GLP-1 receptor agonist market is estimated to be valued at USD 31.71 Bn in 2026 and is expected to reach USD 72.33 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 12.5% from 2026 to 2033.
To learn more about this report, Request Free Sample
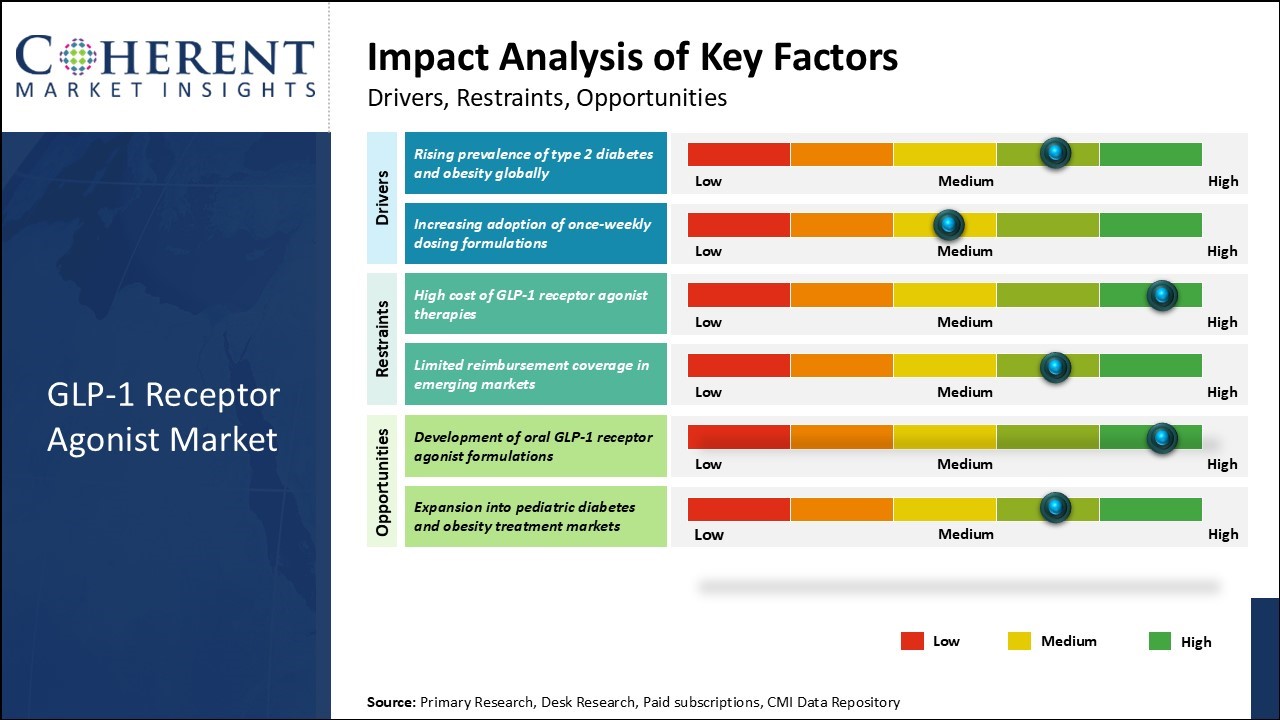
The market growth can be attributed to factors such as rising prevalence of type 2 diabetes and obesity across countries. Global GLP-1 receptor agonist market is expected to witness lucrative growth over the forecast period as increasing number of patients are opting for GLP-1 drugs due to their greater efficacy and lesser side effects compared to conventional diabetes medications. Furthermore, rising launch of novel formulations such as long-acting therapies with dosing convenience can drive the market growth.
However, the high development cost of GLP-1 drugs can hamper the global GLP-1 receptor agonist market growth.
|
Current Events |
Description and its impact |
|
U.S. FDA and Regulatory Expansion Initiatives |
|
|
Global Supply Chain and Manufacturing Disruptions |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The world is witnessing an alarming surge in the prevalence of type 2 diabetes and obesity, which is a major catalyst for the rapid expansion of the GLP‑1 receptor agonist market. Type 2 diabetes—marked by insulin resistance and disrupted glucose metabolism—poses one of the most pressing public health challenges. GLP‑1 receptor agonists offer a therapeutic breakthrough by mimicking the glucagon‑like peptide‑1 hormone, boosting insulin secretion and suppressing glucagon release, thereby enhancing glycemic control.
For instance, according to the latest International Diabetes Federation (IDF) Diabetes Atlas (2025), approximately 11.1% of adults aged 20–79 years—about 1 in 9 people—are living with diabetes globally, and more than 4 in 10 of those individuals remain undiagnosed.
In terms of drug type, liraglutide is expected to contribute 26.5% share of the market in 2026, as its once-daily subcutaneous administration provides consistent glycemic control throughout the day.
Clinical trials have demonstrated Liraglutide's ability to lower HbA1c levels by up to 25.5% on an average, with weight loss of 2-3 kg observed as an added benefit. The drug also helps reduce cardiovascular risks in Type 2 diabetes patients. These therapeutic advantages have led to increasing prescriptions by clinicians for glycemic management.
To learn more about this report, Request Free Sample
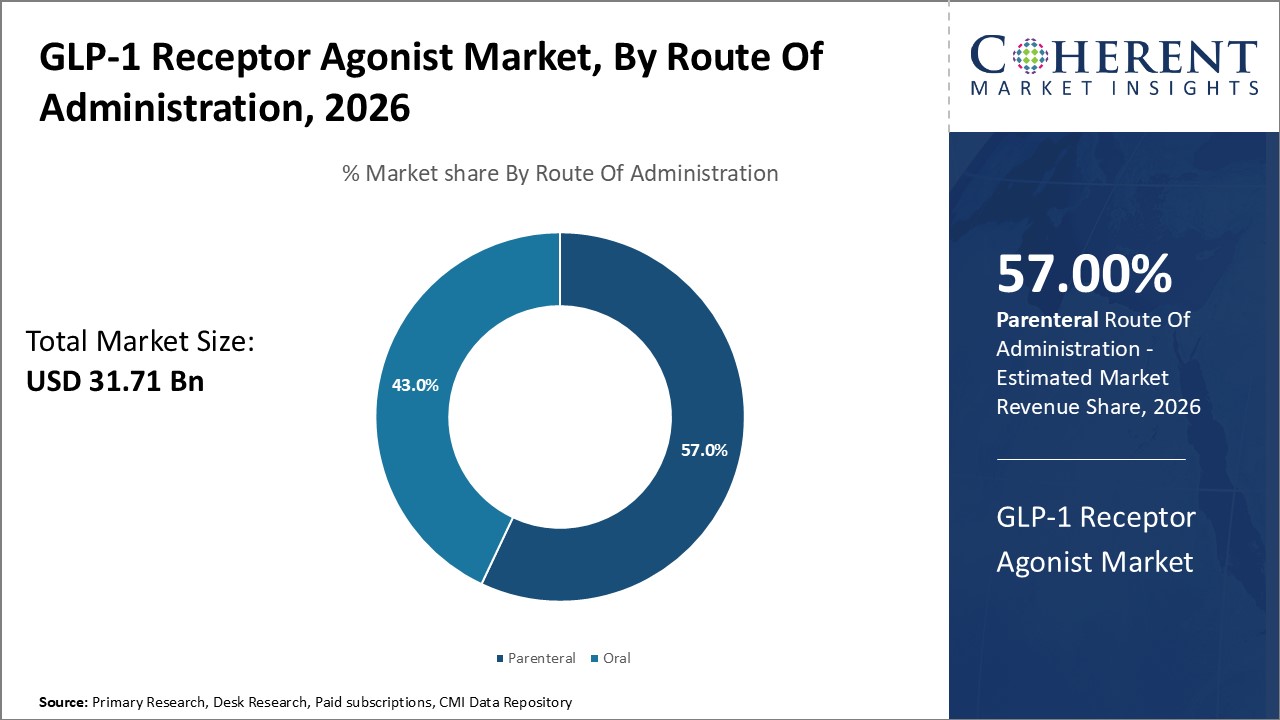
By route of administration, the parenteral segment is estimated to contribute the highest market share owing to its unparalleled convenience. While oral formulations offer non-invasive dosing, their low bioavailability and potential gastrointestinal side effects have remained major limitations.
Injectable GLP-1 receptor agonists provide predictable absorption of the active drug directly into systemic circulation, resulting in consistent glucose-lowering activity. Prefilled multi-dose pens and autoinjector devices have further elevated the ease of self-administration via the subcutaneous route. This improved convenience has led to greater patient acceptance and better treatment compliance compared to other complex anti-diabetes regimens.
By application, type 2 diabetes mellitus segment is estimated to contribute the highest market share of, due to their augmented effectiveness in diabetes care. These drugs aid glycemic control through multiple mechanisms like enhancing insulin secretion, suppressing glucagon levels, and reducing appetite to induce weight loss.
Studies have shown that GLP-1 receptor agonists provide better glycemic control and lower the risks of hypoglycemia compared to other anti-diabetes medications. Weight reduction achieved through these agents facilitates management of other diabetes co-morbidities and reduces cardiovascular complications. Furthermore, GLP-1 receptor agonists allow greater treatment customization based on individual patient's disease progression and therapeutic response.
To learn more about this report, Request Free Sample
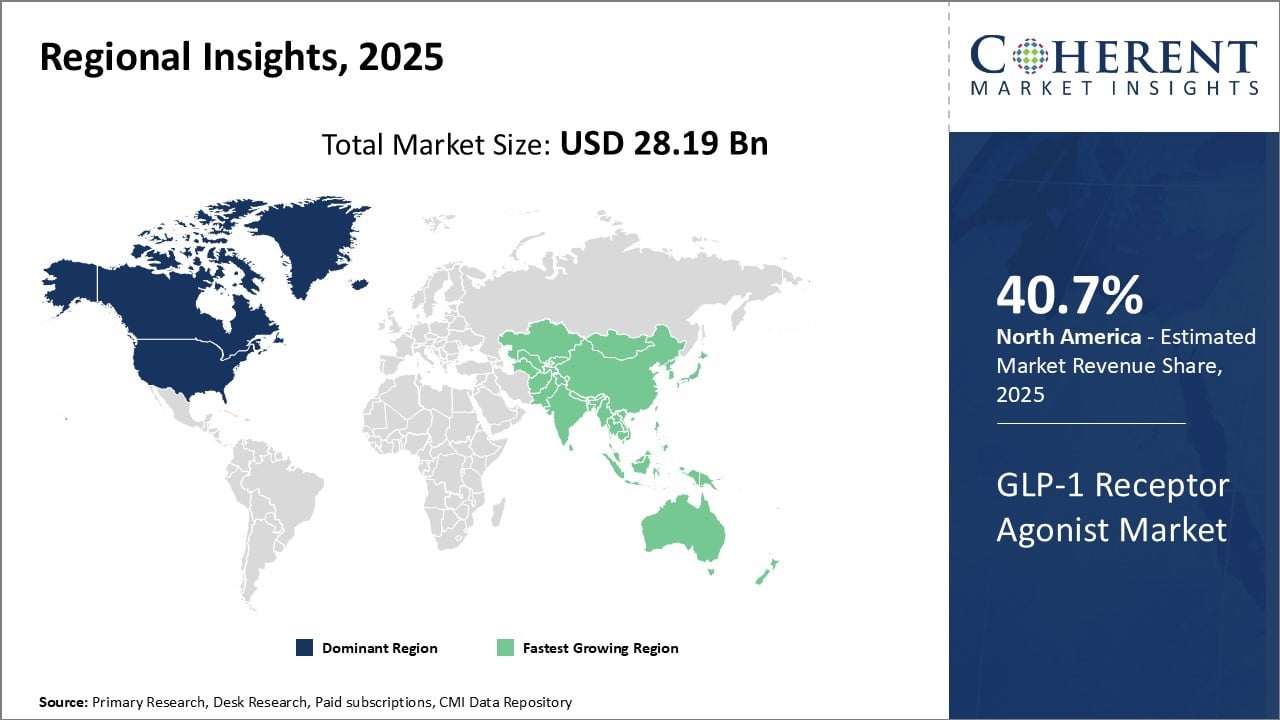
North America dominates the global GLP-1 receptor agonist market over with an estimated market share of 40.7% in 2026. The region has a highly developed healthcare infrastructure and availability of advanced treatment options. Healthcare expenditure is also higher in the U.S. and Canada compared to other parts of the world, enabling more patients to access costly drugs such as GLP-1 receptor agonists.
For instance, a specific RAND survey isn't publicly validated, but a 2024 poll by the American Medical Association estimates that about 16% of U.S. adults—roughly 1 in 8—have used GLP‑1 medications such as Ozempic or Wegovy.
Asia Pacific is poised to register the fastest growth in the global GLP-1 receptor agonist market during the forecast period. Rapid economic development, rising healthcare expenditure, growing diabetes epidemic and increasing focus of international players on emerging Asian markets can drive the market growth.
For instance, according to the International Diabetes Federation Diabetes Atlas (2021 edition), China and India were estimated to have 140.9 million and 74.2 million adults with diabetes in 2021, and these numbers are projected to increase to 174.4 million and 124.9 million, respectively, by 2045.
The U.S. stands as the foremost national market for GLP‑1 receptor agonists, fueled by its sophisticated healthcare infrastructure, effective reimbursement mechanisms, and aggressive public awareness campaigns. Direct-to-consumer advertising has elevated patient demand, while robust clinical and prescribing ecosystems make the U.S. the global engine of GLP‑1 adoption.
China represents a major emerging market, driven by an increasing diabetes burden, expanding healthcare access, and supportive government initiatives. Major drug manufacturers have actively engaged with local partners, establishing production and distribution channels tailored to the country’s needs.
India’s market for GLP‑1 receptor agonists is gaining significant momentum, driven by unmet clinical demand, rising awareness, and a steadily expanding healthcare ecosystem. The introduction of generics, such as Glenmark’s liraglutide biosimilar launched at dramatically lower prices, reflects the push toward affordability and broader access in a cost-conscious market.
Japan remains a key regional player, distinguished by its strong healthcare infrastructure and universal insurance coverage. Key GLP‑1 therapies are integrated into national insurance schemes, and the country’s aging population underscores its strategic importance in diabetes and obesity management.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 31.71 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 12.5% | 2033 Value Projection: | USD 72.33 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Eli Lilly and Company, Sanofi, Novo Nordisk A/S, AstraZeneca, Perrigo Company PLC, 3M Company, Bausch Health, Taro Pharmaceutical Industries, Novan Inc., Serum Institute of India Pvt. Ltd., Orgenesis biotech company, Verrica Pharmaceuticals, AbbVie Inc., Lee's Pharmaceutical Holdings Ltd, and Cassiopea, and Glenmark Pharmaceuticals |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Global GLP-1 receptor agonist market involves pharmaceutical products that mimic the effects of naturally occurring glucagon-like peptide-1 (GLP-1). GLP-1 receptor agonists work by stimulating the release of insulin from the pancreas in a glucose-dependent manner. These are used as treatment options for type 2 diabetes by helping to control blood sugar levels. These have also been shown to promote weight loss in overweight/obese individuals with diabetes. The market includes drugs like exenatide, liraglutide, semaglutide, lixisenatide, and albiglutide.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients