Global Trazodone Market Size and Forecast – 2026 To 2033
The global trazodone market is expected to grow from USD 687.2 Mn in 2026 to USD 907.4 Mn by 2033, registering a compound annual growth rate (CAGR) of 4.1%. The global trazodone market is poised for significant expansion, fueled by the rising prevalence of mental health disorders (particularly depression and insomnia).
According to World Health Organization, an estimated 280 million people, including 5% of all adults, experienced depression. Additionally, more than 10% of pregnant women who have just given birth experience depression.
(Source: World Health Organization)
Key Takeaways of the Global Trazodone Market
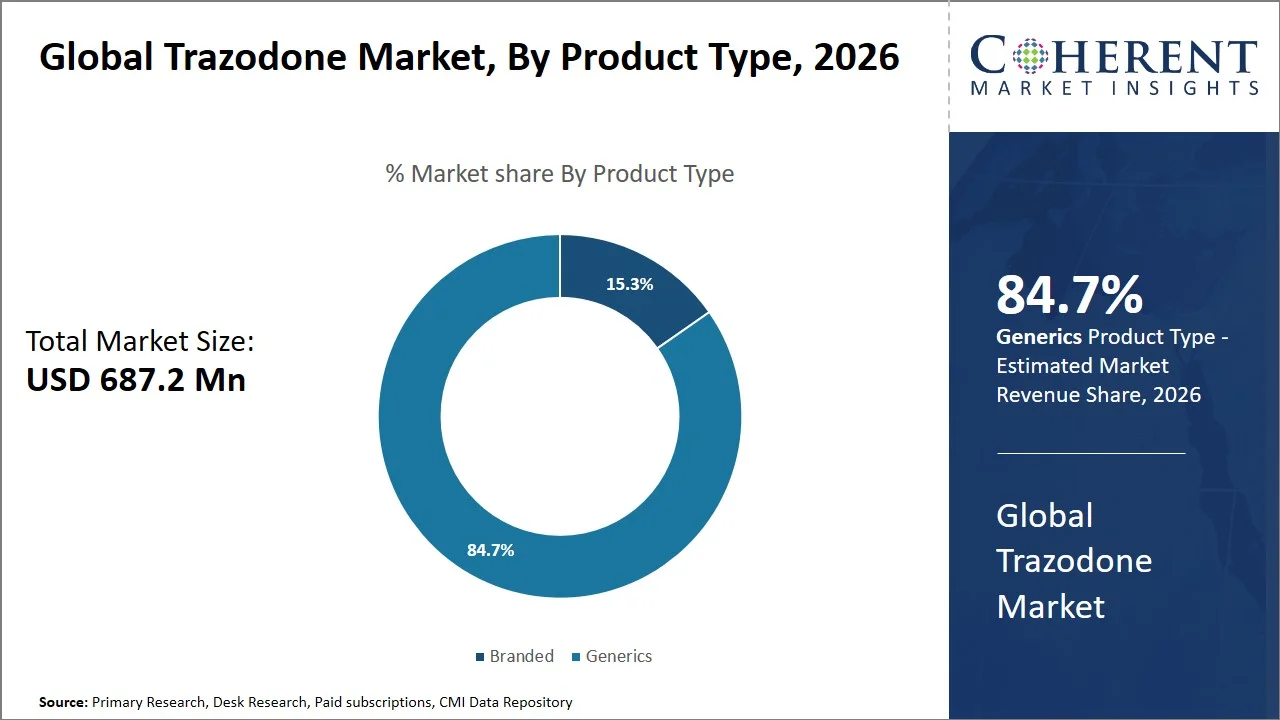
- Generics are projected to hold 84.7% of the global trazodone market share in 2026, making it the dominant product type segment. The high levels of prescriptions for conditions like depression enhance generic substitutions in these areas. For example, several generic forms of trazodone hydrochloride have been approved by the U.S. FDA using the Abbreviated New Drug Application (ANDA) process, which promotes the widespread availability of the drug at reduced costs, thus favoring the use of generics.
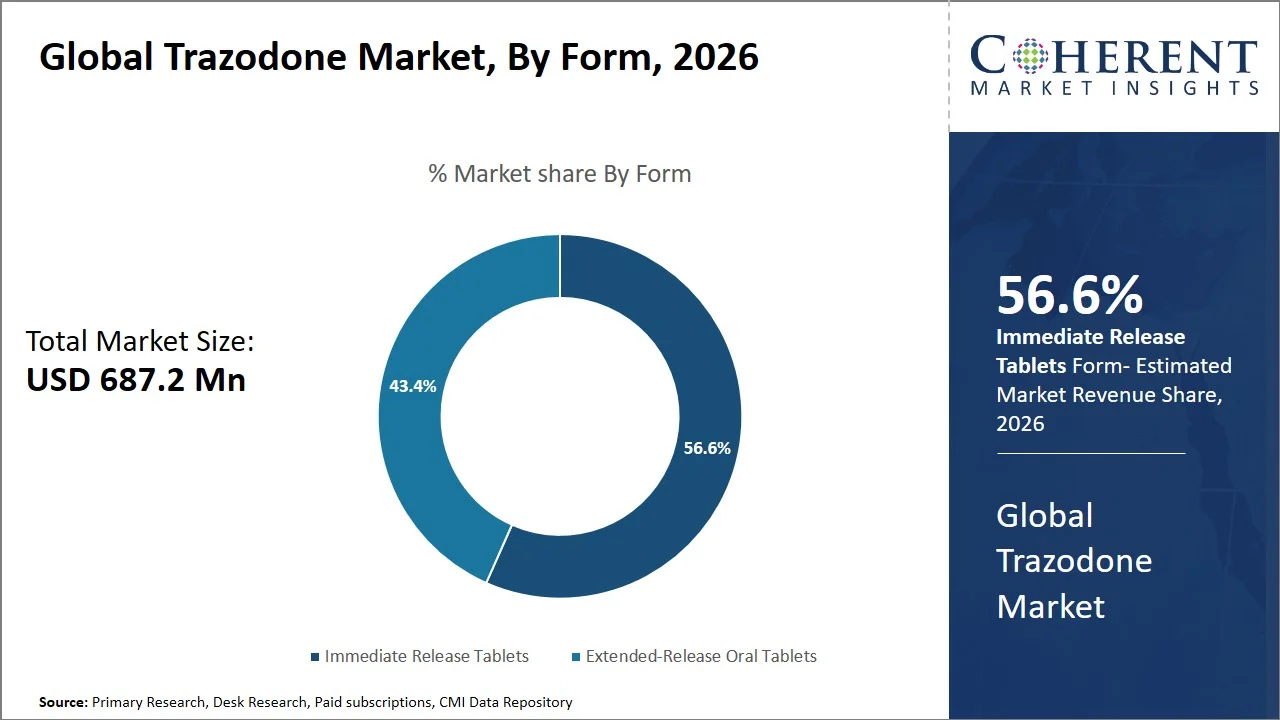
- Immediate release tablets are projected to 56.6% of the global trazodone market share in 2026, making it dominant form segment, given the high demand in both Asia Pacific and North America, as they are used for first-line treatment and dosage adjustment. In emerging Asia Pacific nations, doctors favor immediate-release versions owing to cost-effectiveness and titration ease, whereas in North America, doctors use them for quick relief of symptoms. For example, the immediate release trazodone hydrochloride tablets (available in strengths such as 50 mg and 100 mg) have been extensively used to treat depression as well as off-label insomnia, because of their ability to easily adjust the dose and quick onset.
- 50 mg segment is projected to hold 42.5% of the global trazodone market share in 2026, making it dominant strength segment, particularly in Europe and North America, since it forms the basis of the initial dosage for depression and insomnia-based conditions. In clinical settings in these areas, increasing the dosage gradually is common practice; hence, 50 mg becomes the most widely prescribed and dispensed dosage. For example, when using trazodone to treat insomnia and mild depression, an initial dose of 50 mg is taken before sleep, with the doctor gradually increasing the dosage depending on the individual’s reaction.
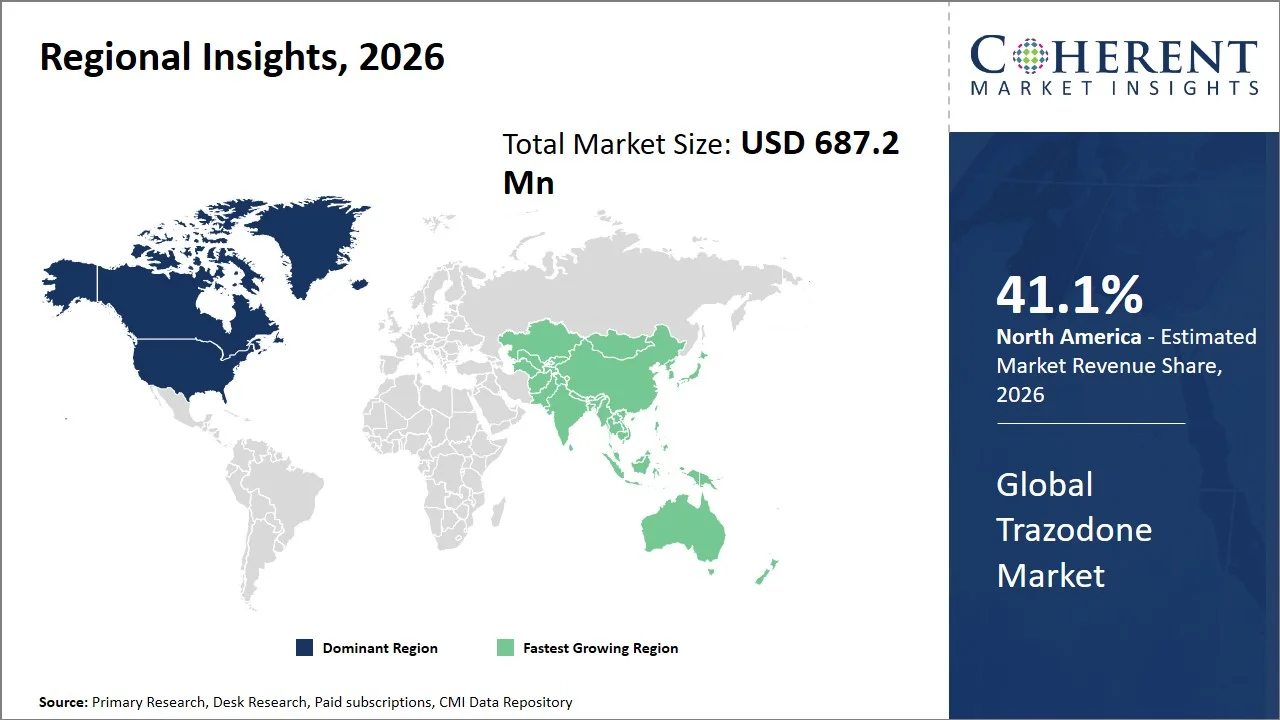
- North America market maintains dominance with an expected share of 41.1% in 2026, driven by increased prescriptions for antidepressants, strong diagnostic capabilities, and strong reimbursement programs (such as Medicare and Medicaid in the U.S.). The U.S. environment is characterized by high awareness of mental disorders, along with established clinical practice guidelines, which ensures the regular use of antidepressants such as trazodone. Also, favorable government regulations (e.g., Abbreviated New Drug Application (ANDA) pathway for generics, Hatch-Waxman Act promoting generic competition, and FDA Orange Book listing for therapeutic equivalence) by organizations like the U.S. FDA encourage the availability of branded as well as generic products, ensuring market leadership.
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 23.3% share in 2026, backed by a significant amount of untreated patients and growing government efforts to promote mental health programs. Countries such as India and China have been seeing a growing number of diagnosed cases and better accessibility to medications (such as the National Mental Health Program of India and the National Mental Health Work Plan [2023-2027] of China).
- Growing Use in Geriatric Psychiatry and Sleep Management: Trazodone is gaining popularity among the elderly because of its lesser anticholinergic side effects. There is increased need for safer dual-acting drugs among the elderly since the number of old people are increasing and hence the growing market segment for trazodone.
- Potential in Combination Therapies and Personalized Psychiatry: New studies are investigating the utility of trazodone in combination therapies alongside other drugs that treat depression or anxiety disorders. In addition, its application in pharmacogenomics-guided therapy offers significant potential.
Why Do Generics Dominate the Global Trazodone Market?
Generics are projected to hold the market share of 84.7% in 2026, because of the regulatory-driven substitution policy (e.g., U.S. state-level mandatory generic substitution laws and the Hatch-Waxman Act, which promotes generic drug entry through the ANDA pathway), and stiff competition after expiration of patents, thus ensuring increased accessibility.
As per data from the U.S. Food and Drug Administration, a single generic competitor can lead to price reductions of 30%, while five generics competing are associated with prices drops of nearly 85% compared to branded medicines, which makes them a lot more affordable (Source: U.S. Food and Drug Administration). In addition, the U.S. FDA mandates that generic medicines be proven to be bioequivalent and as effective, safe, and high-quality as the branded products. These regulatory safeguards, combined with substantial cost reductions, are key factors driving the dominance of generic trazodone across global markets. (Source: U.S. Food and Drug Administration)
Why are Immediate Release Tablets the Most Preferred Form?
To learn more about this report, Request Free Sample
Immediate release tablets are projected to hold a market share of 56.6% in 2026, because of the rapid onset of action and flexibility of dose titration and split dosing, especially needed in the treatment of depression and insomnia. The U.S. FDA describes immediate-release medications as those that rapidly dissolve in the gastrointestinal tract, thus facilitating quick absorption and fast action. (Source: U.S. Food & Drug Administration)
For trazodone, immediate-release tablets provide flexibility in terms of dose adjustment and splitting, as well as easy adjustment of the timing of administration, which can be helpful for personalized treatment approaches and side effect management. Another advantage of immediate-release trazodone tablets are their relatively low cost (e.g., NHS Drug Tariff prices indicate trazodone costs around USD 36–USD 46 per pack (€33–€42) depending on dosage strength—such as 100 mg at ~USD 46 (€42), 150 mg at ~USD 36 (€33), and 50 mg at ~USD38 (€35)—with regulated reimbursement). This is due to the widespread availability of generic versions. This makes them the most commonly prescribed dosage forms in clinical practice. (Source: NHS)
50 mg Segment Dominates the Global Trazodone Market
The 50 mg segment is projected to hold a market share of 42.5% in 2026, owing to its broad application as a first-line and maintenance dosage in the treatment of depression and insomnia, especially among geriatric and vulnerable populations. In accordance with the guidelines laid out by the U.S. FDA labeling information, trazodone is generally started with smaller dosages (for instance, 50-100 mg a day), thereby minimizing the incidence of adverse reactions and ensuring that the dosage is adjusted in relation to the patients' response to medication. (Source: U.S. Food & Drug Administration)
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Updated U.S. FDA Guidance on Trazodone Bioequivalence & Formulations (Regulatory Shift – 2024) |
|
|
Rising Generic Drug Approvals Strengthening Market Competition (Industry / Regulatory Shift – 2024–2025) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: U.S. Food and Drug Administration, U.S. Food and Drug Administration)
Global Trazodone Market Dynamics
To learn more about this report, Request Free Sample
Market Drivers
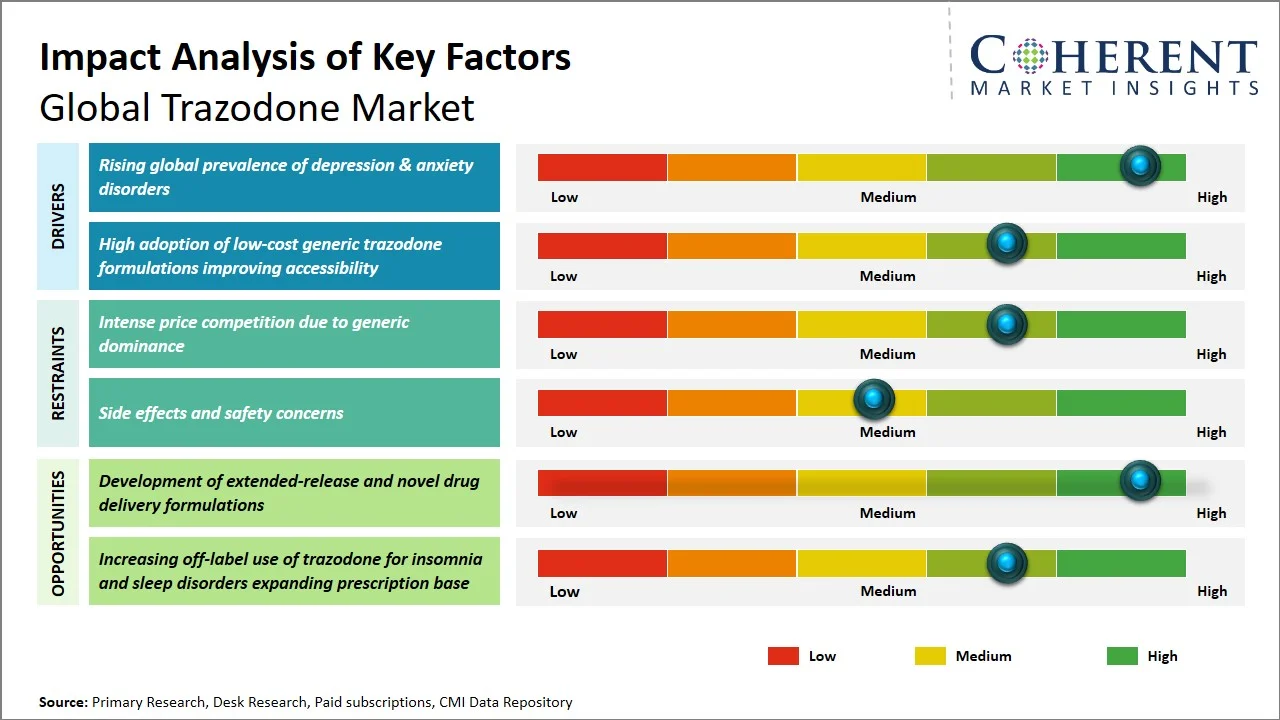
- Rising global prevalence of depression & anxiety disorders: The rising burden of mental health disorder is a significant factor fueling the trazodone market (commonly prescribed for both depression and anxiety). The World Health Organization estimates that more than a billion people worldwide are grappling with mental health conditions. Anxiety disorders, for instance, impact roughly 4.4% of the global population. This surge in prevalence is directly boosting the need for antidepressants such as trazodone owing to its ability to address both depression and sleep- related disorders. (Source: World Health Organization)
- High adoption of low-cost generic trazodone formulations improving accessibility: The widespread availability of inexpensive trazodone generics has greatly enhanced its treatment availability, especially in regions that cannot afford expensive medicines. The U.S. FDA has revealed that in the U.S., generic drugs make up a significant portion of all prescriptions filled, as they make significant savings on health care costs. (Source: S. Food & Drug Administration) This high utilization rate of generics are reflective of the worldwide trend of seeking cheaper medicines, thus enhancing the treatment rate of mental disorders. Trazodone is an off-patent drug and thus available largely as generics; this makes its availability crucial for its market success.
Emerging Trends
- Increasing Off-Label Use for Insomnia and Anxiety: The use of Trazodone is increasing for treating insomnia and anxiety because of its sedative effect and lesser chances of addiction than other drugs used for sleep induction. Its use is also increasing outside its intended application of treating depression patients.
- Shift Toward Generic Dominance and Cost-Effective Therapies: Increasing prevalence of generics in the market is being witnessed owing to patent expiration and cost-control measures. Consequently, trazodone’s availability across the globe is on the rise, along with price pressures on drug manufacturers.
Regional Insights
To learn more about this report, Request Free Sample
Why is North America a Strong Market for Trazodone?
North America leads the global trazodone market, accounting for an estimated 41.1% share in 2026, owing to the prevalence of diagnoses and treatments for mental disorders, an advanced health care system in the region, and regulatory policies encouraging the sale of generics (e.g., the Hatch-Waxman Act and U.S. state-level generic substitution laws). As per the data presented by Centers for Disease Control and Prevention, 40% or 2 in 5 U.S. high school students reported experiencing symptoms of depression—felt so sad or hopeless every day for two or more weeks in a row that they stopped doing some usual activities in the past 12 months. Additionally, 5% or 1 in 20 U.S. adults regularly reported feelings of depression. (Source: Centers for Disease Control and Prevention) This combination of high prevalence of diseases, coupled with good health care facilities, and regulatory structures continues to contribute to increased demand of trazodone in North America.
Why Does Asia Pacific Trazodone Market Exhibit High Growth?
The Asia Pacific trazodone market is expected to exhibit the fastest growth with an estimated contribution of 23.3% share to the global market in 2026, owing to the surging prevalence of depression and mental health conditions and an expansion in mental healthcare initiatives led by governments (such as the National Mental Health Programme and the Mental Healthcare Act, 2017 in India, and the National Mental Health Work Plan in China).
In prominent countries of Asia Pacific such as India, government-backed data suggest that nearly 15% of adults require mental health intervention while around 1 in 20 adults suffers from depression (Source: World Health Organization). Additionally, India bears a high burden of mental health illnesses, 2,443 disability-adjusted life years (DALYs) per 10,000 population, the age-adjusted suicide rate per 100,000 population is 21.1. While, economic loss due to mental health conditions, between 2012-2030, is estimated at USD 1.03 trillion. (Source: World Health Organization)
Why are the U.S. Leading Innovation and Adoption in the Trazodone Market?
The U.S. plays a critical role in terms of innovation and adoption of trazodone because of its robust regulatory environment, high rates of prescriptions for psychiatric medications and persistent generics development. The U.S. FDA is keen on supporting the market growth through the Abbreviated New Drug Application (ANDA) process that helps to accelerate generics approvals and promotes competition. (Source: U.S. Food & Drug Administration) In addition, the U.S. healthcare sector reveals high usage of antidepressant medication owing to its clinical awareness and availability. Approvals and developments of new formulations contribute to innovation in the field and guarantee continued adoption of trazodone in the U.S.
Is U.K. a Favorable Market for Trazodone Market?
The U.K. represents an ideal target market for the sale of trazodone since it exhibits extensive usage of antidepressants, robust public healthcare services, and a larger population seeking mental illness services. According to the official report released by the NHS based on information provided by the government, approximately 8.7-8.9 million people in England received antidepressant medications between 2023 and 2025.(Source: NHS Business Services Authority) Additionally, over 92 million antidepressants were prescribed annually, thus demonstrating extensive demand for the pharmaceutical medication for treating depression and similar disorders. (Source: NHS Business Services Authority)
Furthermore, the long-term strategy by the NHS emphasizes further development of mental health services, community care, and treatment access. These factors combined with high numbers of patients undergoing treatment, increasing numbers of prescriptions, and strong governmental support (e.g., NHS Long Term Plan funding expansion, NHS Talking Therapies improving access to care, and post-COVID mental health recovery initiatives by the U.K. Government) for a robust healthcare system will ensure that trazodone will find its place on the British markets.
Is Japan Emerging as a Key Growth Hub for the Trazodone Market?
Japan appears to be a major growth hub for the trazodone market owing to the high incidence of aging population and occurrence of sleep disorders and depression. The Statistics Bureau of Japan reports that about 29.3% of the population in Japan is 65 years or older (as of 2024), which is among the highest ratios in the world (Source: Statistics Bureau of Japan). These individuals are at higher risk for insomnia and psychological disorders, and hence there is a greater need for treatment via medicines such as trazodone, which treats both problems.
Why Does Germany Top the European Trazodone Market?
Germany leads the market of trazodone in Europe fueled by the prevalence of mental diseases among Germans, high prescription rates, and the existence of a reimbursement policy. In Germany, government statistics show that nearly 17% of the adult population suffers from depression, while approximately one out of four adult patients suffer from a mental disorder yearly. As stated in the report from the Federal Statistical Office of Germany, Germany's statutory health insurance scheme provides coverage for almost 90% of its citizens. This ensures wide accessibility to antidepressants in Germany since the majority of the people have health insurance. (Source: Robert Koch Institute)
Trazodone market is evolving in China due to a growing number of mental disorders and greater involvement of the government to increase drug accessibility. In accordance with statistics compiled by the WHO, there are about 54 million depression patients and ~41 million patients with anxiety disorders in China (Source: WHO). In addition, the National Health Commission of China has launched a program (such as the National Mental Health Work Plan) aimed at enhancing the mental health sector and providing access to mental health services throughout the cities through the construction of specialized centers during 2023–2027, thus addressing problems of underdiagnosis and undertreatment.
Regulatory Landscape Governing the Global Trazodone Market
|
Region |
Key Regulations |
Regulatory Overview |
Impact on Market |
|
North America |
U.S. Food and Drug Administration (FDA) – ANDA pathway, Controlled Substances Act (where applicable), HIPAA |
Well-established regulatory framework for generic drug approvals through ANDA, with strict requirements on bioequivalence, manufacturing (cGMP), and labeling standards |
Ensures high drug quality and safety, boosting adoption; however, stringent approval processes increase compliance costs and time-to-market |
|
Europe |
European Medicines Agency (EMA), EU Pharmaceutical Directive, GDPR |
Harmonized regulatory system across EU member states with strong emphasis on drug safety, pharmacovigilance, and data protection |
Facilitates cross-border drug approvals and market access but adds complexity due to multi-country compliance requirements |
|
Asia Pacific |
Central Drugs Standard Control Organization (CDSCO), National Medical Products Administration (NMPA), PMDA (Japan) |
Diverse and evolving regulatory landscape with increasing alignment to global standards; growing focus on generic drug approvals and quality compliance |
Offers high growth potential due to expanding healthcare access, though regulatory variability can create entry barriers |
|
Latin America |
ANVISA (Brazil), COFEPRIS (Mexico) |
Developing regulatory frameworks with improving standards for generic drug approvals and manufacturing practices |
Supports gradual market expansion, but regulatory delays and varying standards may impact product launch timelines |
|
Middle East & Africa |
Saudi FDA (SFDA), South African Health Products Regulatory Authority (SAHPRA) |
Emerging regulatory systems with increasing focus on drug quality, import regulations, and local manufacturing policies |
Creates opportunities for market entry, though inconsistent regulations and approval timelines may pose challenges |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is the development of extended-release and novel drug delivery formulations creating new growth opportunities in the trazodone market?
The evolution of extended-release and new drug delivery systems are generating new revenue streams in the trazodone market through better patient compliance, improved dosing regimens, and enhanced efficacy, especially for chronic depression therapy. For example, in January 2024, the U.S. Food and Drug Administration approved an oral liquid formulation of Trazodone (Raldesy) for major depressive disorder, offering a ready-to-use solution that enables precise dose titration and improved compliance (Source: U.S. Food and Drug Administration) Besides, regulatory mechanisms like the FDA’s 505(b)(2) application pathway facilitate the development of innovative formulations (such as oral liquid and ER formulations) from already available compounds (Source: U.S. Food and Drug Administration). Such innovations improve patient satisfaction and open up new areas of application, leading to increased commercialization of the product despite the matured generics market environment.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In November 2024, Kamat Pharmatech LLC filed a 505(b)(2) New Drug Application (NDA) with the U.S. FDA for Raldesy, a trazodone oral solution. The agency is currently examining the labeling and clinical data. This move signals a push to expand the trazodone market by offering options other than traditional tablets.
- In August 2024, Granules India obtained approval from the U.S. FDA under its Abbreviated New Drug Application (ANDA) for Trazodone Hydrochloride Tablets (50 mg to 300 mg) which is bioequivalent to the reference listed drug Desyrel.
Competitive Landscape
The trazodone market is characterized by intense competition as it is largely dominated by generics, widely used in medical practice, and faces considerable pressure in terms of prices in global pharma markets. Companies operating in this industry utilize their capacities for mass production, approvals, and reliable distribution channels to retain market share. Since the drug is already off-patent, the competition in the industry focuses mainly on cost-effectiveness and diversification. Key focus areas include:
- Expansion of generic drug portfolios
- Focus on cost leadership and large-scale manufacturing efficiencies
- Regulatory approvals (ANDA filings)
- Strategic partnerships and distribution agreements
Market Report Scope
Trazodone Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 687.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.1% | 2033 Value Projection: | USD 907.4 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Teva Pharmaceutical Industries Ltd., Viatris Inc., Sandoz, Pfizer Inc., Sun Pharmaceutical Industries Ltd., Angelini Pharma S.p.A., The Piramal Group, Fermion Oy, Apotex Inc., and Zydus Lifesciences Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- The future outlook of the trazodone market is likely to be stable with a moderate growth, mainly due to the persistent global prevalence of depression, anxiety, and insomnia conditions. Although there will be continued development of other antidepressants in the coming years, the future of trazodone should not be ignored owing to the drug’s dual application as an antidepressant and sleep inducer. Other factors that are set to fuel growth in the market include the increasing use of generics and the utilization of trazodone for indications other than its label claim.
- Maximum opportunities are anticipated for the sleep disorders and depression management application in the U.S. market due to the high rate of diagnoses, prescriptions, and off-label usage of trazodone for treating insomnia. Besides that, new market opportunities will also be provided by markets like India and Brazil because of growing awareness regarding mental health and better health care facilities.
- In order to create a competitive advantage, firms need to concentrate on the generic production approach of cost leadership, expansion in emerging markets that have high growth potential, and differentiation by formulating better versions such as extended-release products. Companies need to collaborate strategically with healthcare professionals, and conduct research in real world evidence and pharmacogenomics in order to position their products better. In addition, regulatory affairs and pharmacovigilance systems should be strengthened for greater efficacy.
Market Segmentation
- Product Type Insights (Revenue, USD Mn, 2021 - 2033)
- Branded
- Generics
- Form Insights (Revenue, USD Mn, 2021 - 2033)
- Immediate Release Tablets
- Extended-Release Oral Tablets
- Strength Insights (Revenue, USD Mn, 2021 - 2033)
- 50 mg
- 100 mg
- 150 mg
- 300 mg
- Indication Insights (Revenue, USD Mn, 2021 - 2033)
- Depression
- Anxiety
- Insomnia
- Others
- Distribution Channel Insights (Revenue, USD Mn, 2021 - 2033)
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Teva Pharmaceutical Industries Ltd.
- Viatris Inc.
- Sandoz
- Pfizer Inc.
- Sun Pharmaceutical Industries Ltd.
- Angelini Pharma S.p.A.
- The Piramal Group
- Fermion Oy
- Apotex Inc.
- Zydus Lifesciences Ltd.
Sources
Primary Research Interviews
- Trazodone manufacturers and generic drug producers
- Pharmaceutical distributors and hospital pharmacy managers
- Psychiatrists and neurologists prescribing antidepressants
- Clinical researchers specializing in depression and sleep disorders
Stakeholders
- Providers of Trazodone Solutions
- End-use Sectors
- Hospitals
- Specialty Clinics (Psychiatry & Neurology)
- Retail Pharmacies
- Online Pharmacies
- Regulatory & Health Bodies: U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), Central Drugs Standard Control Organization (CDSCO), World Health Organization (WHO)
Databases
- U.S. Food and Drug Administration (FDA) – Drug approvals & Orange Book database
- World Health Organization (WHO) – Global Health Observatory (GHO)
- OECD Health Statistics – Mental health and pharmaceutical consumption data
- National Institutes of Health (NIH) – Clinical trials and research database
Magazines
- Pharmaceutical Technology – Antidepressant drug development trends
- BioPharma Dive – CNS drug market developments
- Drug Discovery & Development – Advances in psychiatric medications
- Healthcare Business Today – Mental health treatment market trends
Journals
- The Lancet Psychiatry – Depression and antidepressant efficacy studies
- Journal of Clinical Psychiatry – Trazodone clinical usage and outcomes
- CNS Drugs – Pharmacological profiles of antidepressants
- BMJ (British Medical Journal) – Mental health treatment studies
Newspapers
- The Wall Street Journal – Pharmaceutical industry and generics market trends
- Financial Times – Global pharma market and patent expiry analysis
- Business Standard – India generics and antidepressant market insights
- The Economic Times – Pharmaceutical sector developments
Associations
- American Psychiatric Association (APA)
- National Institute of Mental Health (NIMH)
- World Federation for Mental Health (WFMH)
- European Psychiatric Association (EPA)
Public Domain Sources
- World Health Organization – Mental health and depression statistics
- U.S. National Institutes of Health (NIH) – Depression and treatment research
- U.S. FDA – Drug safety and labeling information for trazodone
- European Medicines Agency (EMA) – Drug approvals and pharmacovigilance
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients