U.S. Perinatal Tissue-derived Allograft Market Size and Forecast – 2026-2033
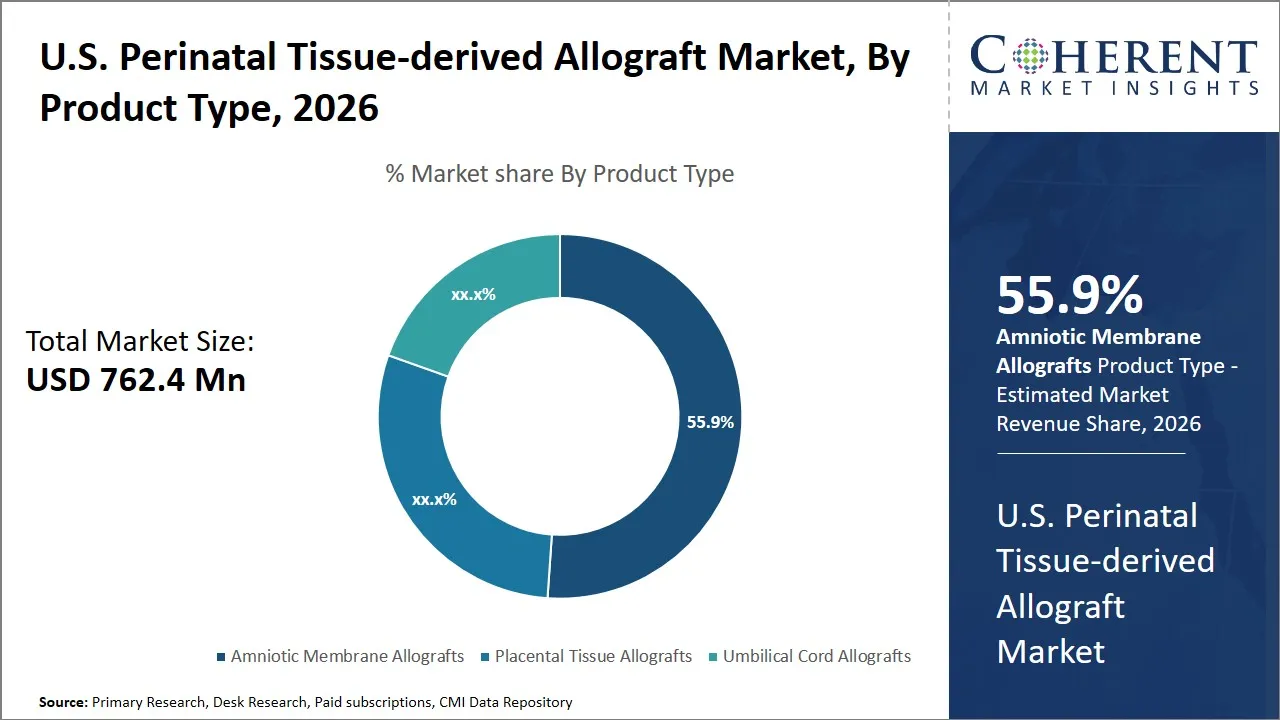
The U.S. perinatal tissue-derived allograft market is estimated to be valued at USD 762.4 Mn in 2026 and is expected to reach USD 1,226.3 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 9.8% from 2026 to 2033. This growth reflects increasing adoption of regenerative medicine and advanced tissue therapies across various medical fields, supported by ongoing research and favorable reimbursement policies.
Key Takeaways of the U.S. Perinatal Tissue-derived Allograft Market
- Amniotic membrane allografts segment is expected to lead the U.S. perinatal tissue-derived allograft market, capturing 55.9% share in 2026.
- Single layer segment is estimated to represent 54.8% of the U.S. perinatal tissue-derived allograft market share in 2026.
- Wound care segment is projected to dominate with 31.0% of the U.S. perinatal tissue-derived allograft market share in 2026.
Market Overview
- The U.S. perinatal tissue-derived allograft market is centered on placental, amniotic, and related birth tissue products used mainly in wound care, surgery, and ophthalmology, with the category steadily moving beyond standard chronic wound treatment into more specialized clinical settings.
- Beyond wound care, the market is expanding into orthopedic and sports medicine applications, where placental and amniotic tissue allografts are being explored for tendon, ligament, cartilage, and osteoarthritis-related uses.
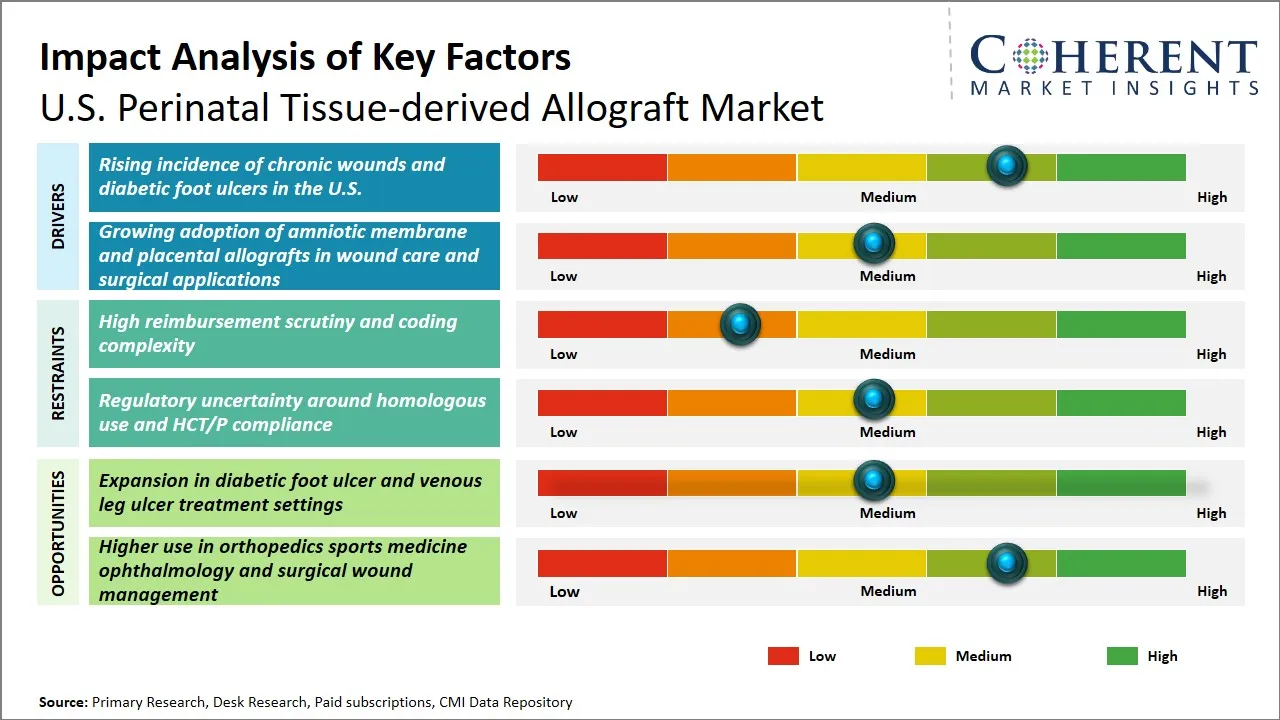
- Regulatory scrutiny remains a defining market factor in the U.S., as these products are assessed under the Food and Drug Administration’s Human Cells, Tissues, and Cellular and Tissue-Based Products framework, with ongoing focus on minimal manipulation and homologous use.
- Shelf stable and workflow friendly formats are gaining importance because hospitals and outpatient centers want grafts that are easier to store, faster to apply, and better aligned with operating room efficiency. MIMEDX said its thyroid product is shelf stable for up to five years and designed for easier application.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Medicare reimbursement reset for skin substitute products |
|
|
Product innovation moving beyond standard wound care |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Amniotic Membrane Allografts Segment Dominate the U.S. Perinatal Tissue-derived Allograft Market in 2026?
The amniotic membrane allografts segment is expected to hold the largest market share of 55.9% in 2026, driven by their unique biological properties and versatile clinical applications. The allografts of amniotic membrane have the inherent property of anti-inflammatory, anti-fibrotic, and anti-microbial, which promotes rapid wound healing and tissue regeneration. These allografts are rich in collagen and growth factors that forms an extracellular matrix that promote cellular migration as well as proliferation that is essential in repairing damaged tissues.
Moreover, they are naturally biocompatible, which reduces chances of immune rejection and inflammation making them desirable in implantation in different surgical settings. Their ease of processing and sterilization with their capability to be held without losing biological activity also adds to their attractiveness to healthcare providers. The fact that amniotic membrane allografts are used in so many specialties speaks to that fact, including wound care, reconstructive surgery, and ophthalmology, among others, which guarantees continued demand.
For instance, in June 2024, Axogen announced the launch of Avive+ Soft Tissue Matrix, a resorbable multi-layer amniotic membrane allograft developed as a soft tissue barrier for peripheral nerve healing and surgical applications. The launch strengthened Axogen’s regenerative medicine portfolio and highlighted continued innovation in amniotic membrane allografts beyond traditional wound care use cases.
(Sources - Axogen)
Why Does the Single Layer Segment Dominate the U.S. Perinatal Tissue-derived Allograft Market in 2026?
The single layer segment is expected to hold 54.8% of the market share in 2026, owing to the clinical advantages single layer allografts offer in terms of ease of handling, surgical application, and targeted tissue repair outcomes. Single layer membranes are thinner and more flexible and the surgeons can be able to exactly fit the allograft to odd or complicated wound locations without rejecting the structural integrity or biological activity.
This aspect is especially advantageous in the use of single layer allografts where delicate surgeries like an ophthalmic surgery are required to ensure the patient does not lose his sight but rather he is healed through the use of a thin and clear membrane. The decreased thickness is also helpful in the exchange of oxygen and nutrients which are important considerations that enhance the best cell viability and assimilation with the native tissues. Operationally, the single layer allografts save time and complexity in the operating room and boost the efficiency of the procedure.
For instance, in April 2024, Xtant Medical announced the full commercial launch of SimpliGraft, a single-layer amniotic membrane allograft for chronic and acute wounds in the U.S. The product was introduced alongside SimpliMax and was designed as a dehydrated, terminally irradiated sheet that serves as a protective barrier over wounds. This launch is relevant because it directly supports demand in the single-layer segment, which is favored for easier handling and broader wound care use.
(Source - Xtant Medical)
The Wound Care Segment Dominates the U.S. Perinatal Tissue-derived Allograft Market
The wound care segment is expected to hold the largest market share of 31.0% in 2026, reflecting the critical need for effective solutions in the management of acute and chronic wounds. Due to the rising incidence of diabetes, obesity, and peripheral vascular disease, the number of chronic non-healing wounds, including diabetic foot ulcers and pressure sores among others, has risen dramatically. These circumstances are highly challenging to treat and have an impact on healthcare systems that are associated with high morbidity and costs.
Perinatal tissue allografts especially amniotic membranes and umbilical cord tissues have highly biological activity which enhances faster and complete wound healing when compared with conventional dressings. They have anti-inflammatory and regenerative capabilities to decrease the risk of infection, tune the microenvironment of the wound, and promote neovascularization, which is essential in healing chronic wounds. Besides, the capability of using these allografts as bio-compatible dressing or graft will enable least invasive and repeatable interventions, which enhance patient compliance and comfort.
Impact of Medicare Reimbursement per Application on the U.S. Perinatal Tissue-derived Allograft Market
- The issue of Medicare reimbursement per application has become a significant factor on the U.S. perinatal tissue-derived allograft market as payment policy is now more directly influencing product economics and physician uptake. In the case of 2026, CMS has finalized the skin substitute products would be paid as incident to supplies under the Physician Fee Schedule used in a non-facility setting, whereas hospital outpatient payment was updated under new payment groupings of skin substitute products. This is important in that reimbursement is not only about clinical value, but it now directly influences product access, site of care economics, and willingness of provider to use more expensive grafts.
- Medicare at the claim level also closely correlates payment with the amount actually paid as opposed to waste, where this puts strain on the utilization management and documentation. According to CMS billing guidelines, the amount actually used on the patient is the only amount which should be reimbursed at the per square centimeter rate, and the rest of the unused or discarded amounts are not paid separately. Practically, it compels manufacturers and providers to emphasize graft sizing, pricing discipline as well as product formats that are more suitable to wound sizes and minimize unreimbursed wastage.
Market Players, Key Developments, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- On March 5, 2026, MIMEDX announced the commercial launch of AMNIOFIX Thyroid Shields, a line extension of its dehydrated human amnion/chorion membrane allograft portfolio for endocrine surgery. The preconfigured grafts are applied during thyroidectomy to protect the recurrent laryngeal nerve and parathyroid glands, supporting healing and reducing surgical complications. The product is shelf-stable and designed to improve operating room efficiency and expand surgical use cases of placental allografts.
- In December 2025, BioStem Technologies introduced its American Amnion product line at the 2025 Desert Foot Multi-Disciplinary Limb Salvage and Wound Care Conference in Phoenix. The launch included American Amnion AC and American Amnion, two placental-derived allografts for wound care and protective covering applications, expanding the company’s U.S. perinatal tissue allograft portfolio.
- In November 2025, Cellution Biologics introduced AmnioPlast 1 and AmnioPlast Double, dehydrated amnion membrane allografts for ocular surface repair. The products are designed as protective barriers after ocular reconstruction procedures and expand the company’s U.S. regenerative ophthalmology portfolio.
Regulatory Scenario of the U.S. Perinatal Tissue-derived Allograft Market
|
Regulatory area |
Current scenario in the U.S. Perinatal Tissue-derived Allograft Market |
Market impact |
|
FDA product classification |
Perinatal tissue-derived allografts are generally regulated as human cells, tissues, and cellular and tissue-based products if they meet the criteria under Section 361 of the Public Health Service Act and 21 CFR Part 1271. The main regulatory tests remain minimal manipulation and homologous use. Products that do not meet these standards can be regulated as drugs, devices, and or biologics under Section 351, which carries a much higher approval burden. |
This creates a split market where compliant 361 products can move faster commercially, while higher-risk claims or processing methods can trigger stricter regulation and slow expansion. |
|
FDA enforcement environment |
FDA scrutiny remains active in this category. Recent warning letters in 2025 cited companies marketing placental or amniotic products as unapproved drugs or unlicensed biologics, showing that intended use claims, processing methods, and promotional language are still under close review. |
Regulatory enforcement raises compliance costs and increases legal and commercialization risk for manufacturers that stretch beyond permitted HCT/P positioning. |
|
Reimbursement policy |
CMS finalized major 2026 payment changes for skin substitute products under the Medicare Physician Fee Schedule. For 2026, CMS adopted a single national per square centimeter payment rate of about USD 127.28 for relevant products, while noting that some Section 351 biologics will continue under separate methodology. |
Reimbursement is now a major pricing and utilization lever, directly affecting physician economics, product selection, and margin pressure in wound care settings. |
|
Coverage policy |
CMS announced final Local Coverage Determinations for certain skin substitutes in late 2025, but those LCDs scheduled for January 1, 2026, were then withdrawn, meaning the payment rule stayed in place while broad national coverage tightening did not move forward as initially expected. Active local policies still remain with some Medicare Administrative Contractors. |
This has created a mixed landscape with continued reimbursement pressure but less immediate national coverage restriction than many suppliers had anticipated. |
|
Billing and utilization controls |
Medicare billing guidance links payment to the amount actually applied and requires strong documentation around medical necessity, coding, and product use. The 2026 reimbursement reset also puts more focus on square-centimeter efficiency and wastage control. |
Manufacturers are pushed to optimize graft sizing, packaging, and pricing, while providers face tighter documentation and utilization review. |
|
Clinical evidence expectations |
Even where premarket approval is not required for 361 HCT/Ps, payers and providers increasingly expect stronger evidence for wound healing outcomes, surgical utility, and cost justification. Clinical trial activity and evidence generation continue to support market positioning. |
Companies with stronger published data and clearer indication-specific value are better positioned for formulary access and sustained adoption. |
|
Core regulatory challenge |
The biggest challenge is that the line between compliant tissue allograft use and a more heavily regulated biologic product remains commercially critical. FDA guidance continues to emphasize that broad disease-treatment claims and non-homologous positioning can shift regulatory status. |
This limits aggressive marketing claims and keeps regulatory strategy central to product development, labeling, and go-to-market planning. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Top Strategies Followed by U.S. Perinatal Tissue-derived Allograft Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Leading companies stay ahead by investing in R&D to develop more effective, safer, and longer shelf-life allograft products. They also use partnerships with healthcare providers and industry players to expand reach, speed up commercialization, and strengthen customer retention. At the same time, they grow through wider distribution and entry into new markets with products tailored to local clinical and regulatory needs. |
BioStem Technologies strengthens its leadership through placental allograft product innovation, differentiated processing technology, and an exclusive distribution partnership with Venture Medical to expand commercialization and market reach in the U.S. |
|
Mid-Level Players |
Mid-level players compete by offering clinically reliable allografts at more affordable price points, making them attractive to smaller hospitals, outpatient centers, and price sensitive buyers. They often strengthen their position through partnerships that improve manufacturing scale, technology access, and distribution efficiency, while also focusing on flexible production to respond faster to market demand. |
Gland Pharma represents a strong mid-level player approach. The company focuses on cost-efficient sterile injectable manufacturing and supplies a broad portfolio of generic injectables to the U.S. hospital market at competitive pricing. It has strengthened its position through strategic partnerships with global pharma companies for technology transfer and commercialization, while leveraging large-scale, flexible manufacturing facilities in India to ensure consistent supply and faster response to demand fluctuations. |
|
Small-Scale Players |
Small scale players compete by targeting niche clinical uses and offering specialized allograft formats that larger companies may not prioritize. Their advantage is speed, since they can move faster on product design, localized partnerships, and focused physician outreach in areas such as wound care, ophthalmology, or orthopedic support. |
Stimlabs focuses on specialized placental allograft formats such as Revita, a full-thickness placental allograft positioned for chronic wounds and surgical wound applications, and markets it across focused specialties including podiatry, vascular surgery, dermatology, and orthopedic surgery. That is the kind of niche-led strategy smaller players use to compete without going head-to-head with broad portfolio companies. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
U.S. Perinatal Tissue-derived Allograft Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 762.4 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.8% | 2033 Value Projection: | USD 1,226.3 Mn |
| Segments covered: |
|
||
| Companies covered: |
MIMEDX Group, Inc., Organogenesis, Inc., Smith+Nephew plc, Amnio Technology, LLC, VIVEX Biologics, Inc., Integra LifeSciences Holdings Corporation, Celularity Inc., Applied Biologics LLC, Surgenex, LLC, Pinnacle Transplant Technologies, BioStem Technologies, Inc., StimLabs, LLC, BioTissue Holdings, Inc. / AMNIOX Medical, Inc., AlloSource, and MTF Biologics |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
U.S. Perinatal Tissue-derived Allograft Market Dynamics
To learn more about this report, Request Free Sample
U.S. Perinatal Tissue-derived Allograft Market Driver - Rising Incidence of Chronic Wounds and Diabetic Foot Ulcers in the U.S.
The growing number of chronic wounds, especially diabetic foot wound, in the U.S. contributes largely to the demand of perinatal tissue-derived allografts. Chronic wounds are a significant healthcare issue because they require a long duration to heal and could be highly infected with chances of serious complications such as amputation. Diabetic foot ulcers are one of the complications associated with diabetes that have increased with the increased number of diabetics, which leads to a high demand to find effective ways of treatment. Tissue-derived allografts that are perinatal with their regenerative effects and potential to promote tissue healing have become an exciting intervention technique to treat these complicated wounds. These allografts improve healing through the supply of important growth factors and extracellular matrix constituents, which generate cell proliferation and decrease inflammation.
For instance, in October 2025, MIMEDX announced the launch of EPIXPRESS, a next generation lyophilized human placental allograft for acute and chronic hard to heal wounds. The product features a thick tri layer configuration with fenestrations to improve fluid movement and is positioned for wound types such as diabetic foot ulcers and venous leg ulcers, as well as deep or tunneling wounds.
(Source - MIMEDX)
U.S. Perinatal Tissue-derived Allograft Market Opportunity - Expansion in Diabetic Foot Ulcer and Venous Leg Ulcer Treatment Settings
The U.S. market for perinatal tissue-derived allograft is an excellent growth opportunity due to the increased incidence of chronic wounds, specifically Diabetic Foot Ulcers (DFUs) and Venous Leg Ulcers (VLUs). The disorders are a significant clinical and financial burden to the health care system and diabetic foot ulcers are present in a population of diabetic patients approximately 15 percent of their lifetime and venous leg ulcers comprise about 70 percent of the chronic lower extremity ulcers. Tissue-derived allografts perinatal due to their special biological characteristics, including high growth factor concentration, extracellular matrix substances, and anti-inflammatory agents, have increased potential wound healing. This has resulted in an upsurge in its application in the management of wound care particularly in situations where there is little efficacy of traditional treatments.
For instance, in October 2024, BioStem Technologies announced the nationwide launch of Vendaje AC through Venture Medical after establishing reimbursement across all Medicare Administrative Contractor regions. The placental allograft product was positioned to expand access in advanced wound care, including treatment settings focused on diabetic foot ulcers and venous leg ulcers, making it a relevant launch tied to growing demand in chronic wound management.
(Source - BioStem Technologies)
Analyst Opinion (Expert Opinion)
- The U.S. perinatal tissue-derived allograft market is also picking up as hospitals and experts embrace the use of placental based grafts in caesural wound, surgery and ophthalmology. Targeted product innovation is supporting growth, and MIMEDX rolled out AMNIOFIX Thyroid Shields in March 2026 to support endocrine surgery and BioStem American Amnion in December 2025 to support advanced wound care. The main potential is in the specialty procedures and outpatient application, whereas the pressure of reimbursement and regulatory review continue to restrain quicker growth.
- Visibility in the industry has also enhanced by wound care forums and clinical meetings that enable the physicians to evaluate new evidence and product formats. The 2024 Symposium on Advanced Wound Care continued to be a key knowledge sharing event, and the 2026 acquisition of BioTissue Holdings by BioStem of the surgical and wound care business demonstrated the scale focus, portfolio diversity, and enhanced hospital accessibility of the market.
Market Segmentation
- Product Type Insights (Revenue, USD Mn, 2021 - 2033)
- Amniotic Membrane Allografts
- Placental Tissue Allografts
- Umbilical Cord Allografts
- Type Insights (Revenue, USD Mn, 2021 - 2033)
- Single Layer
- Multiple Layer
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Wound Care
- Orthopedics
- Ophthalmology
- Dental
- Reconstructive Surgery
- Dermatology
- Other Applications
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinics
- Others
- Key Players Insights
- MIMEDX Group, Inc.
- Organogenesis, Inc.
- Smith+Nephew plc
- Amnio Technology, LLC
- VIVEX Biologics, Inc.
- Integra LifeSciences Holdings Corporation
- Celularity Inc.
- Applied Biologics LLC
- Surgenex, LLC
- Pinnacle Transplant Technologies
- BioStem Technologies, Inc.
- StimLabs, LLC
- BioTissue Holdings, Inc. / AMNIOX Medical, Inc.
- AlloSource
- MTF Biologics
Sources
Primary Research Interviews
Industry Stakeholders list
- Tissue bank directors
- Regulatory affairs leaders in human tissue establishments
- Hospital procurement managers for biologics and surgical products
- Wound care product formulary decision makers
- Ophthalmic surgery center administrators
- Reconstructive and endocrine surgery specialists
End Users list
- Podiatrists
- Wound care physicians
- Plastic and reconstructive surgeons
- Ophthalmologists and corneal specialists
- General surgeons using biologic grafts
- Outpatient wound clinic managers
Government and International Databases
- U.S. Food and Drug Administration
- Centers for Medicare & Medicaid Services
- Health Resources and Services Administration
- Organ Procurement and Transplantation Network
- ClinicalTrials.gov
Trade Publications
- Wounds
- MedTech Dive
- STAT
- AHC Media
- The American Journal of Managed Care
- Managed Healthcare Executive
Academic Journals
- Advances in Wound Care
- Cell and Tissue Banking
- Ophthalmology
- Cornea
- Plastic and Reconstructive Surgery
- JAMA Surgery
Reputable Newspapers
- Financial Times
- The Wall Street Journal
- The Washington Post
- Los Angeles Times
Industry Associations
- Association for Advancing Tissue and Biologics
- Wound Healing Society
- American Academy of Ophthalmology
- American College of Surgeons
- Alliance for Regenerative Medicine
- American Medical Association
Public Domain Resources
- Federal Register
- NIH RePORTER
- FDA HCTERS Public Query Application
- PubMed Central
- ClinicalTrials.gov results database
- CMS Physician Fee Schedule Look-Up Tool
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 8 Years
Share
Share
About Author
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients