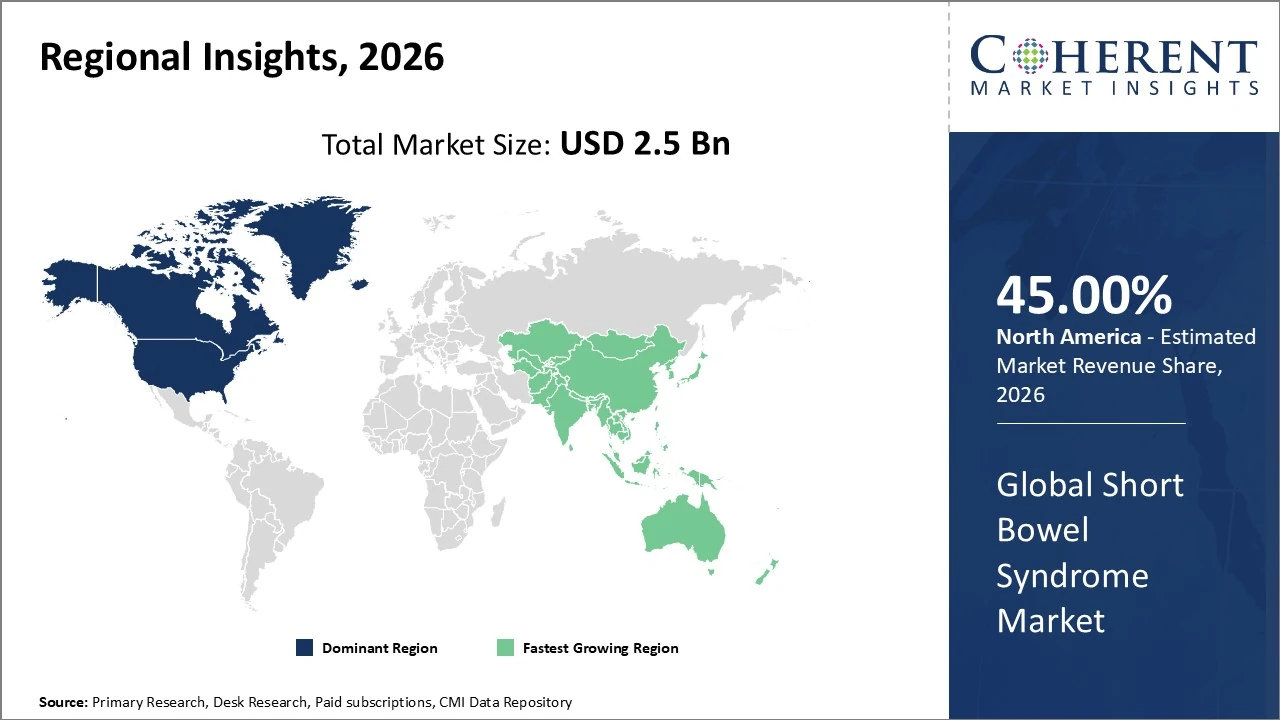
North America dominates the Short Bowel Syndrome (SBS) market, accounting for approximately 45% of global market revenue.
The Asia Pacific Short Bowel Syndrome (SBS) market is the fastest-growing region, expanding at an estimated ~9.5% CAGR.
The United States dominates the Short Bowel Syndrome market, accounting for the largest share within North America (~75% regional share).
The Germany Short Bowel Syndrome (SBS) market is valued at approximately USD 72.29 million in 2026.
Short Bowel Syndrome Market Segmentation Analysis
To learn more about this report, Request Free Sample
Short Bowel Syndrome Market Insights, By Product Type
The Short Bowel Syndrome (SBS) market, by product type, is led by GLP-2 analogs, accounting for approximately 65% of total revenue due to their effectiveness in improving intestinal absorption and reducing parenteral nutrition dependence. Parenteral nutrition (PN) holds around 15%, as it remains essential for severe SBS patients. Enteral nutrition contributes nearly 10%, supporting dietary management. Growth hormones represent about 4%, used selectively to enhance intestinal adaptation. Anti-diarrheal drugs and other supportive therapies collectively account for roughly 6%. Market shares vary slightly by region and treatment guidelines but reflect current therapeutic adoption trends.
Short Bowel Syndrome Market Insights, By End-User
The Short Bowel Syndrome market, by end-user, is led by Hospitals, which account for about ~55% of total market share due to their role in acute care, specialized treatment initiation, and parenteral nutrition administration. Home healthcare settings represent roughly ~30%, driven by increasing patient preference for long-term outpatient and home-based nutritional support. Specialty clinics hold about ~15%, catering to focused SBS management and rare disease expertise. These segments reflect current care delivery trends, with homecare growing fastest as portable therapies and multidisciplinary support expand beyond traditional hospital settings.
Short Bowel Syndrome Market Trends
Increasing clinical use of glucagon-like peptide-2 analogs (e.g., teduglutide) is driving improved patient outcomes and reducing dependence on long-term parenteral nutrition.
Expansion of home parenteral and enteral nutrition services is boosting market demand as patients and providers favor outpatient management.
Enhanced dietary solutions, tailored nutrient formulations, and multidisciplinary care programs are becoming key components of comprehensive SBS management.
Short Bowel Syndrome Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Short Bowel Syndrome Market Analysis and Trends
North America dominates the Short Bowel Syndrome (SBS) market, accounting for approximately 45% of global market revenue due to advanced healthcare infrastructure, early adoption of innovative biologic therapies, and strong reimbursement frameworks. The United States leads, with high utilization of GLP-2 analogs like teduglutide and well-established home parenteral nutrition programs driving demand. Early diagnosis, rising awareness, and substantial rare disease support policies further bolster regional growth. Telehealth and home-based care expansion enhance long-term management. Continued R&D investment and clinical trial activity solidify North America’s leadership, while payer support for orphan drugs sustains market momentum.
Asia Pacific Short Bowel Syndrome Market Analysis and Trends
The Asia Pacific Short Bowel Syndrome (SBS) market is the fastest-growing region, expanding at an estimated ~9.5% CAGR through 2033 as healthcare access, diagnostics, and specialist care improve across China, Japan, India, South Korea, and Australia. Increased gastrointestinal surgeries and rising SBS incidence are key demand drivers. The region holds around ~23% of the global SBS market share, reflecting significant growth potential against more mature markets. Adoption of advanced therapies like GLP-2 analogs and enhanced parenteral nutrition, along with expanding reimbursement and rare-disease awareness initiatives, further accelerate regional uptake and investment.
Short Bowel Syndrome Market Outlook for Key Countries
USA Short Bowel Syndrome Market Analysis and Trends
The United States dominates the Short Bowel Syndrome market, accounting for the largest share within North America (part of ~75% regional share) due to advanced healthcare infrastructure, strong rare‑disease reimbursement, and a high diagnosed patient pool supporting growth. U.S. SBS market size is projected to reach an estimated ~USD 1.9 billion by 2033, driven by widespread use of GLP‑2 analog therapies that reduce dependence on parenteral nutrition and improve outcomes. Increasing gastrointestinal surgeries, improved diagnosis, and expanding home parenteral nutrition programs further fuel uptake. Emerging therapies and R&D investments are key growth drivers ahead.
Germany Short Bowel Syndrome Market Analysis and Trends
The Germany Short Bowel Syndrome (SBS) market is valued at approximately USD 72.29 million in 2026, with a projected ~15.2% CAGR through the forecast period, reflecting strong growth in rare disease treatment adoption and biotech investment. Germany benefits from high healthcare spending, advanced clinical research, and supportive reimbursement for orphan drugs, driving demand for GLP‑2 analogs and nutritional therapies. Hospitals and home care services lead care delivery, while increasing clinical trials and novel therapies expand local treatment options. Germany’s share within Europe’s SBS market underscores its role as a key regional growth hub backed by regulatory support and pharmaceutical innovation.
Analyst Opinion
Analysts highlight GLP‑2 analog therapies as the primary revenue driver, improving patient outcomes and reducing long-term parenteral nutrition reliance.
Expansion of home parenteral and enteral nutrition services is expected to accelerate adoption and reduce hospital dependency.
Asia Pacific and Latin America are seen as high-growth regions due to improving healthcare infrastructure, increasing surgeries, and growing awareness.
Tailored dietary and nutritional therapies are gaining importance, enhancing SBS management and patient quality of life.
Orphan drug incentives and rare disease reimbursement policies strongly influence market dynamics and investment decisions.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.5 Billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 19.3% | 2033 Value Projection: | USD 7.9 Billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Takeda Pharmaceutical, Nestlé Health Science, NPS Pharmaceuticals, Ferring Pharmaceuticals, Ipsen, Mallinckrodt Pharmaceuticals | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Short Bowel Syndrome Market Growth Factors
The SBS market is expanding due to rising gastrointestinal surgeries and increased incidence of Crohn’s disease, mesenteric ischemia, and congenital intestinal disorders. Advancements in therapies, especially GLP‑2 analogs, improve intestinal absorption and reduce reliance on parenteral nutrition, driving adoption. Growing awareness and early diagnosis enhance treatment initiation, while home healthcare and outpatient parenteral nutrition programs support long-term management. Supportive policies, orphan drug incentives, and reimbursement schemes encourage innovation and market entry. Additionally, nutritional and personalized dietary solutions improve patient quality of life, further fueling demand. Together, these factors contribute to steady market growth through 2033.
Short Bowel Syndrome Market Development
In July 2025, Russian biotech firm Biocad initiated Phase II trials of its original drug BCD-261 for inflammatory bowel diseases, including Crohn’s disease and moderate-to-severe ulcerative colitis, following approval from the Russian Ministry of Health. The company expects to invest over 3 billion rubles in the drug’s development and research.
Key Players
Leading Companies of the Market
Takeda Pharmaceutical
Nestlé Health Science
NPS Pharmaceuticals
Ferring Pharmaceuticals
Ipsen
Mallinckrodt Pharmaceuticals
Key players in the Short Bowel Syndrome market include Takeda Pharmaceutical, Nestlé Health Science, NPS Pharmaceuticals, Ferring Pharmaceuticals, Ipsen, Mallinckrodt Pharmaceuticals. These companies lead in GLP‑2 analogs, parenteral nutrition, and specialized dietary therapies, driving innovation and market growth.
Short Bowel Syndrome Market Future Outlook
The SBS market is expected to experience robust growth through 2033, driven by rising gastrointestinal surgeries, increasing prevalence of Crohn’s disease, and growing awareness of rare intestinal disorders. GLP‑2 analogs and novel biologics will continue to dominate, improving patient outcomes and reducing long-term dependence on parenteral nutrition. Expansion of home healthcare, outpatient services, and personalized nutrition therapies will support broader accessibility. Emerging markets, particularly in Asia Pacific and Latin America, are anticipated to witness accelerated adoption. Continuous R&D, favorable reimbursement policies, and regulatory support for orphan drugs will further stimulate market development, innovation, and long-term revenue growth.
Short Bowel Syndrome Market Historical Analysis
Historically, the SBS market was limited by lack of effective therapies and high dependence on parenteral nutrition, with patient management largely confined to hospitals. Early interventions focused on supportive care, including nutritional supplementation and anti-diarrheal medications, resulting in modest market growth. The introduction of GLP-2 analogs in the last decade marked a turning point, enhancing intestinal absorption and enabling reduced hospital stays. North America and Europe historically dominated the market due to advanced healthcare infrastructure, higher diagnosis rates, and reimbursement support. Gradual awareness, clinical research, and improvements in home parenteral nutrition programs laid the foundation for today’s expanding global SBS market.
Sources
Primary Research Interviews:
Pharmaceutical & Biotech Companies
Healthcare Professionals
Hospital Administrators & Clinical Directors
Regulatory & Policy Experts
Databases:
PubMed / Medline
ClinicalTrials.gov
World Health Organization
Journals:
Gastroenterology
Journal of Parenteral and Enteral Nutrition
Clinical Nutrition
Alimentary Pharmacology & Therapeutics
Newspapers:
The Wall Street Journal
Financial Times
The Guardian
The New York Times
Associations:
American Gastroenterological Association (AGA)
European Society for Clinical Nutrition and Metabolism (ESPEN)
American Society for Parenteral and Enteral Nutrition (ASPEN)
Pediatric Intestinal Failure and Rehabilitation Consortium (PIFRC)
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients