Global CDMO Injectable Drugs Market Size and Forecast – 2026-2033
The global CDMO injectable drugs market is expected to grow from USD 18,650.1 Mn in 2026 to USD 32,819.9 Mn by 2033, registering a compound annual growth rate (CAGR) of 8.4%. The market is poised for significant expansion driven by the rising use of prescription drugs.
According to the Centers for Disease Control and Prevention, 49.9% of individuals used at least one prescription drug, 24.7% used three or more, and 13.5% used five or more prescription drugs within a 30-day period, reflecting a high and growing demand for pharmaceutical therapies.
(Source: Centers for Disease Control and Prevention)
Key Takeaways of the Global CDMO Injectable Drugs Market
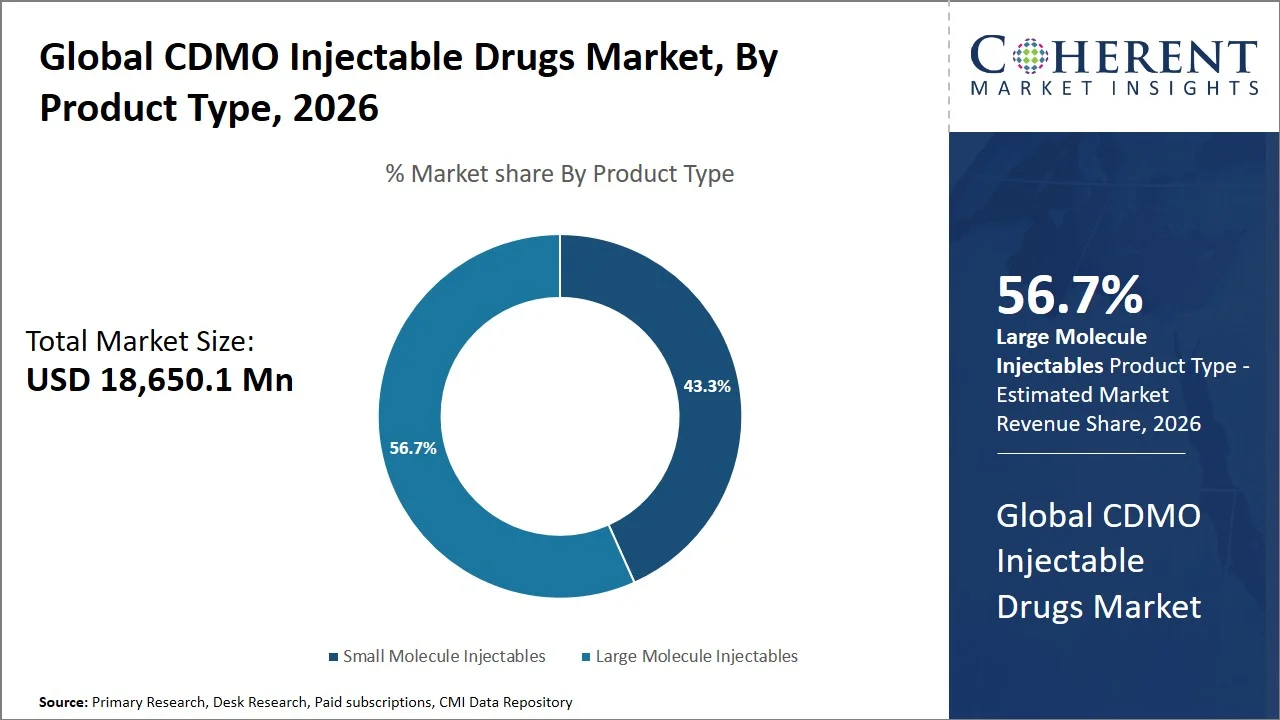
- Large molecule injectables are projected to hold 56.7% of the global CDMO injectable drugs market share in 2026, making it the dominant product type segment, with dominance highly evident in North America because of the high number of approvals for biologics products (e.g., Keytruda, Humira, Opdivo, and Avastin), in addition to biopharmaceutical research and development. The main reasons behind the dominance include the existence of high regulatory standards provided by the U.S. FDA (e.g., Current Good Manufacturing Practice (cGMP) regulations), and the outsourcing trend associated with monoclonal antibody production. For example, some highly effective monoclonal antibodies like Keytruda and Opdivo undergo complex production using very specific methods, which may require the help of contract development and manufacturing organizations (CDMOs).
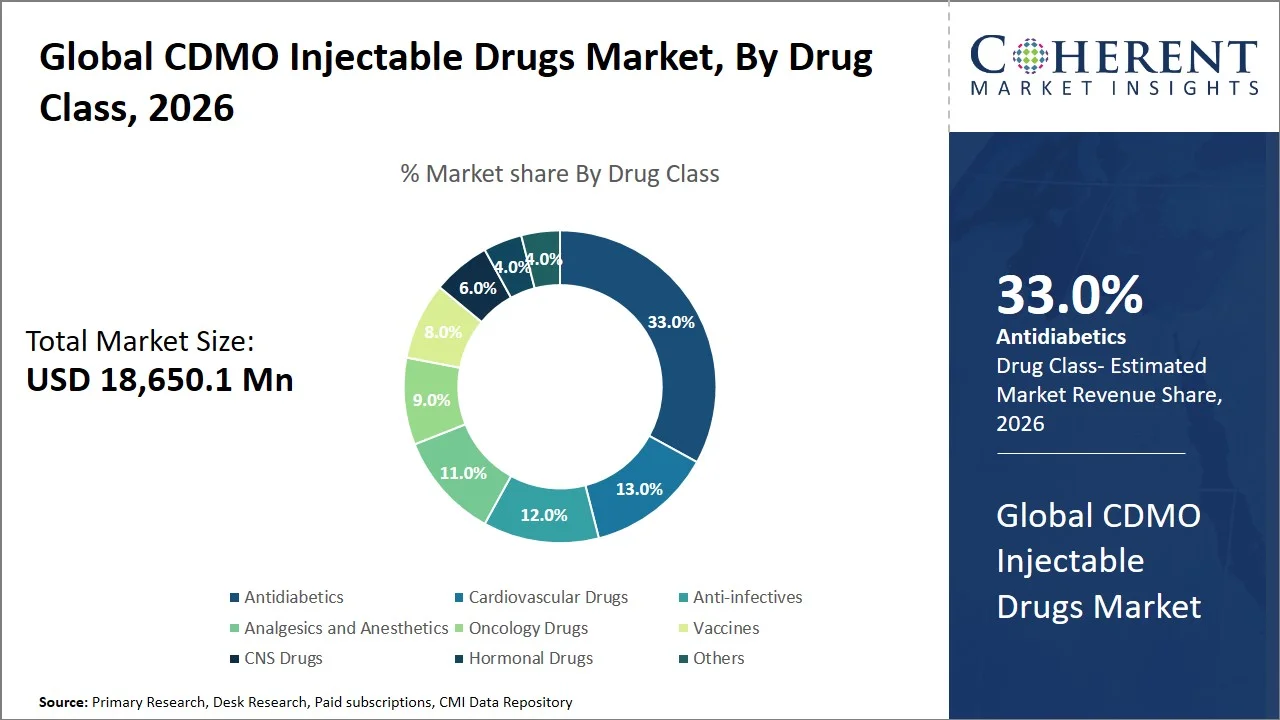
- Antidiabetics are projected to hold 33.0% of the global CDMO injectable drugs market share in 2026, making it the dominant drug class segment, with considerable demand coming from the Asia Pacific region, particularly India and China, because of the increasing prevalence of diabetes and availability of injectables like insulin and GLP-1 receptor agonists. The development of biosimilars and health care growth driven by government initiatives (e.g. Production Linked Incentive (PLI) Scheme and Biopharma Mission in India, and Made in China 2025 and Healthy China 2030 in China) are adding fuel to the CDMO demand in the region.
- Oncology segment is projected to hold 26.9%of the global CDMO injectable drugs market share in 2026, making it the dominant application segment, dominated by North America and Europe, backed by an established pipeline of cancer drugs. The increasing regulatory support and oncology drug approvals by regulatory bodies like the European Medicines Agency (e.g. PRIority MEdicines (PRIME)) have led to a growing demand for sterile and complex injectable manufacturing processes, which has fueled CDMO outsourcing.
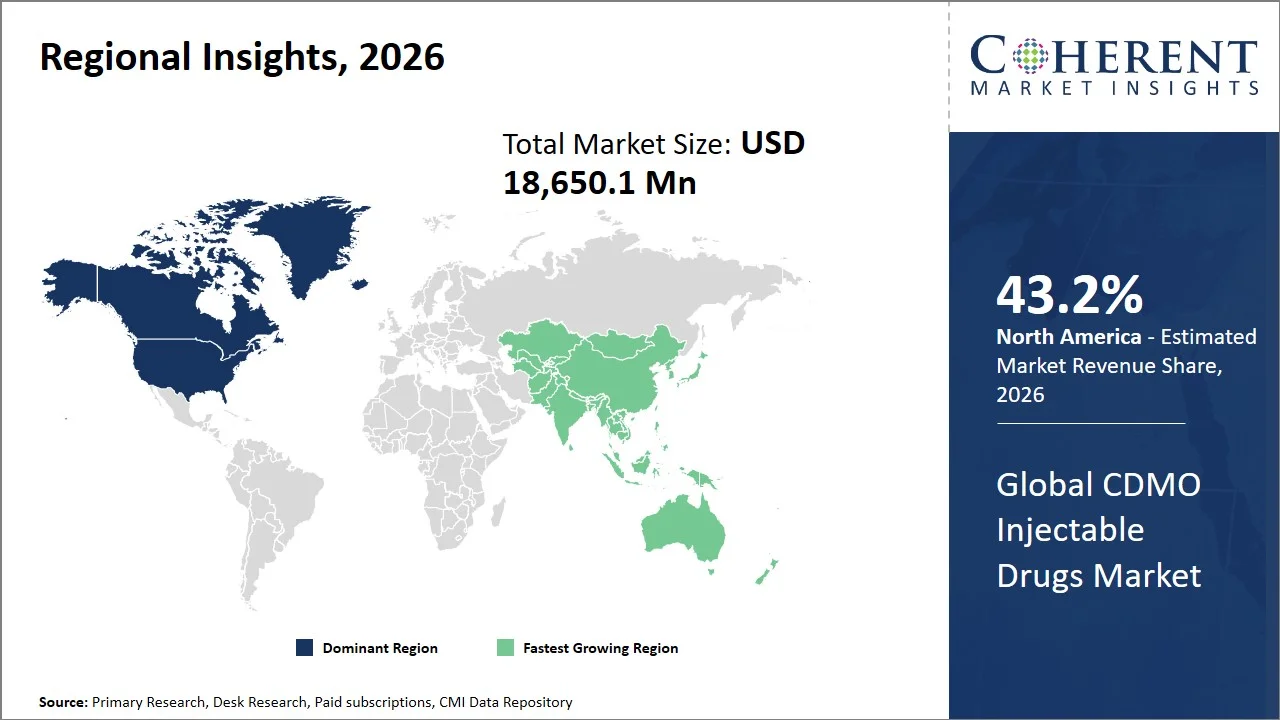
- North America maintains dominance with an estimated share of 43.2%in 2026, driven by the robust pipeline of biological and oncological products, state-of-the-art manufacturing facilities, and high outsourcing. The region is positively impacted by a highly developed regulatory environment, along with major pharmaceutical and biotech firms, ensuring a constant need for CDMO injectables.
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 21.0% share in 2026, facilitated by increased manufacturing capacity, favorable cost dynamics, and growing demand for biological products and biosimilars. This will also be fueled by government policies that support growth, along with greater access to healthcare facilities in developing countries.
- Expansion of High-Potency Injectables: The fast adoption of sophisticated injectable drug products like ADCs, monoclonal antibodies, and GLP-1 agonists has created an urgent need for dedicated manufacturing capacity. This includes the use of highly contained facilities and the need for sterile fill-finish operations, forcing pharmaceutical firms to turn to CDMOs with specific skills. The rising trend in biologics and advanced therapeutics are key driver for CDMO growth worldwide.
- Capacity Constraints in Sterile Fill-Finish & Biologics Manufacturing: Shortages of bioreactor capacities and sterile injection manufacturing slots have forced organizations to outsource to CDMOs. As the trend of outsourcing continues to increase from around 34% in 2014 to about 49% in 2023, there is growing reliance on third-party organizations to manufacture injectables.
Why Do Large Molecule Injectables Dominate the Global CDMO Injectable Drugs Market?
Large molecule injectables are projected to hold the market share of 56.7% in 2026, owing to the increasing trend towards the use of biologics, which essentially need to be processed through sterile injectable and special outsourcing processes. According to the U.S. Food and Drug Administration, over the last few years, there have been a continuous rise in the approval of various biopharmaceuticals, indicating the significant role played by biologics in the drug development process. (Source: U.S. Food and Drug Administration) Concurrently, the World Health Organization has noted the increasing use of biologics in essential treatments. Biologics, such as monoclonal antibodies and recombinant proteins, being highly unstable and not suitable for oral administration, are mainly provided through injections, thus requiring special manufacturing methods (Source: World Health Organization). Consequently, such process complexity, along with high investment costs, compels pharmaceutical manufacturers to resort to CDMOs.
Why are Antidiabetics the Most Preferred Drug Class in Global CDMO Injectable Drugs Market ?
To learn more about this report, Request Free Sample
Antidiabetics are projected to hold a market share of 33.0% in 2026, owing to the increasing prevalence of diabetes, and the lifelong dependence on injectable therapies such as insulin and GLP-1 analogs. As per the World Health Organization, the number of people living with diabetes rose from 200 million in 1990 to 830 million in 2022 (Source: World Health Organization). While International Diabetes Federation estimates the prevalence among adults at 589 million in 2024 and predicts a future estimate of 853 million by 2050 (Source: International Diabetes Federation). Insulin is still necessary for all type 1 diabetics and many with advanced-stage type 2 diabetes; however, only 59% of adult patients received any treatment in 2022, signaling a significant gap in treatment (Source: World Health Organization). The rising numbers of patients, their need for a lifetime treatment option, and the increasing number of treatments available have led to increased demand for the production of high-volume sterile injectable formulations, causing pharmaceutical firms to outsource their manufacturing process to CDMOs.
Oncology Segment Dominates the Global CDMO Injectable Drugs Market
The oncology segment is projected to hold a market share of 26.9% in 2026, owing to the large portfolio of cancer treatments, which include highly complex biologics needing a sterile injectable form and special manufacturing. As per the U.S. Food and Drug Administration, there has been a considerable portion of new approvals from the oncology sub-segment over the years, along with biological drugs including monoclonal antibodies and targeted cancer medicines. Moreover, the National Cancer Institute highlights that today's treatment of cancer depends significantly on biologic drugs and immunotherapies, which can be administered only through injections (Source: National Cancer Institute). This factor, together with a growing trend toward outsourcing drug manufacturing among pharmaceutical companies, further cements the dominant position of the oncology sub-segment in the CDMO injectable drugs market.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
U.S. FDA Initiative to Strengthen Domestic Drug Manufacturing (2026) |
|
|
Tightening Global Regulatory Standards for Sterile Injectable Manufacturing (2025–2026) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: U.S. Food and Drug Administration, U.S. Food and Drug Administration)
Global CDMO Injectable Drugs Market Dynamics
To learn more about this report, Request Free Sample
Market Drivers
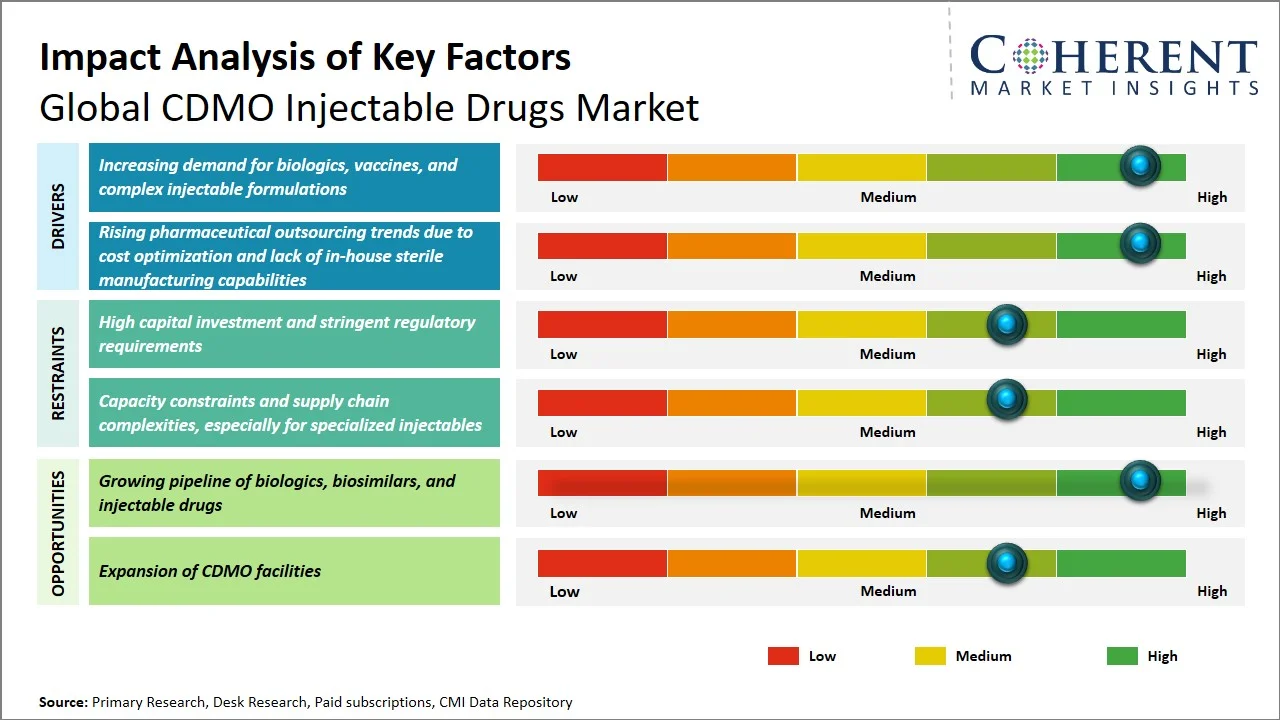
- Increasing demand for biologics, vaccines, and complex injectable formulations: Biologics constitute ~31-32% of all new drugs being approved in 2023–2024, according to statistics matching the criteria laid out by the U.S. Food and Drug Administration (Source: DCAT). Furthermore, as per the World Health Organization, billions of vaccines are being injected each year in the immunization campaigns worldwide, for instance, about 109 million babies were inoculated with three DTP vaccinations in 2024 alone, with about 115 million having received at least one DTP vaccination, illustrating the enormous magnitude of vaccines administered globally (Source: World Health Organization). The rising demand for biologics that often come as injectables and constant requirement for vaccine injections are driving CDMOs towards specialized needs of sterility.
- Rising pharmaceutical outsourcing trends due to cost optimization and lack of in-house sterile manufacturing capabilities: The rising trend of outsourcing of medications due to the benefits of cost reduction and lack of sterile production capacity in-house forms one of the major drivers behind the increasing demand for the CDMO injectable drugs market. The sterile production of any drug according to the U.S. FDA requires the compliance with cGMP standards that involve having clean room facilities and aseptic production processes along with regular quality checks. Sterile production requires heavy investment on the part of the company. Therefore, pharmaceutical companies find it convenient to outsource their production activities to CDMOs. (Source: U.S. Food & Drug Administration)
Emerging Trends
- Expansion of High-Potency API (HPAPI) Manufacturing Capabilities: Contract development and manufacturing organizations (CDMOs) are increasingly investing in containment facilities and specialized infrastructure to handle highly potent compounds, particularly in oncology. This is driven by the growing number of targeted therapies in development, which require strict safety and handling standards.
- Rising Demand for Prefilled Syringes and Advanced Delivery Systems: Use of prefilled syringes and long-acting injectables is gaining momentum due to their user-friendliness, accuracy in dosing, and better patient compliance. CDMOs are upgrading their fill-finish facilities to suit these changing delivery modalities.
Regional Insights
To learn more about this report, Request Free Sample
Why is North America a Strong Market for CDMO Injectable Drugs?
North America leads the global CDMO injectable drugs market, accounting for an estimated 43.2% share in 2026, due to the advanced nature of the regulatory environment, robust biologics pipeline, and higher number of oncology and specialty drug approvals, most of which necessitate sterile manufacturing processes.
According to the U.S. Food and Drug Administration, the U.S. remains the world leader in drug innovation, with as many as 50 novel drugs having been approved in the year 2024, with some of them containing complex biologics (Source: U.S. Food and Drug Administration). In addition to that, the FDA's Center of Excellence for Oncology reported the approval of 89 oncology drugs and biologics in 2024 (Source: U.S. Food and Drug Administration). The strong presence of regulations, the approval of biosimilars and oncology products, along with existing pharmaceuticals and biotech infrastructure, creates high demand for outsourcing services by CDMOs, making North America the dominant region for the CDMO injectable drugs market.
Why Does Asia Pacific CDMO Injectable Drugs Market Exhibit High Growth?
Why is the U.S. Leading Innovation and Adoption in the CDMO Injectable Drugs Market?
The U.S. is the most innovative and prominent country for the CDMO injectable drugs market because of its robust regulatory environment and developing biopharmaceuticals industry. The U.S. FDA emphasizes the growing amount of biosimilars approvals in the U.S. which implies an increase in the number of complicated biologic products that necessitate advanced manufacturing processes (Source: U.S. Food and Drug Administration).
Besides, the publication of novel drug approvals by the U.S. FDA indicates the active work of the drug developers’ pipeline including such areas as oncology and specialty medicines. As many medications belong to the category of injections and require a sterile manufacturing process, pharmaceutical enterprises become increasingly reliant on CDMOs.
Is U.K. a Favorable Market for CDMO Injectable Drugs Market?
The U.K. can be regarded as an ideal market for the CDMO injectable drugs market owing to favorable regulations as well as the country’s preference for biosimilars. The Medicines and Healthcare products Regulatory Agency (MHRA) has developed easier pathways for the use of biosimilars through reduced development processes. (Source: Medicines and Healthcare products Regulatory Agency)
Additionally, the National Health Service encourages the use of biosimilar drugs in major therapeutic classes such as cancer and chronic diseases. According to the NHS, biological drugs have played an important role in treating various ailments, including cancer and auto-immune diseases. In this regard, the NHS has initiated a national approach to speed up the use of biosimilars in clinical practice (Source: NHS). This increasing adoption, given that most biosimilar and biological drugs demand high sterility and advanced manufacturing techniques, only strengthens the dependency on CDMOs in the production of specialized injectable medicines.
Is China Emerging as a Key Growth Hub for the CDMO Injectable Drugs Market?
China appears to be a major growth hub for the CDMO injectable drugs market owing to robust support in manufacturing from the government, growing demands in health care, and advanced biopharmaceutical industry. For example, in recent years, China’s pharmaceutical industry has experienced rapid growth, with the market size expanding from USD 180 billion (1.21 trillion RMB) in 2010 to USD 420 billion (2.97 trillion RMB) in 2024. The industry now accounts for 29.5% of the global research and development (R&D) pipeline, positioning China as the world’s second-largest drug market and a leading hub for drug development and innovation (Source: National Library of Medicine).
Moreover, China encourages innovation in biopharmaceuticals and high-end drug production by implementing industry policies (Made in China 2025, 14th Five-Year Plan for Pharmaceutical Industry Development, and Healthy China 2030 Initiative), which are helping in boosting the production of complex injectables. The combination of massive manufacturing capacity, increased spending on health care, and drug development is making China a potential outsourcing destination for injectable drugs.
Why Does Germany Top the European CDMO Injectable Drugs Market?
Germany leads the market for CDMO injectable drugs in Europe fueled by its well-developed pharmaceutical manufacturing industry, its leadership in biopharmaceuticals production, and strong export capacity. Germany Trade & Invest estimates that biopharmaceuticals made up about one-third of the entire pharmaceutical market in 2023, owing to the nation’s high emphasis on biologics which need injection and are difficult to manufacture (Source: Germany Trade & Invest). In addition to being among the major exporters of pharmaceutical goods internationally, this advanced network, in tandem with high biologicals penetration levels, fuels substantial demand for CDMO operations, thus making Germany the dominant market for outsourcing injectable drugs in Europe.
Is CDMO Injectable Drugs Market Developing in India?
The CDMO injectable drugs market in India is witnessing considerable growth due to substantial government backing and development in biologic infrastructure. The Indian government has implemented various schemes like the Biopharma SHAKTI program worth USD 1.07 billion (₹10,000 crore) to improve its biologics and biosimilars production capacity (Source: Government of India). India’s vast pharmaceutical production capabilities and cost efficiencies make it suitable for outsourcing. With the expansion of biologics and biosimilars, there is a rise in demand for sterilized and sophisticated production processes, placing India at the forefront of CDMO injectable drugs market.
Regulatory Landscape Governing the Global CDMO Injectable Drugs Market
|
Region |
Key Regulations |
Regulatory Overview |
Impact on Market |
|
North America |
U.S. Food and Drug Administration (FDA) – cGMP (21 CFR Parts 210/211), NDA/ANDA/BLA pathways, Drug Supply Chain Security Act (DSCSA) |
Highly stringent regulatory framework governing sterile manufacturing, quality control, and supply chain traceability; strong oversight on biologics and injectables |
Ensures high product quality and safety, boosting client trust; however, increases compliance costs and time-to-market for CDMOs |
|
Europe |
European Medicines Agency (EMA), EU GMP Guidelines (EudraLex Volume 4), Falsified Medicines Directive (FMD), GDPR |
Harmonized regulatory system across EU with strict requirements for sterile manufacturing, pharmacovigilance, and serialization |
Facilitates cross-border operations and approvals; however, multi-country compliance and documentation add operational complexity |
|
Asia Pacific |
NMPA (China), CDSCO (India), PMDA (Japan), TGA (Australia) |
Diverse and evolving regulatory landscape with increasing alignment to global GMP standards and focus on biologics and generics manufacturing |
Offers cost advantages and faster approvals in some regions; regulatory variability and frequent updates can pose entry challenges |
|
Latin America |
ANVISA (Brazil), COFEPRIS (Mexico), INVIMA (Colombia) |
Developing regulatory frameworks with improving GMP standards and increasing alignment with international guidelines |
Supports growing outsourcing opportunities; however, longer approval timelines and inconsistent regulations may delay market entry |
|
Middle East & Africa |
Saudi FDA (SFDA), South African Health Products Regulatory Authority (SAHPRA), UAE MOHAP |
Emerging regulatory systems with rising emphasis on quality standards, import controls, and localization policies |
Creates new growth avenues for CDMOs; however, fragmented regulations and evolving policies may increase operational complexity |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is the growing pipeline of biologics, biosimilars, and injectable drugs creating new growth opportunities in the CDMO injectable drugs market?
The rising pipeline of biologics, biosimilars, and injectables has provided immense growth prospects for the CDMO injectable drugs market through higher demands for development and manufacturing services. As per the U.S. Food and Drug Administration (FDA), biological products constitute an important segment in the approval of drugs in which several products involve sophisticated processes and Biologics License Application (BLA) pathway for delivery through injections. (Source: U.S. Food and Drug Administration)
Additionally, the European Medicines Agency stated that there has been a steady rise in approved biosimilars in Europe, with over 86 biosimilar medicines approved in the EU since 2006, demonstrating sustained growth in biologic alternatives. (Source: European Medicines Agency) The rising pipeline, along with the intricacy involved in manufacturing such biologics, is motivating pharmaceutical organizations to collaborate with CDMOs in relation to aseptic filling and manufacturing activities.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- On April 1, 2026, ROiS CDMO finalized the purchase of an injectable manufacturing facility located in the U.S. This acquisition bolstered its sterile manufacturing capabilities and broadened its presence in North America. The decision underscores the rising investments by CDMOs, aimed at increasing injectable production capacity to satisfy the escalating demand for outsourcing.
- In October 2025, Terumo Corporation finalized the purchase of a drug product manufacturing facility located in Leverkusen, Germany, from WuXi Biologics. This acquisition bolsters Terumo Corporation's capabilities as a contract development and manufacturing organization (CDMO) for injectables. The deal also expands Terumo Corporation's global production capacity and its ability to respond to market demands, a clear indication of the company's growing investments in sterile injectable manufacturing infrastructure.
Competitive Landscape
The CDMO injectable drugs market is characterized by intense competition fueled by a multitude of international and domestic entities providing specialized manufacturing solutions. The competitive landscape is largely determined by technical proficiency, adherence to regulatory standards, and cost-effectiveness, reflecting the intricate and sterile requirements inherent in injectable drug production. To distinguish themselves from competitors, companies are concentrating more on enhancing their capabilities, utilizing sophisticated aseptic techniques, and providing complete service packages. Key focus areas include:
- Capacity expansion and sterile manufacturing capabilities
- Cost leadership and operational efficiency
- End-to-end service offerings
- Regulatory compliance and quality standards
- Focus on complex injectables and biologics
Market Report Scope
CDMO Injectable Drugs Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 18,650.1 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.4% | 2033 Value Projection: | USD 32,819.9 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Lonza Group, Thermo Fisher Scientific, Catalent, Inc., Samsung Biologics, WuXi Biologics, FUJIFILM Diosynth Biotechnologies, Boehringer Ingelheim, Recipharm AB, Siegfried Holding AG, and AGC Biologics |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- The future growth trajectory of the CDMO injectable drugs market would be heavily influenced by the fast-paced adoption of biological drugs, biosimilar products, and more complex sterile pharmaceuticals. In view of pharmaceutical firms adopting the light assets model, the outsourcing of fill-finish, aseptic processes, and high-potency injectables will be the norm, thus making CDMOs business partners of choice.
- The largest market opportunities will arise from the production of biologics and biosimilars, particularly those pertaining to oncology and immunology, where the U.S. and China will be the focal points for expansion. For the U.S., there is an abundance of market opportunity due to innovation and high consumption of biologics. On the other hand, the scope of growth in China is enormous owing to the creation of many pipelines for biologics locally, along with the growing tendency for outsourcing.
- For any competitive advantage, the players need to concentrate on their capacity growth in sterile manufacturing, their investments in modern fill-and-finish equipment, and their regulatory excellence. It will be necessary for companies to have end-to-end capabilities from the developmental stage to large-scale production processes. Partnerships with biotechnology companies, automation in manufacturing, and supply chain optimization will help create more differentiation for top CDMOs.
Market Segmentation
- Product Type Insights (Revenue, USD Mn, 2021 - 2033)
- Small Molecule Injectables
- Large Molecule Injectables
- Drug Class Insights (Revenue, USD Mn, 2021 - 2033)
- Antidiabetics
- Cardiovascular Drugs
- Anti-infectives
- Analgesics and Anesthetics
- Oncology Drugs
- Vaccines
- CNS Drugs
- Hormonal Drugs
- Others
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Oncology
- Diabetes
- Autoimmune Diseases
- Hematological Disorders
- Cardiovascular Diseases
- Infectious Diseases
- Neurological Disorders
- Pain Management
- Rare Diseases
- Others
- Route of Administration Insights (Revenue, USD Mn, 2021 - 2033)
- Intramuscular (IM)
- Intravenous (IV)
- Subcutaneous (SC)
- Others
- Scale of Operations Insights (Revenue, USD Mn, 2021 - 2033)
- Preclinical Development
- Clinical Development (Phases I–III)
- Commercial / Large-Scale Manufacturing
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Biopharmaceutical Companies
- Generic Drug Manufacturers
- Contract Research Organizations (CROs)
- Academic and Research Institutes
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Lonza Group
- Thermo Fisher Scientific
- Catalent, Inc.
- Samsung Biologics
- WuXi Biologics
- FUJIFILM Diosynth Biotechnologies
- Boehringer Ingelheim
- Recipharm AB
- Siegfried Holding AG
- AGC Biologics
Sources
Primary Research Interviews
- CDMO injectable drug manufacturers and contract service providers
- Pharmaceutical companies outsourcing sterile manufacturing and fill-finish services
- Hospital procurement managers and supply chain distributors for injectable drugs
- Clinical researchers and regulatory experts specializing in biologics and injectable formulations
Stakeholders
- Providers of CDMO Injectable Drug Solutions
- End-use Sectors
- Hospitals
- Specialty Clinics (Oncology, Immunology, Infectious Diseases)
- Retail Pharmacies
- Online Pharmacies
- Regulatory & Health Bodies: U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), Central Drugs Standard Control Organization (CDSCO), World Health Organization (WHO)
Databases
- U.S. Food and Drug Administration (FDA) – Drug approvals & Orange Book database
- World Health Organization (WHO) – Global Health Observatory (GHO)
- OECD Health Statistics – Pharmaceutical manufacturing and consumption data
- National Institutes of Health (NIH) – Clinical trials and biologics research database
Magazines
- Pharmaceutical Technology – Injectable drug manufacturing and CDMO trends
- BioPharma Dive – Biologics and contract manufacturing developments
- Drug Discovery & Development – Advances in sterile manufacturing and injectables
- Healthcare Business Today – Pharma outsourcing and CDMO market trends
Journals
- Journal of Pharmaceutical Sciences – Injectable formulation and manufacturing studies
- International Journal of Pharmaceutics – Sterile drug delivery systems research
- PDA Journal of Pharmaceutical Science and Technology – Aseptic processing and GMP compliance
- BMJ (British Medical Journal) – Clinical use of injectable therapies
Newspapers
- The Wall Street Journal – Pharmaceutical outsourcing and CDMO developments
- Financial Times – Global pharma manufacturing and biologics market trends
- Business Standard – India pharmaceutical manufacturing and CDMO insights
- The Economic Times – Pharma sector and contract manufacturing updates
Associations
- Parenteral Drug Association (PDA)
- International Society for Pharmaceutical Engineering (ISPE)
- Biotechnology Innovation Organization (BIO)
- International Federation of Pharmaceutical Manufacturers & Associations (IFPMA)
Public Domain Sources
- World Health Organization (WHO) – Injectable medicines and biologics guidelines
- U.S. National Institutes of Health (NIH) – Biologics and injectable drug research
- U.S. FDA – Sterile manufacturing and injectable drug approvals
- European Medicines Agency (EMA) – Biologics and injectable drug regulations
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients