Global Difluprednate Market Size and Forecast – 2026-2033
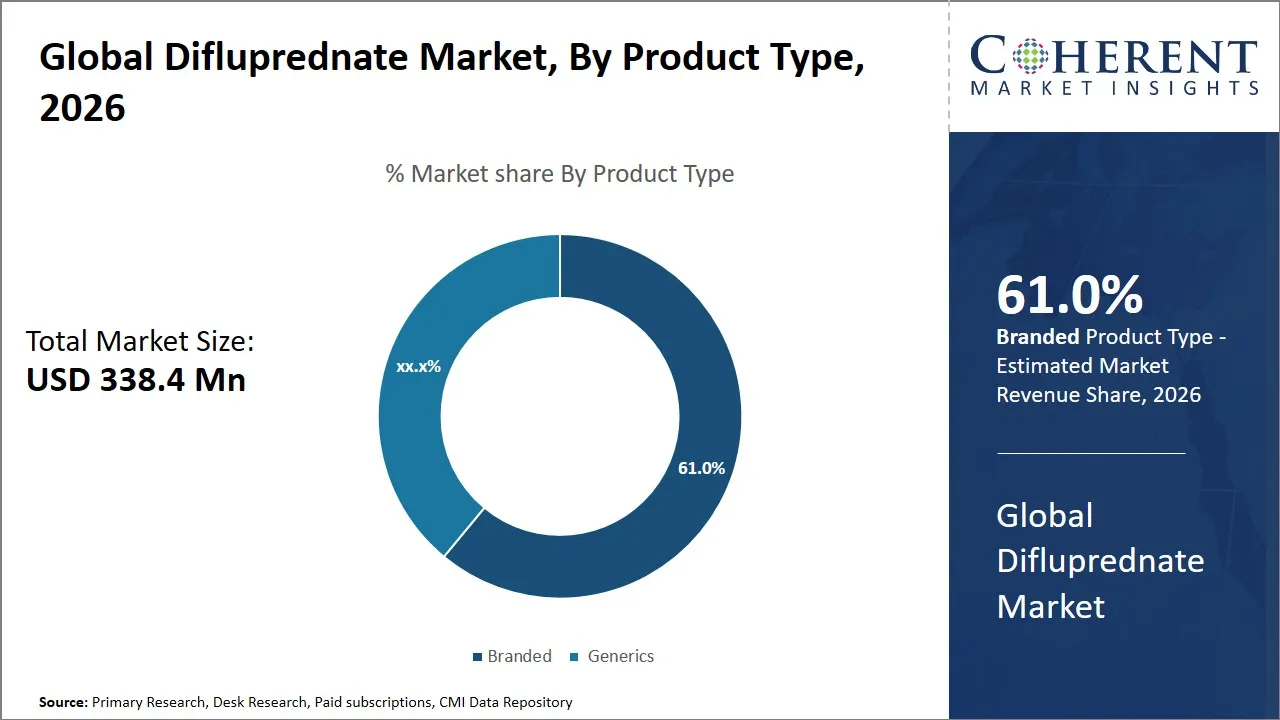
The global difluprednate market is estimated to be valued at USD 338.4 Mn in 2026 and is expected to reach USD 467.5 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 4.7% from 2026 to 2033. This steady growth is due to the increasing demand for effective anti-inflammatory ophthalmic drugs. In addition, the expanding applications of the drugs are propelling the market's growth. Furthermore, the increase in the prevalence of eye disorders, coupled with the advancements in drug delivery systems, is also contributing to the growth of the market.
Key Takeaways of the Global Difluprednate Market
- The branded segment is expected to dominate with 61.0% of the global difluprednate market share in 2026.
- The post-operative ocular inflammation segment is expected to dominate the difluprednate market, accounting for 47.0% share in 2026.
- The hospital pharmacies segment is projected to capture 52.0% of the global difluprednate market share in 2026.
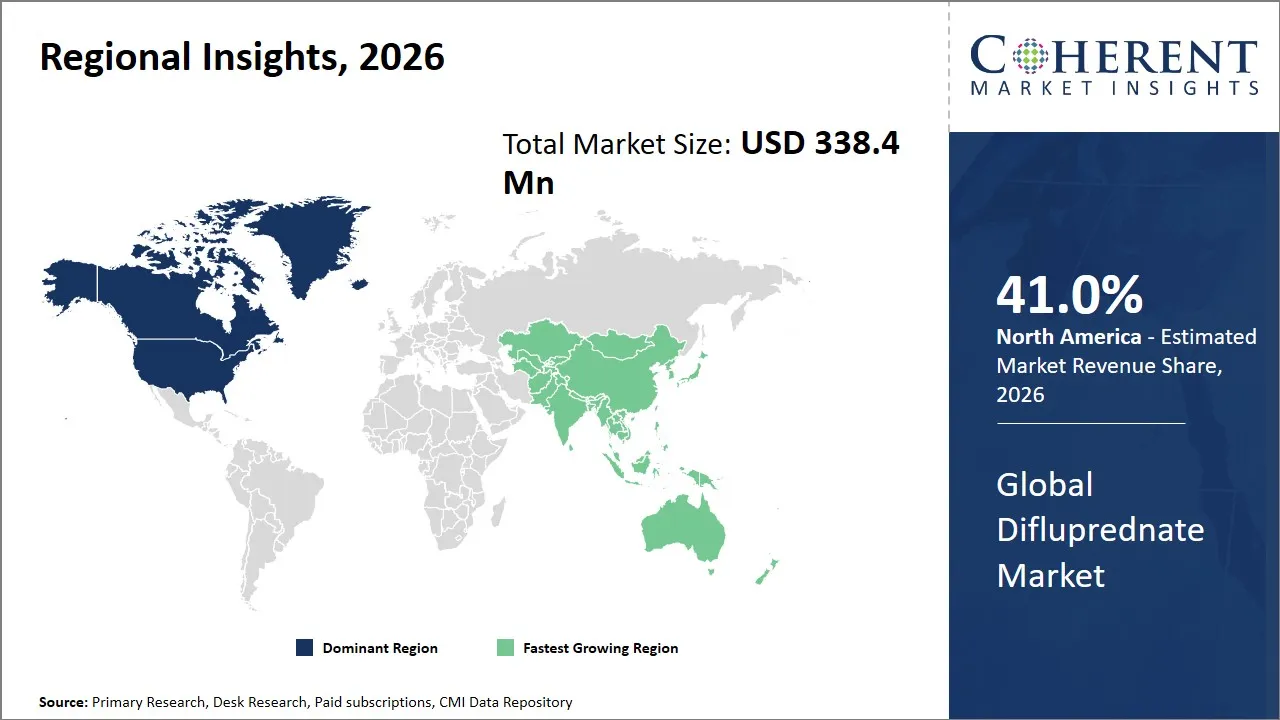
- North America is expected to dominate the global difluprednate market in 2026, accounting for 41.0% share.
- Asia Pacific is anticipated to witness the fastest growth, holding a market share of 26.0% in 2026.
Market Overview
- Difluprednate is a topical ophthalmic corticosteroid used for the management of post-operative ocular inflammation, pain, and anterior uveitis. Difluprednate functions by decreasing eye inflammation. This makes it a significant medication for post-surgical care and management of eye inflammation.
- The difluprednate market is expanding, and its growth is expected to continue due to rising eye surgeries, including cataract and refractive surgeries. The prevalence of eye inflammatory disorders is also on the rise. Difluprednate is found to be very effective, and its anti-inflammatory properties and bioavailability compared to conventional corticosteroids make it a preferred choice for eye disorders.
- In addition, there is a significant expansion of generic forms of difluprednate, and developments in ophthalmic drug delivery systems, including eye emulsions, have improved patient compliance. The difluprednate market is expected to continue its growth due to advancements and developments, as well as rising healthcare infrastructure and awareness of eye health.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Increasing Volume of Ophthalmic Surgeries |
|
|
Expansion of Generic Difluprednate Approvals |
|
|
Rising Prevalence of Ocular Inflammatory Disorders |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Branded Segment Dominate the Global Difluprednate Market in 2026?
The branded segment is anticipated to hold a market share of 61% in 2026, due to high levels of trust among physicians, efficacy, and validation. These products have the advantage of extensive clinical trials and quality formulation, making them trustworthy for managing sensitive ocular conditions. They are preferred by physicians, especially in complicated cases such as post-operative inflammation and uveitis, where the efficacy and safety of a drug are of prime importance. In addition, these branded products have high levels of engagement among physicians and patients, making them easily accessible in all segments of the market.
Branded difluprednate is constantly developing and improving drug delivery mechanisms and efficacy, such as ocular penetration and dosage. They have high levels of marketing and presence in the market, making them easily accessible and visible in the market, despite the presence of generic products.
Why Does the Post-Operative Ocular Inflammation Segment Dominate the Global Difluprednate Market in 2026?
The post-operative ocular inflammation segment is expected to hold the highest market share of 47% in 2026, due to the urgent requirement for the management of inflammation during eye surgeries like the removal of cataracts, corneal transplantations, glaucoma surgery, etc. Inflammation is a common complication during eye surgeries, and the failure of the drug to effectively control the same may have an adverse impact on the outcome of the surgery. Therefore, the drug is extensively used because of its efficacy in the management of inflammation. The rising number of eye surgeries across the globe, especially cataract surgeries, is an added advantage for the drug.
Moreover, the drug is also preferred by eye care practitioners because of its strong efficacy in the management of ocular inflammation. In addition to this, the drug is preferred because of the short duration of action of the drug. The rising acceptance of advanced surgical techniques is an added advantage for the drug. Therefore, the aforementioned factors have made post-operative ocular inflammation the largest segment of the difluprednate market.
For instance, in December 2024, the DailyMed database of the U.S. National Library of Medicine reaffirmed the specific indication of difluprednate ophthalmic emulsion for the treatment of inflammation and pain following ocular surgery, such as cataract surgery, thereby directly supporting the dominance of this segment.
(Source: National Library of Medicine)
The Hospital Pharmacies Segment Dominates the Global Difluprednate Market
Hospital pharmacies segment is expected to dominate with 52% of the market share in 2026. The hospital pharmacies are an integral part of eye care treatment and post-operative care. In most cases, eye surgeries are done in hospitals, along with the treatment of various eye-related complications. Therefore, hospital pharmacies are considered to be a major source of difluprednate because they are easily accessible during critical stages of treatment.
They have a direct association with pharmaceutical companies that ensure the availability of brand-name drug products. This is important in eye care treatment after eye surgeries. In addition to that, hospital pharmacies are well equipped with ophthalmology services that provide critical care information pertaining to the use of corticosteroids to ensure a safe outcome during treatment. With the increase in demand for institutionalized treatment and development in the field of medicine, hospital pharmacies are considered to be a trustworthy source of difluprednate in comparison to other sources like retail pharmacies and online pharmacies.
Regulatory Landscape & Clinical Practice Guidelines Influencing Difluprednate Adoption
|
Region |
Approval Status of Difluprednate |
Clinical Practice Guidelines |
Key Impact on Market |
|
North America |
Fully approved for post-operative inflammation & pain |
Strong recommendation for corticosteroids posts cataract surgery |
High adoption due to clear guidelines and strong reimbursement |
|
Europe |
Approved in major countries (Germany, France, U.K., Spain, and Italy) |
Widely included in post-operative inflammation management protocols |
Moderate to high adoption; varies by country reimbursement |
|
Asia Pacific |
Approved in selected countries (Japan, India) |
Increasing adoption aligned with rising surgical volumes |
Growing demand due to expanding healthcare infrastructure |
|
Latin America |
Limited but expanding approvals |
Adoption influenced by physician preference and availability |
Emerging market with gradual uptake |
|
Middle East & Africa |
Limited approvals |
Less standardized guidelines; dependent on imports |
Lower penetration but improving access |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Regional Insights
To learn more about this report, Request Free Sample
North America Difluprednate Market Analysis and Trends
The North America region is projected to lead the market with a 41% share in 2026, attributed to the presence of a strong medical infrastructure and awareness of advanced eye care treatments. The region is characterized by the presence of strong regulations from the FDA, thereby maintaining the quality of the products. In addition, the presence of strong market players such as Alcon (Novartis), Bausch + Lomb, and Valeant Pharmaceuticals (Bausch Health) ensures the innovation of products and marketing strategies.
Additionally, the efforts of the government in controlling eye care disorders and investment in eye care R&D activities have further added to the dominance of the market in the region of North America. The presence of strong and well-established distribution channels ensures the sustainability of the market.
For instance, in March 2024, the U.S. FDA completed its review and recommended approval of Clobetasol Propionate Ophthalmic Suspension 0.05% for post-operative inflammation and pain associated with ocular surgery based on positive Phase 3 trials for efficacy and safety.
(Source: U.S. Food & Drug Administration)
Asia Pacific Difluprednate Market Analysis and Trends
Asia Pacific is anticipated to witness the fastest growth, holding a market share of 26.0% in 2026, owing to factors such as better access to healthcare services, high incidence of ocular diseases, and better awareness among patients in emerging markets such as India, China, Japan, and South Korea. Development of pharmaceutical manufacturing capabilities and government initiatives in supporting healthcare infrastructure development also contribute significantly to the growth of the difluprednate market in Asia pacific. Pharmaceutical companies such as Sun Pharmaceutical Industries and Lupin Limited are currently engaged in expanding their product portfolios in the Asia Pacific region and forming partnerships with multinational corporations for improving drug accessibility in Asia Pacific.
Additionally, factors such as better urbanization, high incidence of ocular diseases among elderly patient populations, and better affordability of Difluprednate formulations owing to development in generic drug formulations also contribute significantly to the dynamic nature of the Asia Pacific difluprednate market.
Difluprednate Market Outlook for Key Countries
How is the U.S. Helping in the Growth of the Difluprednate Market?
There is a huge demand for advanced anti-inflammatory ophthalmic preparations in the U.S. due to the rising number of eye surgeries such as cataract and retina repair, where difluprednate is used. Companies such as Alcon and Bausch + Lomb have a large share of the market due to their continuous investment in advanced drug delivery systems and eye drops. The reimbursement policies and patent position of the U.S. make it a lucrative market for investment in launching drugs and conducting clinical studies on difluprednate.
How is India Helping in the Growth of the Difluprednate Market?
The difluprednate market in India is growing significantly owing to the increasing penetration of healthcare in the country, both in rural and urban areas. At the same time, the availability of generic forms of difluprednate is making it possible for a large patient pool to access the drug. Companies such as Sun Pharmaceutical and Cipla are playing a crucial role in the production and distribution of difluprednate. These local players are sometimes partnering with international pharmaceutical brands to increase production and availability. Initiatives taken by the government in promoting eye health through vision care programs are also helping in increasing the market for difluprednate.
For instance, in August 2023, according to a Press Information Bureau release issued under the Ministry of Health & Family Welfare, a total of 83,44,824 cataract surgeries were conducted in FY 2022-23 under the National Programme for Control of Blindness and Visual Impairment (NPCBVI). This reflects a large demand for post-operative eye care products like difluprednate in India.
(Source: Ministry of Health and Family Welfare)
Key Drivers for the Growth of the China Difluprednate Market
China remains at the top of regional demand driven by its vast population and increasing rates of eye diseases caused by pollution and aging populations. Chinese regulatory authorities are becoming increasingly compliant with international regulations, thus speeding up the approval and import of innovative formulations of difluprednate. Multinational firms such as Pfizer and Novartis have been actively collaborating with local firms in manufacturing and marketing, while local firms are going into R&D to address the need for customized treatment.
Germany Difluprednate Market Trends
The difluprednate market of Germany is dominated by a highly advanced pharmaceutical research environment and a highly regulated healthcare system that is focused on providing highly innovative and efficient solutions for eye inflammation. Major pharmaceutical companies of Europe, like Bayer AG and STADA Arzneimittel, have made significant contributions to providing highly advanced formulations of difluprednate with highly efficient efficacy and safety profiles. The fact that Germany is a part of the EU framework provides a healthy competitive environment for the drug difluprednate.
Ocular Disease Epidemiology Influencing Difluprednate Demand
|
Disease Condition |
Global/Regional Prevalence Trend |
Key Patient Demographics |
Relevance to Difluprednate Use |
Market Impact |
|
Cataract |
Leading cause of blindness globally; rising surgical volume annually |
Aging population (60+ years) |
Primary use in post-operative inflammation management |
Major revenue contributor due to high procedure volume |
|
Post-operative Inflammation |
Common after ocular surgeries (cataract, LASIK, etc.) |
All surgical patients |
Direct indication; standard corticosteroid therapy |
High demand driven by increasing surgical interventions |
|
Uveitis |
Moderate prevalence; chronic and recurrent cases |
Adults (20–60 years), autoimmune patients |
Used to control inflammation and prevent complications |
Steady demand due to long-term management needs |
|
Diabetic Macular Edema (DME) |
Increasing with global diabetes prevalence |
Diabetic population |
Adjunct anti-inflammatory role in some treatment protocols |
Emerging opportunity with rising diabetes cases |
|
Allergic Conjunctivitis |
Highly prevalent, especially in urban populations |
Children and young adults |
Limited use; mild steroids used in severe cases |
Low to moderate contribution |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Players, Key Developments, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In January 2026, Alembic Pharmaceuticals has obtained approval from the U.S. FDA for difluprednate ophthalmic emulsion 0.05% (generic) for the treatment of post-operative inflammation and anterior uveitis. This ensured the affordability and availability of the drug in the market.
- In September 2025, Upsher-Smith Laboratories launched difluprednate ophthalmic emulsion 0.05% in the U.S. generics industry. This further intensified the competition in the U.S. generics industry and ensured the implementation of lifecycle management for pharmaceutical companies.
- In December 2024, Caplin Steriles obtained approval from the U.S. FDA for the distribution of difluprednate ophthalmic emulsion 0.05%. This approval ensured the entry of Caplin in the ophthalmic corticosteroids drug segment.
Top Strategies Followed by Global Difluprednate Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established market players prioritize research and development spending, focus on innovative formulations, and pursue global expansion. These companies improve drug efficacy, enhance delivery systems, and expand geographic reach through strong distribution networks and partnerships |
In March 2024, Novartis AG strengthened its presence in the ophthalmology segment of the business through its Alcon division by expanding its surgical and eye care products. This indirectly relates to the adjutant therapy segment of the business, which includes the use of corticosteroid drugs. |
|
Mid-Level Players |
Mid-level players emphasize on cost-effective production, generic launches, and collaborations for market presence in highly competitive markets. These companies aim to achieve cost and quality balance and use collaborations for quicker market access. |
In August 2024, Cipla Limited strengthened its presence in the generic segment of the business by filing various products for approval in the U.S. markets. This once again highlights the company's focus on strengthening its presence in the ophthalmology segment of the business. |
|
Small-Scale Players |
Small-scale players emphasize on niche differentiation and regional expansion, including specialized formulations (preservative-free) and collaborations with regional distributors for market access and visibility. |
In June 2024, LGM Pharma strengthened its presence in the contract development and manufacturing segment of the business by expanding its services to the pharmaceutical industry for the development of complex pharmaceutical formulations. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
Difluprednate Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 338.4 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.7% | 2033 Value Projection: | USD 467.5 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Novartis AG, Amneal Pharmaceuticals LLC, Cipla, Dr. Reddy’s Laboratories Ltd, Alembic Pharmaceuticals Limited, Upsher Smith Laboratories, Axplora, Symbiotec Pharmalab, LGM Pharma, and Viatris Inc |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Difluprednate Market Dynamics
To learn more about this report, Request Free Sample
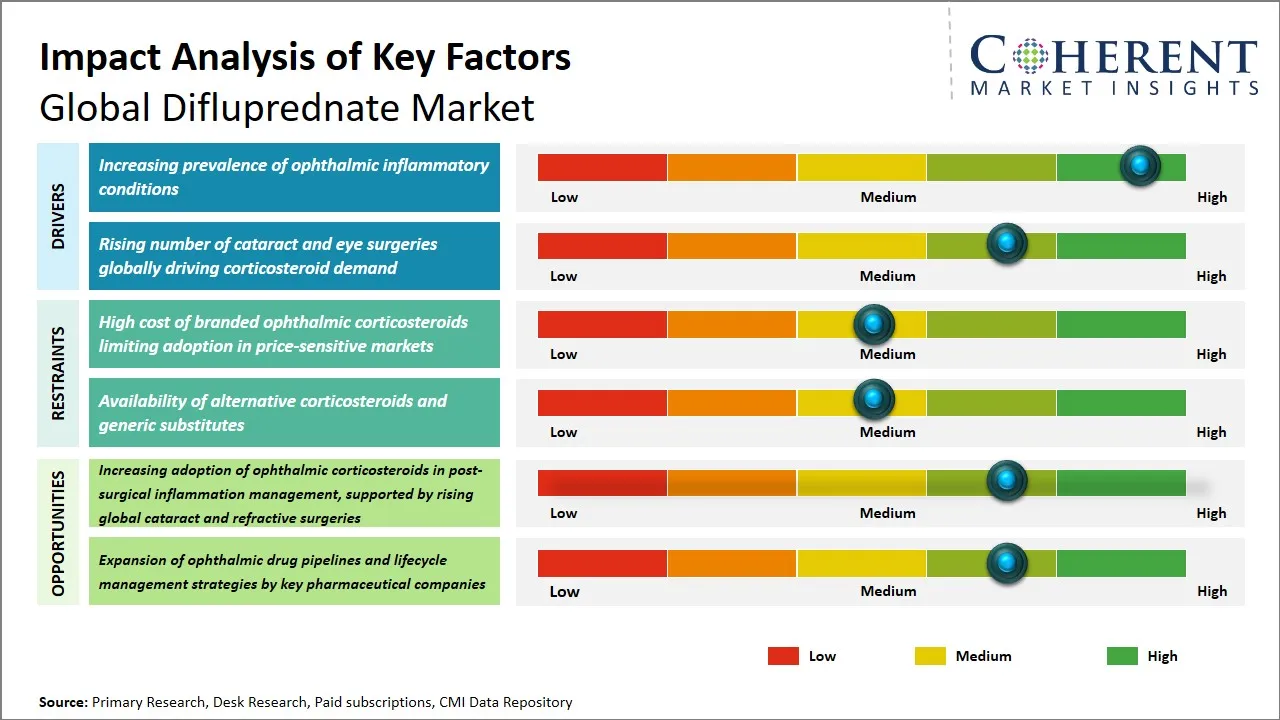
Global Difluprednate Market Driver – Increasing Prevalence of Ophthalmic Inflammatory Conditions
The increase in the occurrence of ophthalmic inflammatory conditions has been a major factor in the increase in demand for difluprednate because of its corticosteroid potency in effectively treating ocular inflammatory conditions. Uveitis, ocular inflammation following a surgical procedure, allergic conjunctivitis, and other inflammatory ocular conditions have been on the increase due to various causes, including the aging factor, increase in autoimmune diseases, and exposure to environmental toxins.
These ocular inflammatory conditions need to be treated effectively to avoid complications that can lead to loss of sight; thus, effective anti-inflammatory agents like difluprednate are necessary in the treatment of ocular inflammatory conditions. In addition, the increase in detection of ocular inflammations through various diagnostic tests has led to therapeutic interventions in the treatment of ocular inflammatory conditions. As people seek to use therapeutic agents that can give quick relief from disease manifestations without side effects, the potency of difluprednate in effectively reducing inflammation in the eye and relieving eye pain makes it a preferable therapeutic agent in the treatment of ocular inflammatory conditions. Therefore, the increase in the occurrence of ocular inflammatory conditions directly translates to an increase in demand for difluprednate treatments.
For instance, in July 2025, a study published in Frontiers in Public Health noted that uveitis has a prevalence of 38-714 cases per 100,000 population, and incidence rates of 17-52 cases per 100,000 population annually, indicating a substantial and increasing public health problem.
(Source: Frontiers Media SA)
Global Difluprednate Market Opportunity - Increasing Adoption of Ophthalmic Corticosteroids in Post-Surgical Inflammation Management, Supported by Rising Global Cataract and Refractive Surgeries
A significant opportunity in the global difluprednate market is the increasing need for effective post-operative inflammation control in eye surgeries, particularly cataract and refractive procedures. The increasing number of patients needing anti-inflammatory treatment after surgery is largely due to the growing number of cataract surgeries.
The aging population, coupled with the rising prevalence of vision correction procedures such as LASIK and PRK, is a significant factor. Properly managing inflammation after surgery is essential as it directly impacts the healing process and minimizes the risk of complications. As a result, there is a greater need for an effective corticosteroid, such as difluprednate.
Ophthalmologists often choose difluprednate because it works quickly, is very potent, and penetrates the eye better than other corticosteroids. In addition, advancements in ophthalmic formulations have improved patient compliance and outcomes. Moreover, the increasing healthcare infrastructure and healthcare expenditure in emerging nations are expected to drive the demand for difluprednate in post-operative management of inflammation in ophthalmic procedures.
For instance, in March 2025, according to the study titled "The future of cataract surgery" published in Springer Nature Limited reported that over 20 million cataract surgeries are performed globally each year, with the number continuing to rise due to aging demographics and improved surgical access.
(Source: Springer Nature Limited)
Analyst Opinion (Expert Opinion)
- The growing trend of ophthalmological procedures, especially cataract and refractive surgery is likely to remain a key driver for the difluprednate market. The anti-inflammatory activity of difluprednate is more potent compared to its peers, and its increased activity in the eyes makes it a more preferred choice for post-surgical care.
- The rise in cases of ocular inflammatory disorders, such as uveitis and post-surgical inflammation is also contributing significantly to market growth. Alongside this, an increase in diagnosis rates and access to eye care is further adding momentum to market growth in general.
- The proliferation of generic difluprednate, coupled with improvements in how eye medications are administered is fueling expansion in developing markets.
Market Segmentation
- Product Type Insights (Revenue, USD Mn, 2021 - 2033)
- Branded
- Generics
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Post-Operative Ocular Inflammation
- Uveitis
- Allergic Conjunctivitis
- Other Eye Disorders
- Distribution Channel Insights (Revenue, USD Mn, 2021 - 2033)
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Novartis AG
- Amneal Pharmaceuticals LLC
- Cipla
- Reddy’s Laboratories Ltd
- Alembic Pharmaceuticals Limited
- Upsher Smith Laboratories
- Axplora
- Symbiotec Pharmalab
- LGM Pharma
- Viatris Inc
Sources
Primary Research Interviews
Industry Stakeholders
- Ophthalmic Drug Manufacturers
- Pharmaceutical Companies
- Contract Manufacturing Organizations (CMOs)
- Raw Material Suppliers (API Manufacturers)
- Regulatory and Compliance Experts
- Distributors and Hospital Procurement Specialists
End Users
- Hospitals and Ophthalmology Clinics
- Ambulatory Surgical Centers (ASCs)
- Eye Care Specialists and Ophthalmologists
Government and International Databases
- World Health Organization (WHO) – Eye health and disease burden reports
- U.S. Food and Drug Administration (FDA) – Drug approvals and safety data
- European Medicines Agency (EMA) – Regulatory approvals and guidelines
- National Institutes of Health (NIH) – Clinical research and ophthalmology studies
- ClinicalTrials.gov – Ongoing and completed ophthalmic drug trials
Trade Publications
- Pharmaceutical Technology
- Fierce Pharma
- BioPharma Dive
- Ophthalmology Times
- Drug Store News
Academic Journals
- American Journal of Ophthalmology
- Journal of Ocular Pharmacology and Therapeutics
- International Journal of Pharmaceutics
- Clinical Ophthalmology
- Drug Design, Development and Therapy
Reputable Newspapers
- The Wall Street Journal – Healthcare & pharma industry insights
- Financial Times – Pharmaceutical market trends
Industry Associations
- American Academy of Ophthalmology (AAO)
- International Council of Ophthalmology (ICO)
- Association for Research in Vision and Ophthalmology (ARVO)
- European Society of Cataract & Refractive Surgeons (ESCRS)
Public Domain Resources
- ClinicalTrials.gov
- WHO Reports
- NIH Databases
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 10 Years
Share
Share
About Author
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients