Global Next-Generation Genome Mapping Market Size and Forecast – 2026 To 2033
The global next-generation genome mapping market is expected to grow from USD 184.2 Mn in 2026 to USD 611.3 Mn by 2033, registering a compound annual growth rate (CAGR) of 18.7%. The global next-generation genome mapping market is poised for significant expansion, fueled by the soaring global burden of genetic disorders.
For instance, the World Health Organization estimated that about 7.9 Mn births or 6% of total births annually occur with a defect of genetic or partially genetic origin. Furthermore, eastern mediterranean countries (such as Saudi Arabia, Iran, Egypt, and Pakistan) contribute up to 26% of global patients with β-thalassemia. As many as 50% can carry the gene for α-thalassemia, while 30% of certain communities have the sickle cell disease.
(Source: World Health Organization)
Key Takeaways of the Global Next-Generation Genome Mapping Market
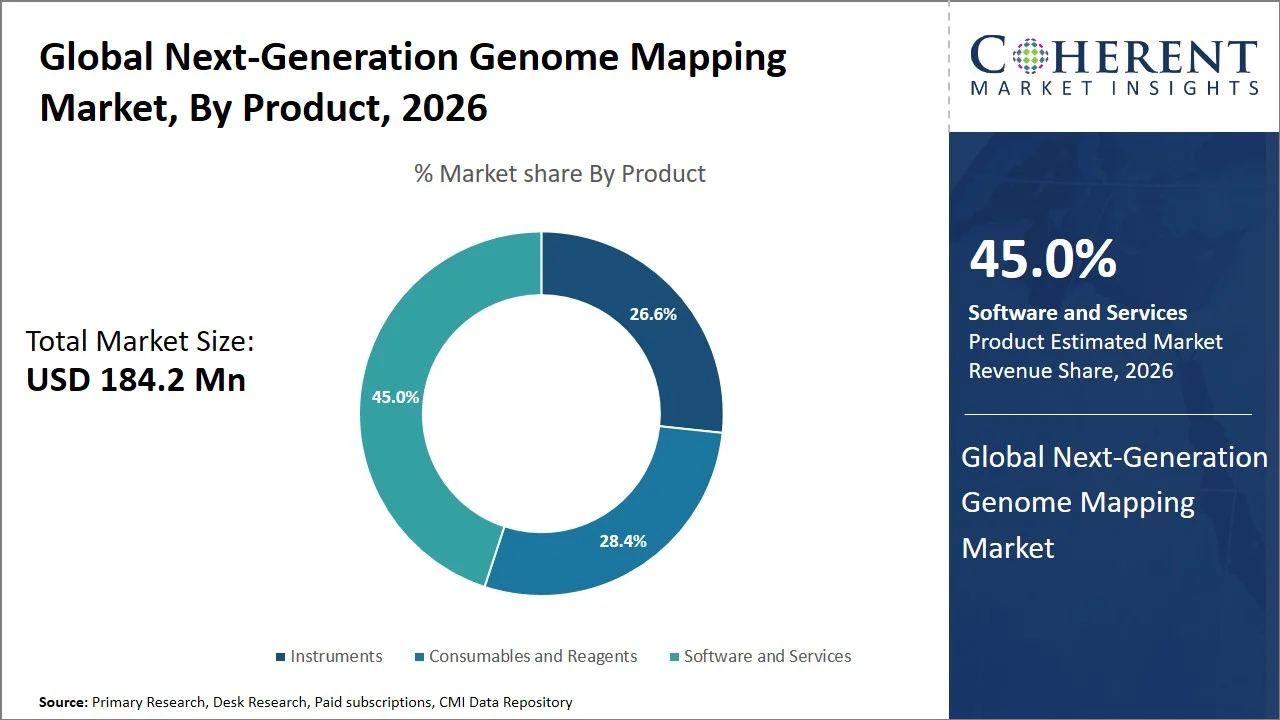
- Software and services are projected to hold 45.0% of the global next-generation genome mapping market share in 2026, making it the dominant product segment, with North America taking the lead in adoption attributed to the presence of a highly evolved bioinformatics network and high incorporation of genomic analysis in the cloud into the workflow process. This domination is fueled by the requirement to analyze data produced by sophisticated mapping technologies. For instance, the National Institutes of Health funds several major genomic data infrastructures, including the “All of Us Research Program”, which is highly dependent on software tools to interpret genomic data, further emphasizing the need for efficient genome mapping software programs. (Source: National Institutes of Health)
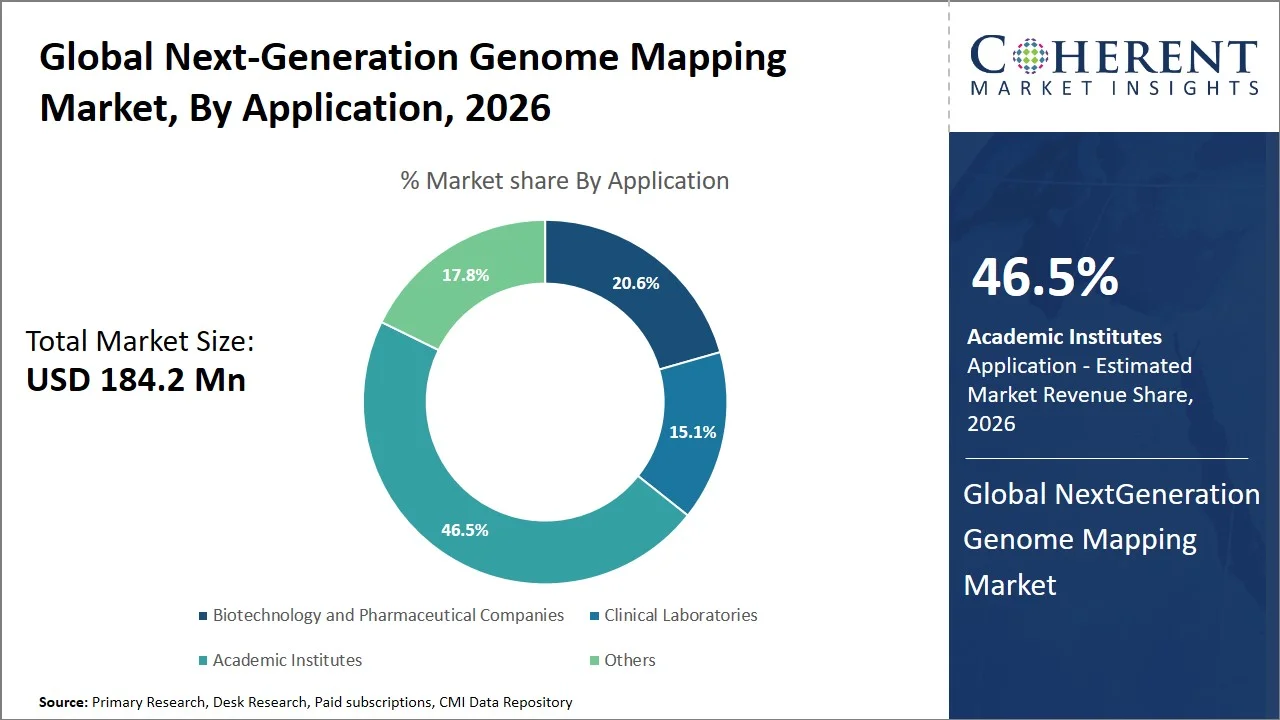
- Academic institutes are projected to hold 46.5% of the global next-generation genome mapping market share in 2026, making it the dominant application segment, with Europe emerging as a central hub owing to its publicly supported genomics research projects and cross-border collaborations (such as 1+ Million Genomes Initiative, ELIXIR). Universities and research institutions serve as the main users of genome mapping technology to analyze genetic differences and diseases. For instance, UK Biobank has sequenced the genomic data of 500,000 people, facilitating extensive studies related to complex diseases, which contributes greatly to the need for next-generation genome mapping services among academia. (Source: UK Biobank Limited)
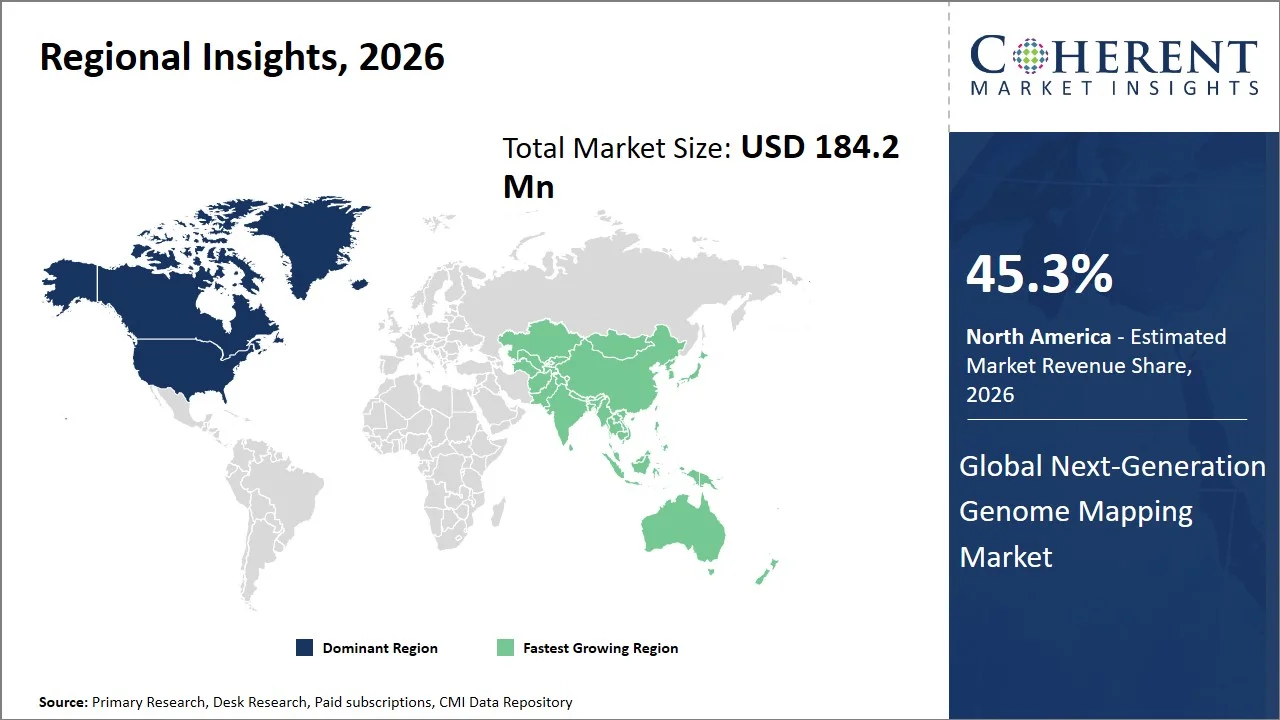
- North America maintains dominance with an expected share of 45.3%in 2026, supported with robust commercialization efforts for genome mapping technologies, combined with quick clinical implementation in oncology and rare disease testing. This region enjoys an advanced approach to coverage, along with the broad application within specialized genomic laboratories. For instance, the National Human Genome Research Institute supports initiatives like the Centers for Common Disease Genomics, where extensive genome sequencing and structural variations are used to study diseases. (Source: National Human Genome Research Institute)
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 21.5% share in 2026, driven by greater use of genome mapping within population studies and growth in biobanks. Countries like Japan and South Korea are implementing genomics within their nation’s health care systems. For instance, the Tohoku Medical Megabank Organization had established a biobanking facility that integrates genomics and clinical information for precision medicine, thus advancing the application of sophisticated genome-mapping technology in the region. (Source: Tohoku University Tohoku Medical Megabank Organization)
- Rising Relevance of Structural Variant Analysis in Complex Disease Research: Conventional sequencing approaches tend to overlook large structural changes, thereby fueling the need for new genome mapping platforms, which enable researchers to obtain a bigger picture of the genome. In cancer, neurological disease, and rare diseases with underlying genetic anomalies, genome structure alterations are particularly relevant, making genome mapping an increasingly important complement to sequence analysis.
- Increasing Adoption in Biopharma R&D for Cell and Gene Therapy Development: Genome mapping is gaining popularity among the steps taken by biopharmaceutical firms, especially those dealing with cell and gene therapies where it is essential to establish the identity of the genome. Genome mapping is critical when trying to ascertain the stability of the genome and checking for any side effects not intended. With the current requirements from regulatory bodies to ensure the safety of the genome and validate it, genome mapping has now become one of the first steps in the pipeline process.
Why Does Software and Services Segment Dominate the Global Next-Generation Genome Mapping Market?
Software and services are projected to hold the market share of 45.0% in 2026, attributed to the essential need for bioinformatics tools to analyze, store, and interpret vast amounts of genomic data generated by the use of high-throughput genome mapping techniques. Unlike hardware, which represents a one-time investment, software platforms and services are needed continuously for detecting variations, analyzing structures, and deriving insights for clinical purposes. This trend is further strengthened by the advent of cloud-based genomics data ecosystems and AI-driven analytics in the industry.
For instance, Illumina, Inc. has bolstered its software environment by creating platforms such as Illumina Connected Analytics that allow cloud-based and scalable computing for big genomic data sets. This platform features enhanced workflows to analyze structural variants as well as perform multi-omics analyses for genome mapping (Source: Illumina, Inc.). Concurrently, Thermo Fisher Scientific Inc. provides bioinformatics integration services via its Ion Torrent, Torrent Suite Software and Ion Reporter Software, which facilitate the entire process of genomics workflow including variant calls, annotations, and reporting. The company’s bioinformatics tools provide an effective solution for analyzing genome mapping data (Source: Thermo Fisher Scientific Inc.). These developments underscore the importance of sustained innovations in the fields of data analytics and interpretation for sustaining the segment’s leading position.
Why are Academic Institutes the Most Preferred Application?
To learn more about this report, Request Free Sample
Academic institutes are projected to hold a market share of 46.5% in 2026, owing to their high emphasis on basic research, validation of new technologies, and early-stage discovery phase studies. Academic institutes make wide use of innovative genome mapping techniques for studying structural variation, gene expression, and genome structure in view of consistent public funding and availability of substantial patient populations for conducting research.
For instance, Human Genome Structural Variation Consortium studies structural variations in the human genome through advanced long-read sequencing and genome mapping technology. The purpose of the consortium is to create genomic data sets with high resolution to enhance knowledge about genetic variation and help in the development of genomic analysis technologies (Source: The International Genome Sample Resource (IGSR)). In addition, Genomics England through the 100,000 Genomes Project allows academic scientists to investigate rare diseases and cancer genomics on a larger scale, thereby promoting the use of the latest genome mapping technology (Source: Genomics England). Such large-scale academic ventures continue to fuel continued demand for next generation genome mapping technologies globally.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
U.S. FDA Final Rule on Laboratory Developed Tests (LDTs) Oversight (April 2024–Ongoing Implementation) |
|
|
UK Genomics England & NHS Genomic Data Integration Framework Expansion (2022–2025 Policy Push) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: U.S. Food and Drug Administration, Department of Health and Social Care)
Global Next-Generation Genome Mapping Market Dynamics
To learn more about this report, Request Free Sample
Market Drivers
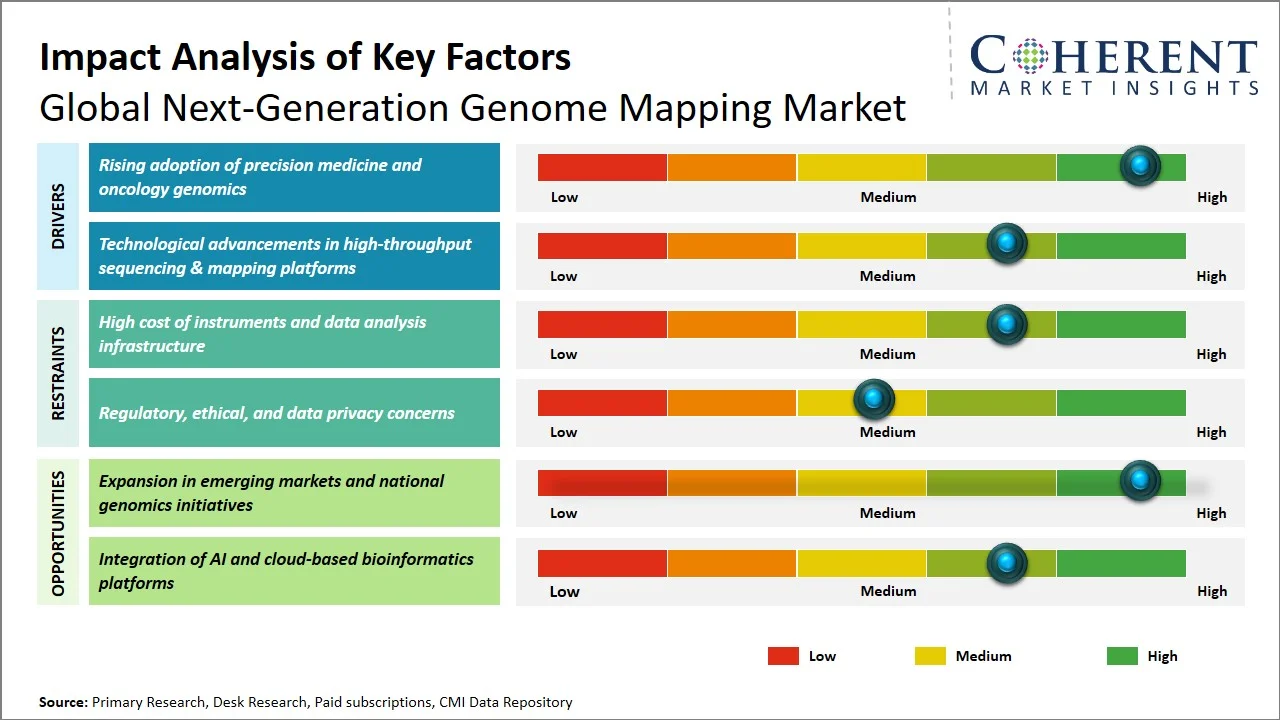
- Rising adoption of precision medicine and oncology genomics: Precision medicine's growing trend of implementation has led to a rising demand for genome mapping technology in the coming years, especially in oncology where genotyping plays an important role in determining patient treatment. Genome mapping provides the advantage of detecting structural variants, copy number variations, and complex mutations which cannot be captured with sequencing methods. For instance, according to the National Cancer Institute, precision oncology makes use of advanced genomic testing in order to link individual patients to targeted drugs. The National Cancer Institute also encourages participation in massive efforts like the Cancer Genome Atlas (TCGA), where genomics have been used to map out changes in various cancers, helping to isolate actionable mutations (Source: National Cancer Institute). With continued advances in cancer treatment through biomarker-guided strategies, the demand for next-generation genome mapping technologies that can offer high resolution is on the rise.
- Technological advancements in high-throughput sequencing & mapping platforms: The consistent evolution in sequencing and genome mapping techniques plays an important role in the growth of the market, as the techniques help in the rapid and accurate analysis of the genomes. With innovations like long-read sequencing and optical genome mapping, it becomes possible to detect complex genetic structures and structural changes efficiently. For instance, PacBio provides HiFi sequencing technology that provides highly accurate sequences with longer reads to detect complex genomic structures. This development results in better genome mapping abilities and more accurate identification of disease-related genetic mutations (Source: PacBio). The improvements in technology have been contributing to better research capabilities and faster adoption among clinical and academic applications.
Emerging Trends
- Convergence of Genome Mapping with AI-Driven Multi-Omics Platforms: Genome mapping is finding increasing use in association with multi-omics approaches facilitated by AI that involves genomic data in conjunction with transcriptomic and epigenomic data. The integration of these technologies allows better interpretation of genomic variation in context. Genome mapping is therefore moving from an independent method to becoming one part of an ecosystem of biological information.
- Shift Toward Decentralized and Cloud-Based Genome Mapping Workflows: There is a shift towards more efficient use of genomic data mapping through cloud computing technology, moving away from lab-based, centralized analysis techniques. It will be helpful in increasing scalability and lowering the dependence on infrastructure, along with allowing real-time data sharing.
Regional Insights
To learn more about this report, Request Free Sample
Why is North America a Strong Market for Next-Generation Genome Mapping?
North America leads the global next-generation genome mapping market, accounting for an estimated 45.3% share in 2026, attributed to the rapid rate at which genomics technologies have been adopted in clinical settings along with initiatives on translational research towards structural variants and rare disease diagnosis. The region possesses an advantage over the existence of advanced infrastructures for clinical genomics, adoption of multi-omics approaches, and high-resolution genome mapping platforms in hospitals. For instance, National Organization for Rare Disorders actively promotes programs that aim to increase genetic tests and enable early identification of rare disorders using genomics approach. The organization promotes projects that foster cooperation among researchers, doctors, and patients to ensure increased application of new genome mapping techniques. (Source: National Organization for Rare Disorders, Inc.)
Additionally, the Geisinger Health System has adopted genomics-enabled care via various programs, including the MyCode Community Health Initiative, where genomics are integrated into the healthcare system. This program links genomic information with patients' electronic medical records for early identification of disease risks and treatment plans (Source: American Medical Association). Such extensive clinical integration and institutional backing ensure that North America will remain a strong market for next-generation genome mapping.
Why Does Asia Pacific Next-Generation Genome Mapping Market Exhibit High Growth?
The Asia Pacific next-generation genome mapping market is expected to exhibit the fastest growth with an estimated contribution of 21.5% share to the market in 2026, owing to the fast growth of national genomics projects, digitalization in healthcare, and investments made by countries towards the implementation of precise medicine infrastructure. For instance, MedGenome received an investment of USD 50 million from Novo Holdings, to aid in developing genome research and increasing its genome sequencing operations in the Asia Pacific region. These massive funding programs have greatly contributed to the adoption of genome mapping technologies. (Source: Novo Holdings)
Furthermore, there is an active development of genomics in healthcare by governments of Asia Pacific, thus driving the demand for innovative genome mapping technology in the region. For instance, China National GeneBank contributes to the generation of genomic information and bio-banking activities that can contribute towards research activities in the field of life sciences (Source: China National Center for Bioinformation). These large investments and genomics programs initiated by governments are anticipated to continue fueling Asia Pacific’s high growth in the next-generation genome mapping market.
Why is the U.S. Leading Innovation and Adoption in the Next-Generation Genome Mapping Market?
The U.S. plays a crucial role in innovation and implementation in the next-generation genome mapping market attributed to its well-developed platform for genomic studies, modern infrastructure, and technology development. The U.S. is known to lead innovations in genomics because of various initiatives like the Human Genome Project, which served as a foundation for the development of genome sequencing techniques used today. Furthermore, there are some leading research centers that specialize in genomics, including the Joint Genome Institute, which offers high-throughput genome sequencing technology. (Source: Joint Genome Institute)
In addition to the existence of large-scale sequencing centers such as the McDonnell Genome Institute that have played a major role in the field of genome sequencing, the country’s ability to innovate has been further enhanced. Moreover, the U.S. always had a heavy investment in genomics, with a significant portion of global government funding allocated to the field, thereby fostering rapid technology advancement and clinical application. For instance, the National Institutes of Health (NIH) invested more than USD 3.8 billion into research programs related to genetics and genomics, including large-scale sequencing, analysis of data, and translational research projects. (Source: U.S. Department of Energy)
Is U.K. a Favorable Market for Next-Generation Genome Mapping Market?
U.K. is perceived to be a favorable market for the next-generation genome mapping owing to the presence of the government’s genomics strategy and its research framework. The U.K. is among those countries that have embraced the adoption of genomics in its health care services through sequencing processes carried out at the national level. This includes initiatives such as those by Genomics England, which has undertaken several sequencing projects including large-scale genome sequencing that aids research on rare diseases and cancers (Source: Genomics England Limited). Additionally, the U.K. has strong network systems for biobanks and cooperation among academic institutions and health systems, fueling demands for genome mapping technologies.
Is China Emerging as a Key Growth Hub for the Next-Generation Genome Mapping Market?
The next-generation genome mapping market is expected to witness significant growth in China, considering the strong government-backed initiatives to encourage precision medicine and the rapid development of sequencing capabilities in the country. The China government keeps on emphasizing the importance of genomics in the national strategy of modernizing healthcare systems. For instance, BGI Genomics, one of the leading genomics companies, has greatly increased its high-throughput sequencing and genome analysis capacity, allowing them to conduct large population and disease genome studies (Source: BGI Genomics). Further, there is an increasing adoption of genomics testing in hospitals and public-private sector collaboration that drives the growth in the demand for next-generation genome mapping market.
Why Does Germany Top the Europe Next-Generation Genome Mapping Market?
Germany is the leading Europe market for next-generation genome mapping attributed to the advanced clinical research environment in Germany, as well as the implementation of genome-based technologies for precision medicine purposes in the country. Indeed, Germany has well-developed cooperation between academia, hospitals, and biotechnology companies that ensures fast transition of genomic discoveries to their implementation. For instance, the German Human Genome-Phenome Archive provides safe storage and exchange of genomic and phenomics data for scientific purposes (Source: The German Human Genome-Phenome Archive). Moreover, the emphasis of Germany on using genomics within healthcare in conjunction with digital health programs further propels the adoption of the next- generation genome mapping tools.
Is Next-Generation Genome Mapping Market Developing in Japan?
The Japan is becoming a promising destination for next-generation genome mapping in the coming years owing to its favorable policies regarding precision medicine and the use of genomic technologies in the healthcare industry (such as the Ministry of Health, Labor and Welfare’s national genomic medicine initiatives and the Japan Agency for Medical Research and Development’s funding programs supporting genomic research and clinical implementation) Indeed, there are efforts being made by the government to ensure that genomic data is maximally exploited using digital health strategies. For instance, the BioBank Japan has created a vast genomic database based on patients, which helps in studying common diseases and developing medicines (Source: BioBank Japan). Such robust policy backing, integration of digital health care, and collaborative research efforts are anticipated to hasten the deployment of next-generation genome mapping market in Japan.
Regulatory Landscape Governing the Global Next-Generation Genome Mapping Market
|
Region |
Key Regulations |
Description |
Impact on Market |
|
North America |
U.S. FDA, CMS (CLIA), FTC |
Genome mapping technologies used in diagnostics fall under medical device and genetic test regulations. Clinical labs must comply with CLIA standards, while U.S. FDA oversight is expanding to ensure analytical and clinical validity of genomic tests. |
Increasing regulatory scrutiny is improving test reliability and clinical adoption. However, stricter validation and compliance requirements may raise entry barriers for emerging genome mapping players. |
|
Europe |
IVDR (In Vitro Diagnostic Regulation), GDPR, national health authorities |
Genome mapping applications are regulated under IVDR for diagnostic tools and GDPR for genomic data privacy. Strong emphasis is placed on patient consent, data protection, and cross-border data governance. |
Stringent data privacy and clinical validation frameworks enhance trust and interoperability, but increase compliance complexity, especially for data-intensive genome mapping platforms. |
|
Asia Pacific |
Country-specific frameworks (e.g., Japan PMDA, China NMPA, India ICMR guidelines) |
Regulatory frameworks across APAC are evolving, focusing on genomic research ethics, biobanking standards, and clinical validation of genetic technologies. Some countries are actively updating policies to accommodate emerging genomic innovations. |
Regulatory diversity creates both opportunity and complexity—faster approvals in some markets drive adoption, while lack of harmonization may limit scalability for global genome mapping providers. |
|
Latin America |
National health agencies, biosafety laws (e.g., Brazil ANVISA, Mexico COFEPRIS) |
Genomic technologies are primarily governed under broader biotechnology and genetic testing regulations, with growing attention to ethical use, patient consent, and biosafety compliance. |
Developing regulatory frameworks present growth opportunities, but limited standardization and infrastructure may slow widespread adoption of advanced genome mapping solutions. |
|
Middle East & Africa |
National health ministries, emerging genomics policies (e.g., UAE, Saudi genomics programs) |
Regulatory systems are in early development stages, often tied to national genomics initiatives and healthcare modernization programs, with increasing focus on data governance and research ethics. |
Early-stage regulatory environment enables pilot projects and innovation; however, lack of mature frameworks may restrict commercialization and cross-border data integration. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How are the integration of AI and cloud-based bioinformatics platforms creating new growth opportunities in the global next-generation genome mapping market?
AI integration with cloud computing is proving to be highly beneficial in terms of growth prospects for the emerging next-generation genome mapping market by providing rapid, scalable, and affordable ways of analyzing big genomic data. AI enhances the ability to detect structural variants and interpret genomes, while cloud computing allows the elimination of high costs associated with building an initial structure.
For instance, DNAnexus offers a cloud solution that is integrated with AI algorithms for the purpose of genomics analysis and is currently being used extensively in large-scale sequencing studies and clinical research (Source: DNAnexus). Additionally, Roche offers NAVIFY Mutation Profiler which is cloud-based software that allows interpretation of genomic information in oncology, thereby enabling faster and better decisions (Source: F. Hoffmann-La Roche Ltd.). This innovation has greatly improved access and efficiency in next-generation genome mapping processes.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In February 2026, Illumina, Inc. unveiled its latest initiative known as TruPath Genome, a state-of-the-art solution meant for improving genomic analysis through improved identification of genomic complexities. The product is geared towards achieving increased accuracy of genome analyses. It indicates efforts by the organization to advance its next generation genome mapping processes.
- In February 2026, Complete Genomics signed a definitive deal regarding its acquisition by Swiss Rockets AG. The objective of the acquisition was to enhance genomics technology skills and to expand precision medicine projects. This is a consequence of the increased strategic investments and consolidation seen in the industry of genome mapping.
- In April 2024, NewBiologix introduced next generation sequencing and optical genome mapping solutions. The innovation is geared towards making structural variation detections and genome mapping more accurate. It also demonstrates the involvement of new firms in boosting genome mapping techniques and stiffening competition in the market.
Competitive Landscape
The global next-generation genome mapping market will likely stay highly competitive in light of increased demand for accurate structural variation detection and higher utilization of genomics technologies in clinical practices and research settings. Ongoing innovations in high-throughput mapping technologies, along with increased use in the diagnosis of oncological diseases and rare disorders, will add to competition in the market. Moreover, the combination of genome mapping with next-generation sequencing and bioinformatics will alter the competitive landscape. Key focus areas include:
- Advancement in high-resolution genome mapping technologies enabling precise detection of large structural variations
- Integration of bioinformatics, automation, and AI-driven analytics to enhance data interpretation and workflow efficiency
- Development of end-to-end solutions combining genome mapping with sequencing and clinical applications
- Expansion of partnerships with research institutions and biopharma companies for translational and clinical adoption
Market Report Scope
Next-Generation Genome Mapping Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 184.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 18.7% | 2033 Value Projection: | USD 611.3 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Bionano Genomics, Inc., Thermo Fisher Scientific Inc., Illumina, Inc., F. Hoffmann-La Roche Ltd., Agilent Technologies, Inc., QIAGEN N.V., Oxford Nanopore Technologies Ltd., Pacific Biosciences of California, Inc., PerkinElmer, Inc., and 10x Genomics, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- In the coming years, the prospects of the next-generation genome mapping market will depend on research market, but it would venture into the fully-integrated clinical market sector, where the technology would find more practical applications, including personalized medicine, cancer diagnostics, and rare disease analysis. With the technological advancements in genome mapping with the application of long-read sequencing and optical genome mapping, the industry would shift focus to the convergence of omics data and genomics analysis using AI methodologies.
- The Asia Pacific region is anticipated to exhibit the highest growth opportunities in the software & services segment, particularly in the economies such as China and India, on account of the growing need for genomic programs and increasing trends towards healthcare digitization in these countries. Moreover, there will be a continued high demand for clinical applications of the technology in oncology and rare diseases in the regions such as North America and Europe.
- To gain a competitive edge, the parties in the market players must focus on developing a platform that will include all aspects of DNA processing, like sequencing, mapping, and intelligent computing analysis, apart from cloud-based bioinformatics. This can be achieved through cooperation with hospitals, universities, research organizations, even governmental agencies involved in genomics studies, while also taking care of financial and regulatory aspects.
Market Segmentation
- Product Insights (Revenue, USD Mn, 2021 - 2033)
- Instruments
- Consumables and Reagents
- Software and Services
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Biotechnology and Pharmaceutical Companies
- Clinical Laboratories
- Academic Institutes
- Others
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Bionano Genomics, Inc.
- Thermo Fisher Scientific Inc.
- Illumina, Inc.
- Hoffmann-La Roche Ltd.
- Agilent Technologies, Inc.
- QIAGEN N.V.
- Oxford Nanopore Technologies Ltd.
- Pacific Biosciences of California, Inc.
- PerkinElmer, Inc.
- 10x Genomics, Inc.
Sources
Primary Research Interviews
- Genome mapping technology providers, bioinformatics experts, and structural variation specialists from leading genomics companies and research labs
- Clinical geneticists and molecular diagnostics professionals involved in rare disease and oncology applications
- CRO executives and genomics service providers engaged in large-scale genome analysis and mapping workflows
Stakeholders
- Next-Generation Genome Mapping Solution Providers: (e.g., genome mapping platform developers, optical mapping technology providers, bioinformatics solution companies)
- End-use Sectors
- Pharmaceutical & Biotechnology Companies
- Academic & Research Institutes
- Contract Research Organizations (CROs)
- Government & Public Health Research Bodies
- Regulatory & Health Bodies: U.S. Food and Drug Administration (FDA), National Institutes of Health (NIH), European Medicines Agency (EMA), World Health Organization (WHO) – genomic data governance, diagnostics validation, and research funding frameworks
Databases
- NCBI Genome & Maps – comprehensive genome sequence and mapping data repository
- dbVar (NCBI) – archive of large-scale genomic structural variations such as insertions, deletions, and translocations
- Database of Genomic Variants (DGV) – curated catalog of human structural genomic variations
- Genome Variation Map (GVM) – repository of SNPs and genomic variation datasets across species
Magazines
- Nature (News & Features section) – genomics technology advancements and structural variation research
- Science Magazine – innovations in genome mapping and genomic medicine
- NIH Research Matters – updates on genome research, mapping technologies, and structural genomics
Journals
- Nature Genetics
- Genome Research
- American Journal of Human Genetics
- Nucleic Acids Research
Newspapers
- Government press releases and official announcements (e.g., NIH, European Commission, national genomics missions)
- Public communication platforms of health ministries and national genomics programs
Associations
- Human Genome Organization (HUGO)
- American Society of Human Genetics (ASHG)
- European Society of Human Genetics (ESHG)
- International Society for Computational Biology (ISCB)
Public Domain Sources
- National Institutes of Health (NIH) – genomics funding programs and research initiatives
- National Human Genome Research Institute (NHGRI) – genome mapping and sequencing programs
- European Commission – Horizon Europe genomics and health data funding
- UK Research and Innovation (UKRI) – national genomics infrastructure investments
- U.S. Department of Energy (DOE) – genome science and bioinformatics programs
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients