Viral Conjunctivitis Drugs Market size is estimated to be valued at USD 394.9 Mn in 2026 and is expected to reach USD 566.8 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 5.3% from 2026 to 2033.
Viral Conjunctivitis Drugs Market growth is rapid due to the rising number of product launches by the key market players, and is expected to propel over the forecast period. For instance, in February 2021, Bausch + Lomb, an eye health products company, announced the launch of Alaway Preservative-Free (ketotifen fumarate ophthalmic solution 0.035%) antihistamine eye drops. It is indicated to relieve eye itchiness caused by eye flu (conjunctivitis). Moreover, it is the first over-the-counter (OTC) preservative-free antihistamine eye itch relief drop approved by the U.S. Food and Drug Administration (FDA).
|
Event |
Description and Impact |
|
U.S.-China Trade Tensions and API Tariffs |
|
|
Regulatory & Therapeutic Advancements |
|
|
Emerging Market Strategies |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The anti-histamines segment is anticipated to lead the global viral conjunctivitis drugs market demand with 37.2% share in 2026, accounting for the largest share among drug classes. This is due to the increasing incidence of viral conjunctivitis, especially in densely populated cities and institutions where outbreaks are common. Anti-histamines are increasingly being used as first-line therapies for symptomatic relief due to their effectiveness in relieving allergic and inflammatory eye symptoms.
These shifts are being made because of pharmaceutical innovations that focus on improving ocular comfort and side-effect profiles. The combination of easy access and a growing preference for self-medication supports the segment’s leading position. Increased R&D aimed at enhancing the tolerability and efficacy of anti-histamine therapies will further drive demand throughout the forecast period.
To learn more about this report, Download Free Sample
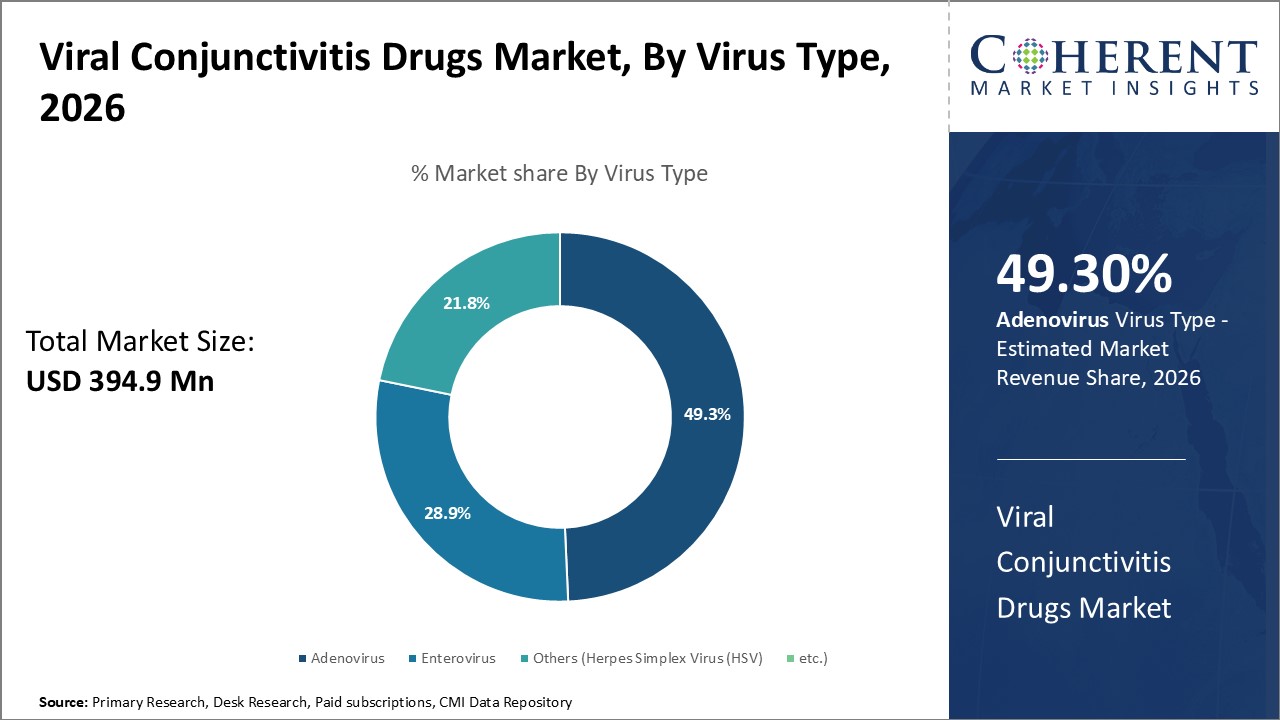
The adenovirus segment is projected to dominate the viral conjunctivitis drugs market forecast by virus type, driven by the high incidence rate of adenoviral infections as the most common viral cause of conjunctivitis. Adenoviruses are especially virulent and easily spread, causing widespread epidemics in schools, military installations, and medical facilities.
This raises the need for more effective therapies. In addition, the recent FDA approvals and new product launches for the treatment of adenoviral ocular infections highlight the focus of the market. A number of biotechnology companies are focusing on the development of drugs for adenovirus infections which is boosting the pipeline of possible therapies. The segment is expected to continue dominating during the forecast period due to the advancement of clinical trials and the introduction of new treatments into the market.
By 2026, hospital pharmacies are projected to capture the largest share of the Viral Conjunctivitis Drugs Market value by distribution channel due to sales volume. This is driven by the increasing number of hospitals globally and particularly in emerging markets with rapidly expanding healthcare infrastructure. Hospital pharmacies are crucial for dispensing prescription viral conjunctivitis treatments, especially for complex cases necessitating a physician’s watchful care.
Furthermore, the confidence derived from hospital-based supply and compliance with stringent laws adds to the importance of this channel. Since most patients with viral conjunctivitis are seen in clinics, ready availability of medications through hospital pharmacies strengthens their critical role in driving market growth.
To learn more about this report, Download Free Sample
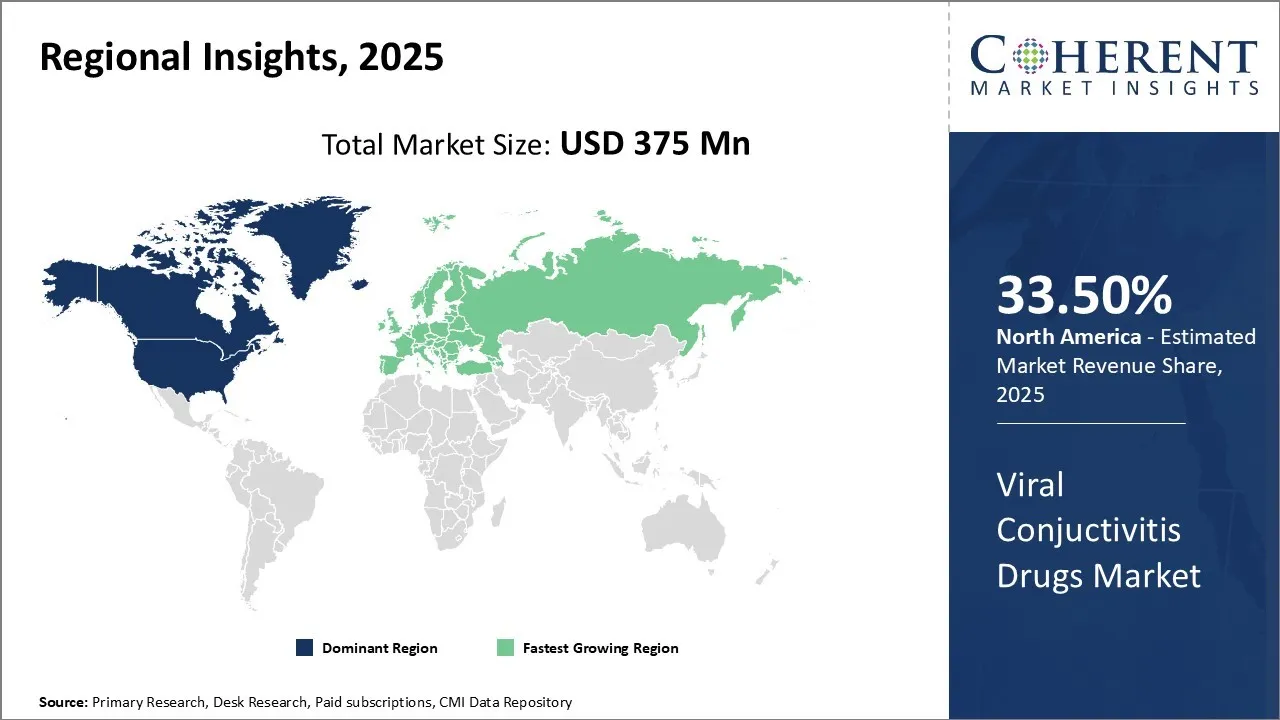
North America is expected to dominate the global viral conjunctivitis drugs market trend in 2026, accounting for an estimated 33.50% share. This leadership is reinforced by well-established regulations, a high rate of eye infections in the population, and frequent use authorization of drugs by the U.S. Food and Drug Administration (FDA). The United States remains an innovation powerhouse as the pharmaceutical industry is aggressively pursuing treatments for viral eye disorders.
One such example is the FDA approval of Alcon Pataday Once Daily Relief and Pataday Twice Daily Relief eye drops for use in allergic conjunctivitis on February 2020. Such approvals demonstrate the growing scope of over-the-counter treatments in the region enhances patient access, and stimulates market expansion. Coupled with its highly sophisticated healthcare system and robust pharmaceutical R&D, North America’s attention to eye care makes it a long-term dominant player in the market for drugs treating viral conjunctivitis.
Asia Pacific is poised to be the fastest-growing region in the viral conjunctivitis drugs market, driven by densely populated countries such as China and India where viral eye infections are more prevalent. The market is driven by enhanced access to healthcare resources, greater understanding of eye care, and an increase in investment for manufacturing generic drugs. You may recall that local pharmaceutical firms are expanding their production facilities and forming regional partnerships to market low-cost antiviral and anti-inflammatory drugs.
These initiatives, coupled with the government’s investments in healthcare, including the diagnostic infrastructure and comminicable diseases public health programs, boost uptake in the region. The prevalence of e-commerce as well as telemedicine is aiding in the availability of eye care products in rural and underserved regions as well.
Europe’s viral conjunctivitis drugs market is witnessing steady growth, supported by the region’s aging population and increasing susceptibility to ocular infections. The pharmaceutical sectors in Germany, the U.K., and France are considered leaders in the region owing to their mature industries and robust regulatory systems.
Stricter consumer demand for better tolerability has shifted the focus of European players to innovation in preservative-free eye formulations and advanced drug delivery systems. Still, the rate of new product launches in the region may be held back by reimbursement policies and longer approval timelines relative to North America. Academic and biotech partnerships, however, continue to foster innovation and strengthen the region’s market presence.
The U.S. stands as the largest contributor to the North American viral conjunctivitis drugs market, fueled by an advanced clinical research ecosystem, frequent FDA drug approvals, and high patient awareness levels. The presence of global eye care leaders like Alcon and Allergan ensures continuous innovation and market responsiveness. The trend toward OTC formulations, driven by consumer demand for self-care medical device, further boosts growth potential.
China is at the center of Asia Pacific's growth because its population is densely situated, has increasing healthcare spending, and is rapidly urbanizing, all of which increases the risk of viral eye infections. Supported by government initiatives, local pharmaceutical firms are working to manufacture generic antivirals and antihistamines on a large scale. Because of its attention to both access and cost, China serves as a major contributor to the growth of Viral Conjunctivitis drugs market around the world.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 394.9 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.3% | 2033 Value Projection: | USD 566.8 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AbbVie Inc., Takeda Pharmaceutical Company Limited, Bausch Health Companies Inc., Panoptes Pharma Ges.m.b.H, NovaBay Pharmaceuticals, Inc., Nicox, NanoViricides Inc., Novartis International AG, Pfizer Inc., Johnson & Johnson, GlaxoSmithKline Plc., Santen Pharmaceutical Co.Ltd., Alcon and Okogen Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Viral conjunctivitis, or pinkeye, is a common, self-limiting condition that is typically caused by an adenovirus. Other viruses that can be responsible for conjunctival infection include herpes simplex virus (HSV), varicella-zoster virus (VZV), picornavirus (enterovirus 70, Coxsackie A24), poxvirus (molluscum contagiosum, vaccinia), and human immunodeficiency virus (HIV). This increasing prevalence of viral conjunctivitis is expected to drive market growth over the forecast period. For instance, on February 15, 2023, according to the report published in National Center for Biotechnology Information, approximately 6 million new cases of viral conjunctivitis occur annually in the U.S.
One of the significant factors influencing the growth rate of the viral conjunctivitis drugs market is the growing healthcare expenditure, which helps in improving its infrastructure. For instance, according to the International Health Care System of the U.S., in June 2020, U.S. government organizations aim to improve the healthcare infrastructure by increasing funding, setting legislation and national strategies, cofounding and setting basic requirements and regulations for the Medicaid program.
Furthermore, in December 2022, according to the report published by the Centers for Medicare & Medicaid Services, reported U.S. health care spending grew 2.7% in 2021, reaching US$ 4.3 trillion, or US$ 12,914 per person, while as a share of the nation's gross domestic product, health spending accounted for 18.3%.
Similarly, in November 2022, the Canadian Institute for Health Information reported that the total health spending in Canada was US$ 331 billion in 2022, or US$ 8,563 per Canadian, while health expenditure represented 12.2% of Canada's gross domestic product (GDP) in 2025, following a high of 13.8% in 2020.
Key players are focused on launching new products from anti-histamine drugs in the North America region, which, in turn, is expected to propel market growth over the forecast period. For instance, in March 2020, Nicox, an international ophthalmology company, announced that its U.S. licensee, Eyevance Pharmaceuticals, a Fort Worth-based company, had launched ZERVIATETM in the U.S. ZERVIATE (cetirizine ophthalmic solution) 0.24% is a histamine-1 (H1) receptor antagonist indicated for treatment of ocular itching associated with allergic conjunctivitis.
Increasing initiatives such as awareness campaigns and others by the governments of respective countries for patients suffering from conjunctivitis are expected to boost market growth over the forecast period. For instance, on August 1, 2023, the Health Minister of Hyderabad, India, conducted an awareness campaign on precautions, prevention, and treatment for conjunctivitis and other seasonal diseases. Under the awareness campaign, necessary medicines, including eye drops and ointments, were readily available in all government hospitals in India.
*Definition: Viral conjunctivitis is an inflammation of the conjunctivitis. Viral conjunctiva is caused by infections from viruses such as adenovirus, herpes simplex virus, varicella zoster virus, Picornavirus, poxvirus, and human immunodeficiency virus. It is also called Pink eye disease. Viral conjunctivitis is characterized by a reddish eye, watery discharge from the eye, and itching or irritation. Persistent conjunctivitis from a virus may lead to corneal keratitis. Treatment for viral conjunctivitis may be symptomatic or virus-specific.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients