The global tumor ablation market was valued at USD 48,870 Mn in 2026 and is forecast to reach a value of USD 88,950 Mn by 2033 at a CAGR of 8.9% between 2026 and 2033.
Point-of-care (PoC) testing has the ability to provide immediate blood gas and other critical care test findings to patients in close proximity. PoC can result in faster therapeutic intervention, fewer preanalytical mistakes, and better patient care when appropriately integrated into the patient care process. However, to avoid errors while sample handling, test inaccuracy, nonlaboratorians training and ongoing competency assessment, and justification of the additional costs of analyzers and test units or cartridges, PoC demands a higher level of oversight and quality control. The advantages of PoC are also influenced by the number of tests performed and the distance between the patient's home and a clinical laboratory. PoC allows for faster clinical decision-making in the areas of diagnosis, therapy selection and monitoring, and prognosis, as well as operational decision-making and resource usage.
|
Current Event |
Description and its Impact |
|
AI and Digital Health Technology Integration |
|
|
Global Healthcare Infrastructure Modernization |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
|
Area |
Market Metric |
Key Impact |
Unique Insights |
|
Diabetes Care |
Over 500 million people projected to live with diabetes worldwide by 2030 |
Enables continuous glucose monitoring and rapid blood sugar checks |
Rising prevalence drives demand for home-use kits and wearable biosensors, making diabetes the largest single application area in Point-of-Care diagnostics |
|
Cardiac Diagnostics |
Rapid troponin PoC tests widely adopted |
Cuts emergency department triage time by up to 50% |
Early detection improves survival rates; AI-powered ECG platforms (e.g., PMcardio) reduce door-to-balloon times by 20–30 minutes |
|
Infectious Disease |
PoC testing grew 25% year-on-year post-COVID |
Respiratory panels lead adoption |
Expansion into multiplex testing for flu, RSV, and COVID; portable kits now used in rural and resource-limited settings |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of technology, the lateral flow assays segment is expected to hold 43.2% share in 2026, due to their simplicity, affordability, and can give quick results. They widely used for infectious diseases like COVID-19, HIV, and the flu, additionally, they require minimal infrastructure and give results in minutes. Since they are easy to move and use, they are essential in hospitals, clinics, and home care, which is why they are used all over the world.
For instance, in August 2025, Biolabs International released its Connected Diagnostics app, which allows people test themselves at home and get instant, professional help. The app combines quick SpeedySwab tests for COVID-19 and the flu with AI-driven analysis and telehealth support.
In terms of application, the diagnostics testing & monitoring segment is expected to lead the market with 72.8% share in 2026, due to rising demand for quick, reliable results for managing infectious and chronic diseases. Point-of-care solutions give both doctors and patients more power, from glucose monitoring to pregnancy kits to cancer diagnostics. They are still at the top as they can speed up processes and get better results.
For example, in October 2025, Co-Diagnostics declared they were making a special tool to help get samples ready for point-of-care testing. This new idea helps diagnostic workflows go faster and be more reliable, which makes testing on the patient side more efficient and accurate, and strengthens the role of diagnostic testing and monitoring.
In terms of end user, the hospitals segment is projected to account for 27% share of the market in 2026. Hospitals are the main end-users because they deal with complicated cases that need quick diagnostics. Point-of-care platforms speed up the process, help with emergency care, and integrate seamlessly in with hospital workflows. Hospitals will always be a key part of adoption because of their infrastructure, the number of patients they see, and their need for quick results, especially for testing for critical and infectious diseases.
For instance, in June 2025, Philips released the Flash 5100 point-of-care ultrasound system, which is meant to provide quick, high-quality imaging right at the patient's bedside. This new technology improves hospital diagnostics by making them faster, more accurate, and efficient. It also helps make decisions about critical care and strengthens the role of point-of-care technologies in modern healthcare settings.
To learn more about this report, Request Free Sample
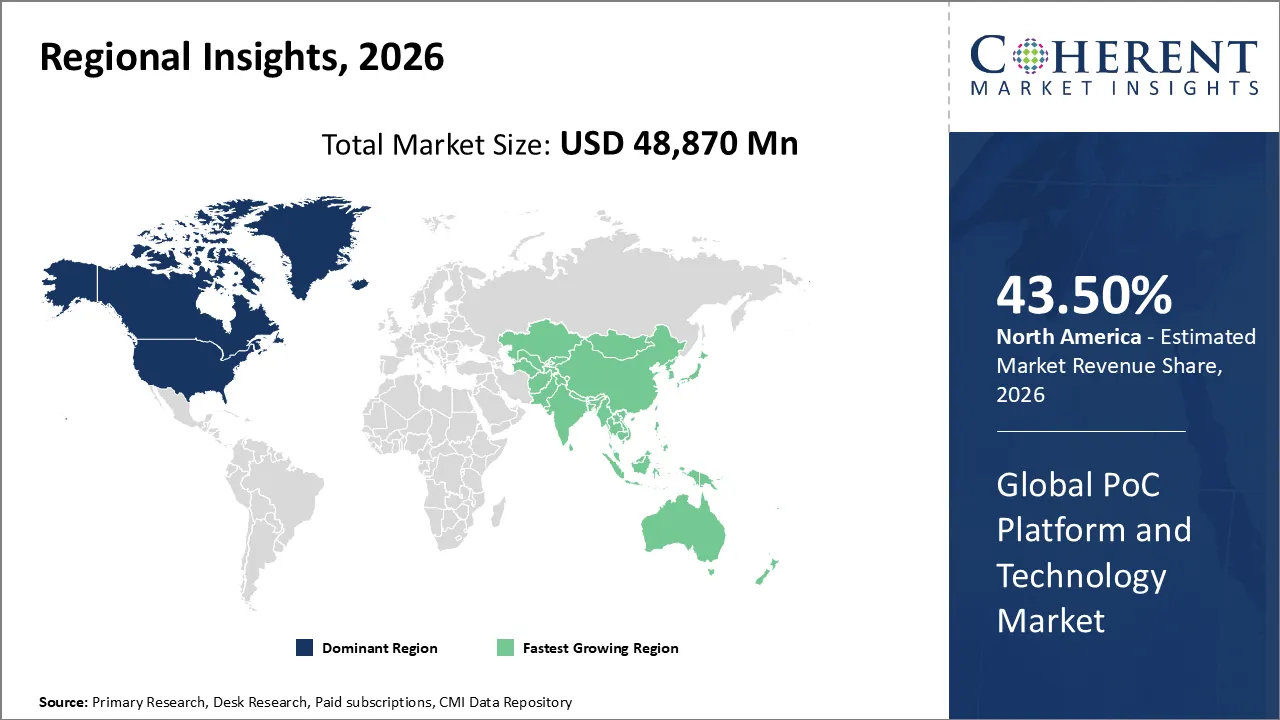
North America is expected to have the largest share of the market, making up about 43.50% of the global market. Advanced healthcare infrastructure, a high rate of adoption of new diagnostic technologies, favorable reimbursement policies, and a high rate of chronic and infectious diseases all contribute to this dominance. The U.S. is in the lead due to its strong investments in research and development and quickly adopts digital health solutions.
For instance, in January 2026, Union Image launched PoC Studio at CES 2026 in Las Vegas. The division's main job is to test proof-of-concept for vision-based technologies like camera modules, imaging devices, and AI-driven vision systems. It connects the idea stage to the product stage, letting innovators test their ideas, make prototypes quickly, and speed up the process of bringing their products to market in the AI era.
The Asia-Pacific region is expected to grow at fastest rate, with a CAGR of 13% through 2026. Growth is being driven by rising healthcare costs, a growing patient population, and a rising need for affordable, decentralized diagnostics. China, India, and Japan are highly investing on PoC technologies to fight both infectious and long-term diseases. This surge makes APAC a key driver of future demand for PoC platform and technology market.
For instance, in January 2026, Monstarlab, which is based in Tokyo, Japan, has made an extensive update to its AI-powered point-of-care development platform, MonstarX. The new release adds specification-driven workflows, which improve efficiency, scalability, and innovation.
The PoC platform and technology market is anticipated to be popular in the U.S. in 2026 because of the country's advanced healthcare infrastructure, the widespread use of new diagnostic tools, and the growing number of people with chronic diseases. Favorable reimbursement policies and integration with digital health ecosystems make adoption even faster, making the U.S. the largest regional market.
For instance, in January 2026, Doceree, which is based in Short Hills, New Jersey, was the only truly direct Point-of-Care engagement platform at scale when it started in 2026. It stands out from aggregator-led models, enabling it has more than 150 direct EHR integrations. This lets healthcare marketers connect with doctors easily at the point of care all over the U.S.
In 2026, the PoC Platform and Technology Market is extremely flourishing in China because healthcare costs are going up, there are an increasing number of patients, and the government is working to modernize diagnostics. Rapid urbanization, a rise in chronic diseases, and the use of cheap decentralized testing solutions all help the market grow even faster, which strengthens China's position in the region.
For instance, in August 2025, POCSTARS, which is based in Shenzhen, China, released its SaaS-based Mobile Device Management (MDM) platform. It was made for Android operations that are essential to the mission. It has centralized control, real-time monitoring, remote commands, secure app/data management, and live tracking. It works with industries like public safety, logistics, transportation, utilities, and industrial operations.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 48,870 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.9% | 2033 Value Projection: | USD 88,950 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Advanced Liquid Logic, Inc., Agilent Technologies, Axxicon Moulds, Eindhoven B.V., Lifesensors, Inc., Abbott Laboratories, Seimens Healthineers AG, LifeScan, Inc., Atomo Diagnostics, SenzaGen, Exalenz Biosciences Ltd., Nanomedical Diagnostics, BGN Technologies, Mesa Biotech Inc., and Millar, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The PoC Platform and Technology Market demand is high because growing need need for quick and accurate testing solutions. Infectious diseases like COVID-19, the flu, and HIV have highlighted the importance of decentralized diagnostics that give results in minutes. This trend is reshaping healthcare is delivered, making it possible to make treatment decisions faster, easing the burden on hospitals, and improving patient outcomes. PoC platforms are becoming important in both developed and emerging markets as healthcare systems focus on making care more accessible and faster.
The PoC Platform and Technology Market share is growing significantly driven by ongoing improvements in microfluidics, molecular diagnostics (NAAT, CRISPR), biosensors, and lateral flow assays. These improvements make devices more accurate, portable, and easy to use, which makes them more reliable and useful in a wider range of clinical settings. Better integration with digital health tools makes adoption even stronger because providers can easily analyze and share diagnostic data. PoC platforms are growing beyond infectious diseases into chronic care, oncology, and personalized medicine as technology improves. This is helping them reach more people.
The PoC platform and technology market forecast highlights there is an enormous opportunity to help people with chronic diseases. Point-of-care platforms are being used steadily to check for diabetes, heart disease, cancer, and other infectious diseases. Not only does this diversification make clinical applications more useful, but it also gives businesses new ways to make funds. PoC technologies help with preventive healthcare and personalized treatment plans by making it possible to find issue early and monitor on them over time. As chronic diseases become more common around the world, this group is likely to lead to long-term market growth and adoption.
The PoC (Point-of-Care) platform and technology market is still changing and evolving. It is a crucial part of decentralized diagnostics and real-time clinical decision support. PoC technologies are becoming more common in healthcare workflows, and adoption rates show that most diagnostic processes now use PoC solutions to speed up turnaround times and improve patient care. These platforms use a wide range of technologies, such as biosensors, lateral flow assays, microfluidics, and AI-enabled systems. They all work together to make it possible to do more tests outside of labs that are in one place.
Application data show that blood glucose monitoring and testing for infectious diseases are two of the greatest portion of the market. This is because people need quick results to manage chronic diseases and in emergencies. Hospitals and clinics are the most common places to use point-of-care (PoC) devices, but there is a lot of growth in outpatient and home healthcare settings as portable, easy-to-use devices become more popular.
North America is still in the lead because it has a well-developed healthcare system and a high rate of technology use. But places like Asia-Pacific are growing quickly because more people can get health care and more money is going into decentralized diagnostic capacity.
Companies are competing in new ways because of new technologies that make things smaller, make biosensing more accurate, and connect data. Strategic partnerships between diagnostic companies and tech companies are making products smarter and helping them work better together in stronger healthcare systems.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients