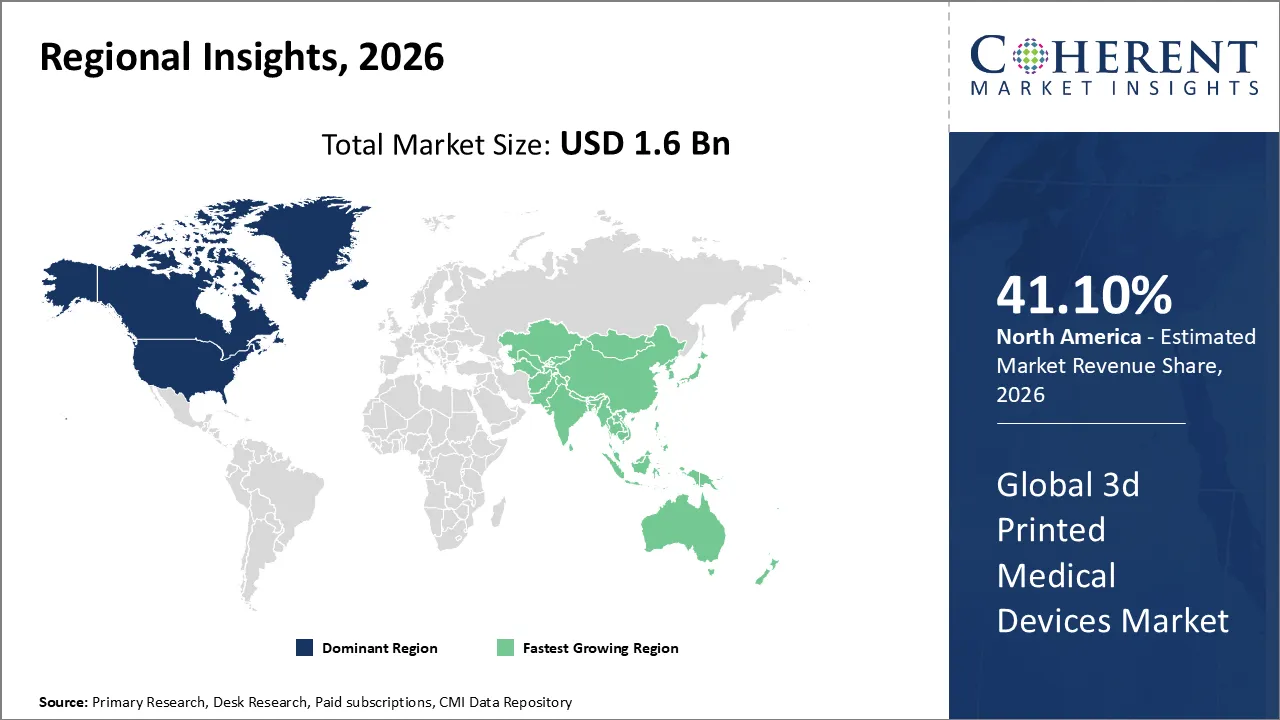
The Global 3d Printed Medical Devices Market is estimated to be valued at USD 1.6 Bn in 2026 and is expected to reach USD 4.2 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 14.2% from 2026 to 2033.
The Global 3D Printed Medical Devices Market actively transforms healthcare by producing patient‑specific implants, prosthetics, surgical guides, anatomical models, and dental devices through additive manufacturing. Advanced printer technologies, biocompatible materials, and streamlined digital workflows allow manufacturers to produce devices faster and improve clinical outcomes. Hospitals and research centers increasingly operate in‑house 3D printing for planning, fabrication, and complex device creation. The market grows as major regions invest in healthcare technology and strengthen regulatory frameworks.
|
Current Events |
Description and its impact |
|
Technological Advancements in 3D Printing Materials and Processes |
|
|
Regulatory Landscape and Compliance Changes |
|
|
Economic Factors and Healthcare Investment Trends |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Orthopedic Implants hold the largest market share of 43.4% in 2026. The orthopedic implants segment in the global 3D printed medical devices market grows as healthcare providers increasingly demand anatomically precise, patient-specific solutions that enhance surgical accuracy and minimize complications. Rising incidences of bone disorders, trauma cases, and age-related joint issues continue to drive the need for advanced implants. 3D printing technology allows manufacturers to create complex porous structures that improve bone integration and durability, while supporting better recovery outcomes. Increasing clinical trust and wider adoption of customized implants further propel segment growth. For instance, in August 2025, OIC International (USA), Medi Mold (AMTZ, India), and AddUp (France) formed a strategic partnership to set up a 3D-printed, precision-engineered orthopedic implant facility at AMTZ, India’s premier medical device park.
Digital Light Processing (DLP) expected to hold largest market share of 34.2% in 2026. Digital Light Processing (DLP) accelerates growth in the global 3D printed medical devices market by producing high-resolution, complex medical components with smooth surface quality. It cures entire layers at once, increasing speed and efficiency for rapid manufacturing and prototyping. Healthcare providers expand its use in dental and surgical applications, while manufacturers develop biocompatible resins to support adoption. Seamless integration with digital design and imaging systems improves precision, and rising demand for customized medical solutions further drives its use. For instance, CollPlant has launched BioFlex, a ready-to-print kit based on its proprietary rhCollagen platform for Digital Light Processing (DLP) 3D bioprinting applications.
Metals and Alloys acquired the prominent market share of 45.8% in 2026. Metals and alloys strengthen the global 3D printed medical devices market by offering high durability, strength, and reliability for critical applications. Manufacturers actively utilize materials like titanium and cobalt-chrome to create implants capable of handling mechanical stress and ensuring long-term performance. Additive manufacturing allows them to design complex geometries and porous structures that improve stability and support bone integration. Rising demand for high-performance implants in orthopedic and dental fields, along with ongoing material innovations, continues to drive the adoption of metal-based 3D printing.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 41.1% in 2026. North America drives key trends in the global 3D printed medical devices market by rapidly adopting patient-specific solutions, integrating digital health technologies, and expanding in-hospital 3D printing labs. Strong healthcare infrastructure and supportive regulatory frameworks promote continuous innovation and clinical application. Technology companies and medical institutions actively collaborate to advance development, while increased use of biocompatible materials and AI-powered design tools improves precision, optimizes workflows, and enables broader use across diverse medical specialties. For instance, in March 2026, 3D Systems received full EU MDR 2017/745 certification, confirming its quality system, documentation, and clinical evidence meet strict medical device standards. The company will now launch MDR-compliant dental products across European markets in a phased rollout.
Asia Pacific is rapidly advancing in the global 3D printed medical devices market as healthcare providers expand infrastructure, attract medical tourism, and invest in advanced manufacturing technologies. Governments and private companies actively foster innovation, driving adoption across hospitals and research centers. Healthcare stakeholders increasingly demand cost-effective, customized solutions and utilize digital design tools to broaden applications. A large patient population and a stronger focus on local manufacturing further accelerate the region’s development in this market. For instance, in November 2025, the Korea Institute of Machinery and Materials (KIMM) has launched a global research network linking China and Vietnam, strengthening collaboration with Jilin University in 3D printing, precision manufacturing, and biomimetic technology.
The United States drives key trends in the global 3D printed medical devices market by widely adopting patient-specific implants, surgical guides, and anatomical models. Hospitals and research centers actively implement in-house 3D printing to streamline surgical planning and shorten production times. Medical device manufacturers and technology companies collaborate closely to foster innovation, while FDA regulatory clarity enables faster clinical approvals. Increased use of biocompatible materials, AI-powered design, and digital workflows enhances precision, efficiency, and the delivery of customized medical care.
China drives trends in the global 3D printed medical devices market by expanding healthcare infrastructure, increasing demand for personalized medical solutions, and investing in advanced manufacturing technologies. Hospitals and research centers actively implement 3D printing to produce customized implants, surgical models, and prosthetics. Local medical device companies collaborate with technology providers to accelerate innovation, while wider access to biocompatible materials and digital design tools improves precision and efficiency. A large patient population and emphasis on domestic production further boost market growth. For instance, in November 2025, Aether launched RapidPrint, a polymer filament line that speeds up 3D printing by up to 5X for high-performance engineering materials, enhancing productivity for aerospace, defense, and industrial users.
The market increasingly emphasizes personalized healthcare solutions, with 3D printing enabling implants, prosthetics, and surgical guides tailored to individual patient anatomy. Clinicians and hospitals leverage imaging data to produce highly accurate, patient-specific devices, improving surgical outcomes and comfort. This trend drives demand for advanced printers and flexible materials capable of producing complex geometries, while encouraging innovation in design software that integrates seamlessly with clinical workflows to meet the growing need for customized medical solutions.
Hospitals and surgical centers are establishing in-house 3D printing labs to produce anatomical models, implants, and prosthetics on-site. This trend shortens lead times, enhances surgical planning, and allows rapid prototyping for complex cases. By integrating point-of-care printing, healthcare providers improve responsiveness to patient needs, reduce dependency on external manufacturers, and optimize clinical workflows, positioning 3D printing as a practical, everyday tool rather than a specialized service limited to high-end research facilities.
The growing demand for customized medical solutions presents a significant opportunity for manufacturers to produce implants, prosthetics, and surgical guides tailored to individual patients. By leveraging advanced imaging, AI, and 3D printing technologies, companies can create devices that improve surgical precision, reduce recovery time, and enhance patient outcomes. Hospitals and clinics increasingly seek these solutions to differentiate care quality, making patient-specific production a lucrative avenue for innovation and market growth.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.6 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 14.2% | 2033 Value Projection: | USD 4.2 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
3D Systems, Inc., Arcam AB, Stratasys Ltd., FabRx Ltd., EOS GmbH Electro Optical Systems, EnvisionTEC, Cyfuse Biomedical K.K., Bio3D Technologies, PrinterPrezz, Carima, Nexxt Spine, and Aurora Labs. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients