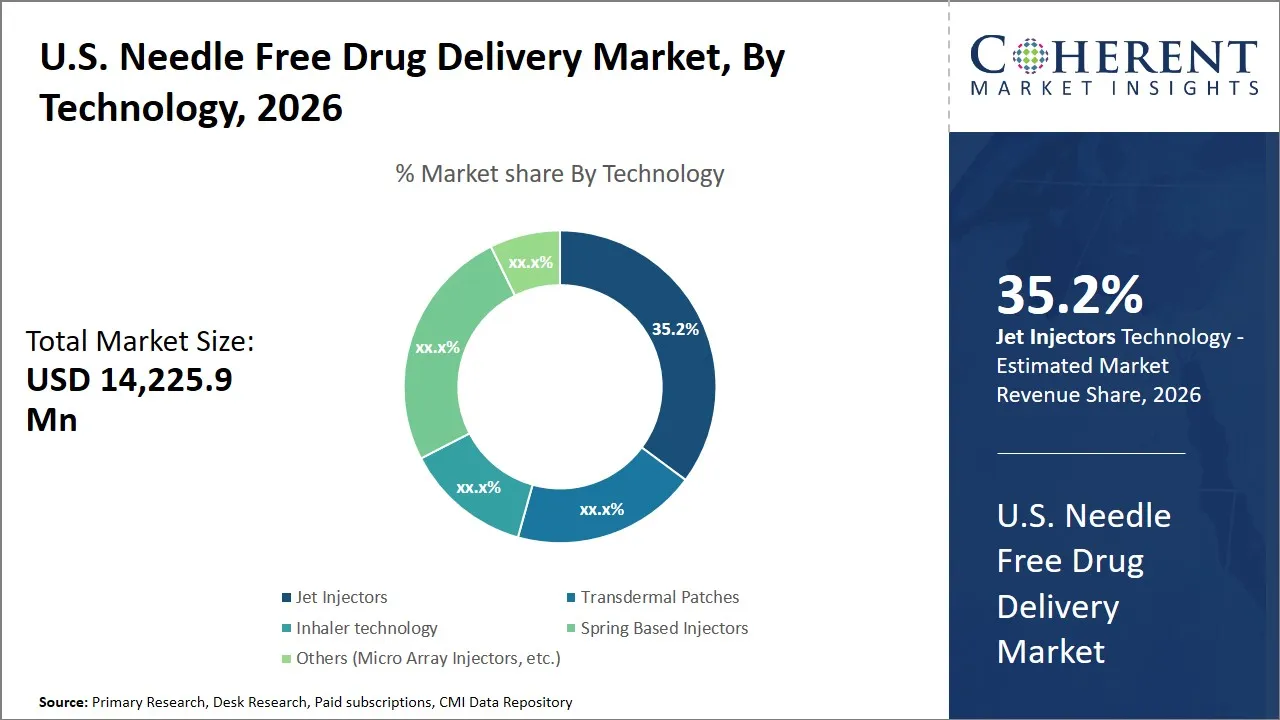
U.S. Needle Free Drug Delivery Market Size and Forecast – 2026-2033
The U.S. needle free drug delivery market is estimated to be valued at USD 14,225.9 Mn in 2026 and is expected to reach USD 29,123.4 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 15.3% from 2026 to 2033. This significant expansion highlights the increasing adoption of needle-free technologies driven by advancements in drug delivery systems and rising demand for safer, non-invasive treatment options across various medical applications.
Key Takeaways of the U.S. Needle Free Drug Delivery Market
- Jet injectors segment is expected to lead the U.S. needle free drug delivery market, capturing 35.2% share in 2026.
- Insulin delivery segment is estimated to represent 28.7% of the U.S. needle free drug delivery market share in 2026.
- Hospitals and clinics segment is projected to dominate with 46.0% of the U.S. needle free drug delivery market share in 2026.
Market Overview
- U.S. needle free drug delivery market is expanding steadily due to increasing demand for painless and safer drug administration technologies across hospitals, vaccination programs, and home care settings.
- The market is driven by rising adoption of biologics and injectable therapies that require patient friendly and self-administration compatible delivery systems.
- It is supported by strong vaccination infrastructure and growing focus on rapid and large-scale immunization programs across the country.
- U.S. needle free drug delivery market is benefiting from technological advancements in jet injectors, microneedle patches, and transdermal delivery platforms improving accuracy and usability.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Expansion of self-injection biologics in the U.S. |
|
|
Strong pharmacy led vaccination activity across the U.S. |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Jet Injectors Segment Dominate the U.S. Needle Free Drug Delivery Market in 2026?
The jet injectors segment is expected to hold the largest market share of 35.2% in 2026, owing to their efficiency and patient-friendly design. The needle free drug delivery market in the U.S has many forms of technology, but jet injectors occupy the lion share mainly because they are effective in the delivery of medication without the use of needles. It is a high-pressure narrow jet technology of the drug formulation to pierce the skin and deliver the drug to the subcutaneous tissue or muscle. The removal of needle-related anxiety and pain is one of the essential drivers in this case and is a major obstacle to treatment adherence in patients who need to receive injections regularly e.g. patients with chronic illnesses. Jet injectors are, therefore, a more comfortable and less frightening alternative, which promotes regular medication adherence.
Moreover, the jet injectors provide fast and efficient delivery of drugs so that therapeutic doses deliver fast and uniformly. This especially proves beneficial in emergency situations or mass immunization where speed and efficiency are of the greatest importance. The safety profile of the jet injectors has also been enhanced by the technological advancements that reduce the risks of cross-contamination using disposable or waste parts, which enhances the confidence of both the healthcare providers and patients.
For instance, on January 9, 2026, PharmaJet announced that its Stratis needle-free injection system is being used to administer DNA immunotherapy for advanced melanoma in a late-stage clinical development program following positive Phase 2 data. The collaboration supports clinical preparations for a registrational trial and demonstrates the growing use of needle-free jet injector technology in oncology and advanced biologic therapies.
(Source- PharmaJet)
Why Does the Insulin Delivery Segment Dominate the U.S. Needle Free Drug Delivery Market in 2026?
The insulin delivery segment is expected to hold 28.7% of the market share in 2026, driven by rising diabetes prevalence and preference for painless therapeutic options. Insulin delivery is the most popular application segment in the U.S. needle free drug delivery market because the burden of diabetes is increasingly growing in the country and the difficulties related to the use of traditional techniques of insulin injection. The patients of diabetes are usually bound with the need of insulin administration that is usually several times a day and needle phobia as well as injection site pain can be major barriers that may raise compliance. Needle-free insulin delivery technologies, thus, fill a gap in critical need that has not been well met as a less invasive alternative and more patient-centered one.
Also, needle-free systems, like jet injectors and transdermal patches offer fast absorption and improved glucose regulation, improving efficacy of treatment overall. This is most useful in the case of type 1 and insulin dependent type 2 diabetes patients who need to be offered a better control of the sugar levels in their bodies without the pain associated with the needles. Both clinical and home setting needle-free insulin delivery is further convenient to enhance adherence and quality of life.
For instance, on February 9, 2026, MannKind Corporation announced the first patient enrollment in its INHALE-1ST pediatric study evaluating Afrezza, an inhaled mealtime insulin, in youth with newly diagnosed type 1 diabetes. The update is significant because Afrezza is a needle-free insulin delivery option, and the study expands its potential use in younger patients, a segment where easier and less invasive administration can improve treatment acceptance and adherence.
(Source- MannKind Corporation)
The Hospitals and Clinics Segment Dominates the U.S. Needle Free Drug Delivery Market
The hospitals and clinics segment is expected to hold the largest market share of 46.0% in 2026, due to their frontline role in administering vaccines, insulin, and other injectable therapies where safety, efficiency, and patient experience are critical factors. The system of needle-free delivery is gaining popularity in such environments due to the fact that it drastically minimizes needle-stick injuries in the healthcare professionals, which is a significant work-related risk. The adoption is encouraged in clinical settings due to the improved safety measures and regulatory focus on preventing such injuries.
Additionally, hospitals and clinics have to deal with a wide range of patients, such as those with chronic conditions or undergoing vaccination, in which case needle-free options will decrease anxiety and enhance comfort of patients during drug delivery. This does not only ensure compliance rates are high, but it also makes operations more efficient since training becomes easier and sharps disposal processes become unnecessary.
U.S. Vaccination Infrastructure Supporting Needle-Free Drug Delivery Adoption
|
Infrastructure metric |
U.S. vaccination infrastructure snapshot |
Relevance to U.S. needle free drug delivery market |
|
Annual vaccination doses administered / distributed |
The U.S. vaccination system handles very large annual volumes. For seasonal influenza alone, CDC reported about 147.6 million doses distributed in the 2024–25 season as of March 8, 2025, and manufacturers projected up to 154 million doses for 2025–26. In addition, the U.S. administered 59.8 million COVID-19 bivalent doses from September 1, 2022 to September 30, 2023. |
High adult and pediatric vaccination throughput support demand for faster, easier, and lower-anxiety delivery platforms such as needle-free injectors, especially in mass seasonal immunization settings. |
|
Number of immunization clinics and vaccination centers |
The U.S. has a broad immunization network rather than a single centralized clinic count. CDC states there are over 37,000 Vaccines for Children (VFC) providers nationwide. CDC pharmacy-based vaccination dashboards also note the U.S. had an estimated 57,000 retail pharmacies in 2022, many of which are major vaccination access points. |
A large decentralized provider base increases the commercial viability of easy-to-administer, low-training-burden needle-free systems across clinics, pediatric practices, and pharmacies. |
|
Government-supported vaccination site footprint |
A 2024 CDC review of the COVID-19 vaccine rollout reported that by the end of the federal public health emergency, roughly 125,000 jurisdiction provider sites, 54,000 individual pharmacy sites, and 5,600 dialysis centers had been enrolled in the U.S. COVID-19 vaccine program, with vaccine shipped to more than 107,500 unique locations. |
This proves the U.S. can operationalize vaccine delivery at national scale, which is favorable for adoption of alternative delivery technologies that improve speed, safety, and patient acceptance. |
|
Government mass vaccination program capacity |
FEMA’s federally supported Community Vaccination Center model defined Type 1 centers at ~6,000 vaccinations/day and Type 2 centers at ~3,000 vaccinations/day. CDC also noted that during December 11, 2020 to March 29, 2022, the U.S. government delivered about 700 million COVID-19 vaccine doses to vaccination sites. |
Large-throughput public vaccination capacity strengthens the case for needle-free devices in surge campaigns, workplace programs, school vaccination drives, and emergency preparedness scenarios. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
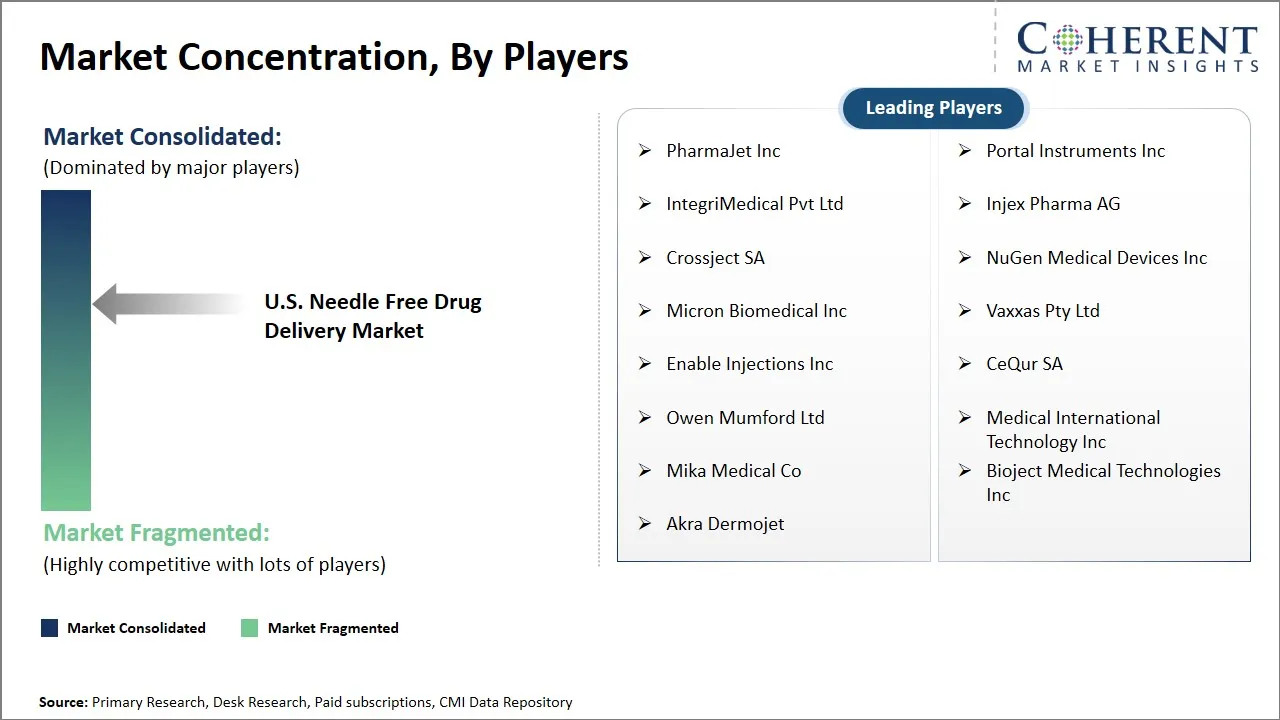
Market Players, Key Developments, and Competitive Intelligence
To learn more about this report, Request Free Sample
Key Developments
- In October 2025, PharmaJet announced plans to develop a new portfolio of proprietary needle-free self-injection pens designed for home use. The devices aim to allow patients to administer therapies without needles, supporting the growing trend toward self-administration and patient-centric drug delivery in the U.S. healthcare system.
- In May 2025, Crossject reported performance milestones for its ZENEO needle-free auto-injector, demonstrating that the system can consistently achieve injection depths comparable to traditional intramuscular injections. The technology is being developed for emergency therapies and other drug delivery applications, strengthening the role of needle-free devices in modern pharmaceutical treatment.
- In January 2025, Portal Instruments launched the PRIME Nexus reusable electronic injector, an advanced needle-free drug delivery platform designed to improve precision and sustainability in injectable therapies. The device is intended to enhance patient comfort and enable pharmaceutical partners to deliver complex biologic drugs without traditional needles.
Expanding U.S. Biologic Pipeline Driving Demand for Needle Free Drug Delivery Systems
- The U.S. biologic drug pipeline is becoming deeper and more complicated, and this is what makes the argument on why sophisticated delivery technologies are strong in the U.S. needle free drug delivery market. According to U.S. FDA, in 2025, CDER approved 46 novel drugs, and CBER also approved new biologics and biologic license applications in vaccines and cell and gene therapies among other specialized products. This is important since a high number of biologics need specificity, good compliance in patients with their repeated administration and therefore delivery mode is a commercial and clinical differentiator and not an afterthought.
- Simultaneously, the U.S. biologics long runway experiencing expansion increases the addressable requirement of next generation administration platforms. This gap indicates the coming wave of biologic launches and follow-on competition is large, with companies insisting on making their products more convenient, requiring less pain, and allowing self-administration, such as needle-free systems, to achieve better adherence, product differentiation, and use in non-infusion primary care.
Top Strategies Followed by U.S. Needle Free Drug Delivery Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Leading players maintain their position through strong R&D investment, continuous device improvement, and partnerships with pharmaceutical companies to strengthen product integration and market reach. They also expand geographically and scale distribution networks to capture broader demand and reinforce brand strength. |
PharmaJet is a strong example, as it has expanded its needle-free platform through vaccine delivery partnerships, clinical collaborations, and broader use cases beyond traditional immunization. |
|
Mid-Level Players |
Mid-level players compete by offering more affordable needle free delivery solutions while focusing on practical use cases such as outpatient care, community clinics, and self-administration. Instead of matching the scale of larger companies, they rely on partnerships, distribution tie ups, and targeted product expansion to improve market reach and keep development costs under control. |
NuGen Medical Devices is a relevant example. The company expanded InsuJet distribution in Canada in April 2025 and also entered a license, supply, and distribution agreement with EziAutoJector in September 2024 to market its needle free technology for insulin and other subcutaneous biosimilar protein products, showing a partnership led and value driven growth model. |
|
Small-Scale Players |
Small scale players compete by focusing on niche technologies, targeted patient groups, and specialized delivery formats rather than broad market coverage. They usually differentiate through innovation in areas such as microneedle patches, precision dosing, or digitally enabled delivery systems, while using local partnerships and pilot collaborations to build early traction. |
Innoture is a relevant example, as it is developing microneedle-based vaccine delivery platforms that target painless, easy to use administration and reflect how smaller innovators are carving out space through highly specialized technology rather than scale. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
U.S. Needle Free Drug Delivery Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 14,225.9 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 15.3% | 2033 Value Projection: | USD 29,123.4 Mn |
| Segments covered: |
|
||
| Companies covered: |
PharmaJet Inc, Portal Instruments Inc, IntegriMedical Pvt Ltd, Injex Pharma AG, Crossject SA, NuGen Medical Devices Inc, Micron Biomedical Inc, Vaxxas Pty Ltd, Enable Injections Inc, CeQur SA, Owen Mumford Ltd, Medical International Technology Inc, Mika Medical Co, Bioject Medical Technologies Inc, and Akra Dermojet |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
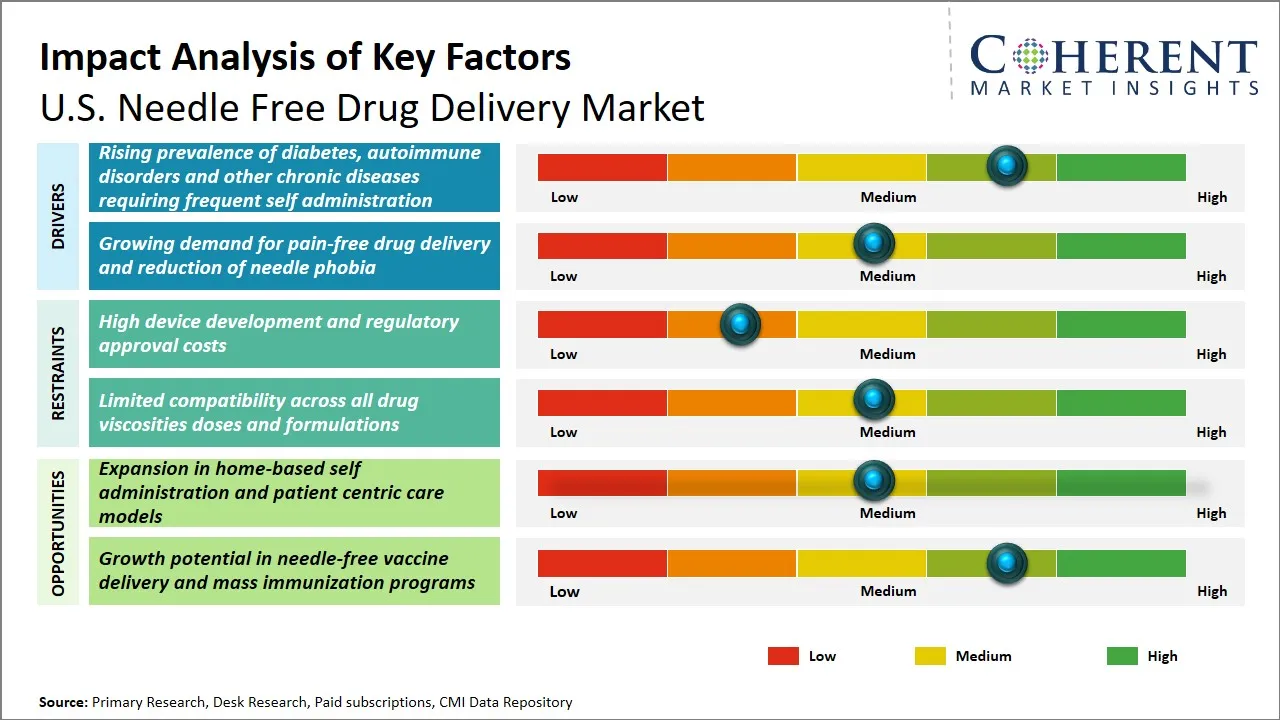
U.S. Needle Free Drug Delivery Market Dynamics
To learn more about this report, Request Free Sample
U.S. Needle Free Drug Delivery Market Driver - Increasing Prevalence of Chronic Diseases Driving Demand for Needle Free Drug Delivery
The growing trends of diabetes, autoimmune diseases and other chronic diseases requiring frequent medication intake by the patient is also a notable force behind the need to support the U.S. needle free drug delivery market. The patients who have to deal with such conditions as diabetes usually have to inject themselves several times a day, which is not only painful but also causes needle-related infections and complications. In the same regard, patients living with autoimmune conditions are often under the care of biologic treatments, which typically incorporate needle delivery mechanisms which results in low patient compliance as a result of needle phobia and reactions to the injections. The lack of needles provides a safer and more painless way of delivering drugs in case of the needle-free drug delivery system, which also increase compliance with the therapy, as well as making the whole therapeutic experience much better.
For instance, on January 26, 2026, MannKind Corporation announced that in the first quarter of 2026, the U.S. Food and Drug Administration approved an updated Afrezza label that provides starting dose guidance for patients switching from multiple daily insulin injections or insulin pump mealtime therapy. The company also stated that the American Diabetes Association Standards of Care in Diabetes 2026 now recommends that clinicians evaluate inhaled insulin as a prandial option at every patient visit, strengthening the role of needle-free insulin delivery in routine diabetes management.
(Source- MannKind Corporation)
U.S. Needle Free Drug Delivery Market Opportunity - Expansion in Home-Based Self Administration and Patient Centric Care Models
The needle free drug delivery industry in the U.S. has a promising future due to the growing tendency of home-based self-administration and patient-centered care model. The growing patient desire to have convenience and control over the management of chronic diseases has boosted the need to find a solution to this by developing a needle-free delivery system that allows self-administration of medications safely, easily, and painlessly, outside of the clinical environment. This change is in line with the increased interest of healthcare providers in patient empowerment, adherence and the quality of life. The use of innovations like jet injectors, nasal sprays, and transdermal patches provide a possible alternative to the needle injection in order to decrease the number of needle-stick injuries and needle phobia, thereby improving patient adherence.
For instance, in July 2025, Becton, Dickinson and Company announced the initiation of the first pharmaceutical-sponsored clinical trial using its BD Libertas wearable injector technology for the delivery of biologic medicines. The system is designed as a prefilled, patient-ready device that enables subcutaneous administration outside clinical settings, supporting the growing shift toward home-based treatment and self-administration.
(Source- Becton, Dickinson and Company)
Analyst Opinion (Expert Opinion)
- The needle free drug delivery market in the U.S. has shifted toward a niche to a practical usage due to the presence of biologic drugs, the trend of self-administration, and the preference of patients to less painful delivery. It is being aided by increasingly effective injector technologies, an increase in interest in vaccination and chronic disease use cases, and government sponsored innovations like the interest of Biomedical Advanced Research and Development Authority (BARDA) in needle free vaccine delivery. The market opportunity is good with scale up yet to be met by addressing cost, reimbursement, and provider workflow barriers.
- The industry conferences like PODD 2024 and 2025, the Pre-Filled Syringes and Injectable Drug Devices conference and PDA workshops have served to drive the market forward by putting pharma, device companies and regulators in the same discussion concerning commercialization, human factors, and combination product strategy. These are platforms that are contributing to the realization of technical promise in real market traction hence the future of the U.S. needle free drug delivery market is encouraging.
Market Segmentation
- Technology Insights (Revenue, USD Mn, 2021 - 2033)
- Jet Injectors
- Transdermal Patches
- Inhaler technology
- Spring Based Injectors
- Others (Micro Array Injectors, etc.)
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Insulin Delivery
- Vaccine Delivery
- Pain Management
- Hormone Delivery
- Opioid Dependence
- Others (In-Vitro Fertilization, Smoking cessation, etc.)
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospitals and Clinics
- Homecare Settings
- Pharmaceutical and Biotechnology Companies
- Others (Research Institutes, etc.)
- Key Players Insights
- PharmaJet Inc
- Portal Instruments Inc
- IntegriMedical Pvt Ltd
- Injex Pharma AG
- Crossject SA
- NuGen Medical Devices Inc
- Micron Biomedical Inc
- Vaxxas Pty Ltd
- Enable Injections Inc
- CeQur SA
- Owen Mumford Ltd
- Medical International Technology Inc
- Mika Medical Co
- Bioject Medical Technologies Inc
- Akra Dermojet
Source
Primary Research Interviews
Industry Stakeholders list
- Hospital pharmacy directors
- Outpatient infusion center managers
- Vaccine program managers
- Drug device combination product regulatory specialists
- Biologics manufacturing and fill finish experts
- Payers and market access directors
End-users list
- Immunization nurses
- Retail pharmacists
- Home healthcare nurses
- Endocrinologists
- Rheumatologists
- Patients using self-injectable biologics
Government and International Databases
- U.S. Food and Drug Administration
- Centers for Disease Control and Prevention
- Centers for Medicare & Medicaid Services Data
- ClinicalTrials.gov
- World Health Organization Data
- OECD Health Statistics
Trade Publications
- BioPharma Dive
- PharmTech
- Pharmaceutical Technology
- Endpoints News
- Drug Target Review
- Innovations in Pharmaceutical Technology
Academic Journals
- Journal of Controlled Release
- Drug Delivery and Translational Research
- AAPS PharmSciTech
- The AAPS Journal
- International Journal of Pharmaceutics
- Pharmaceutical Research
Reputable Newspapers
- Financial Times
- The Wall Street Journal
- The New York Times
- The Washington Post
Industry Associations
- Controlled Release Society
- American Association of Pharmaceutical Scientists
- Parenteral Drug Association
- Biotechnology Innovation Organization
- International Society for Pharmaceutical Engineering
- Drug Information Association
Public Domain Resources
- DailyMed
- OpenFDA
- CDC WONDER
- Federal Register
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 8 Years
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients