The global idiopathic hypersomnia treatment market size is projected to grow from USD 394.7 Mn in 2026 to USD 709.6 Mn by 2033, registering a compound annual growth rate (CAGR) of 8.7% during the forecast period (2026–2033). The reason for the growing market size is the increase in the number of diagnoses for patients suffering from sleep disorders and idiopathic hypersomnia, and the availability of new CNS stimulants and wake-promoting agents.
Stimulant medications segment is anticipated to dominate the idiopathic hypersomnia market in terms of revenue share in 2026, contributing to about 47% of the total revenue share generated by the drug type category. This dominance is primarily attributed to their existing status as first-line treatments for EDS along with high efficacy levels.
Additionally, FDA-approved stimulant and wake-promoting drugs continue to be widely prescribed in sleep medicine practice, reinforcing their dominance in real-world treatment patterns.
In June 2025, Jazz Pharmaceuticals highlighted continued clinical and research advancement of Sunosi (solriamfetol), a wake-promoting stimulant approved by the U.S. FDA for excessive daytime sleepiness in narcolepsy and obstructive sleep apnea
To learn more about this report, Request Free Sample
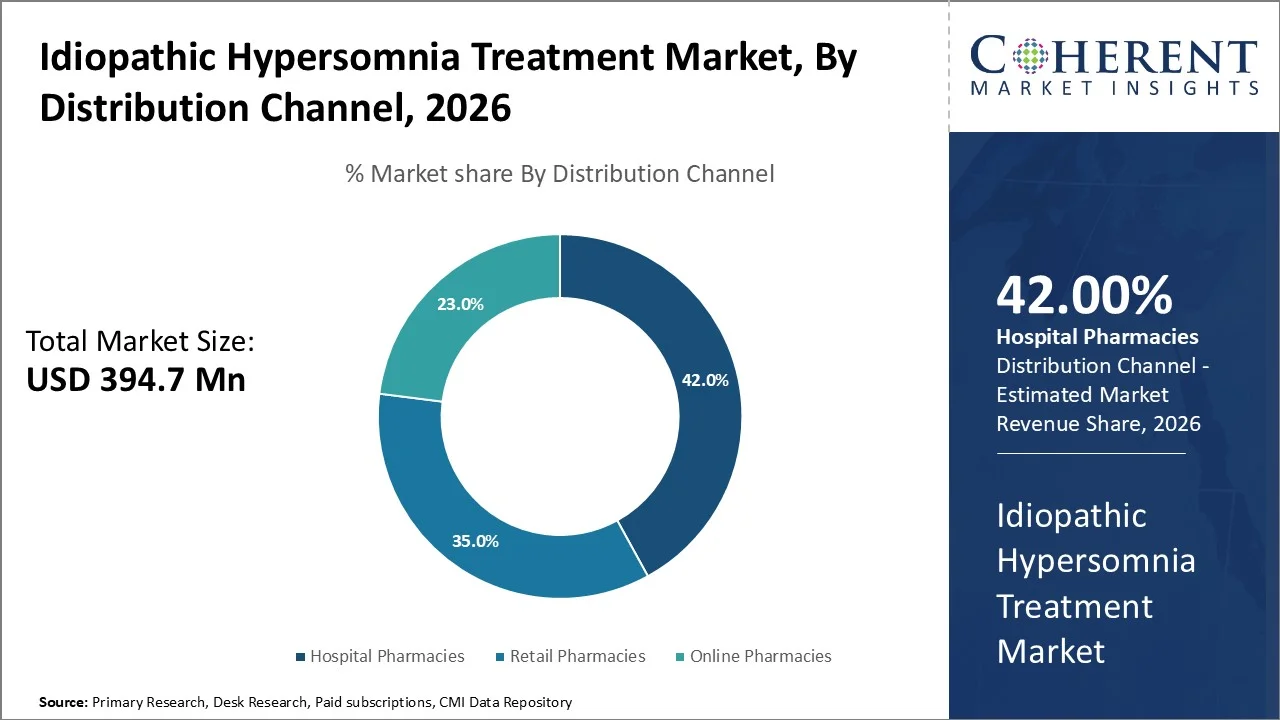
Hospital pharmacies account for the largest share of about 42% in 2026 due to their significant contribution to the specialty diagnosis, treatment initiation, and the regulated supply of CNS acting drugs for hypersomnolence disorders.
Hospitals and sleep centers act as the main source for diagnostic procedures and the start of treatment because of the need for specific tests, including polysomnography and multiple sleep latency test. The National Institute of Neurological Disorders and Stroke in the United States notes that patients with hypersomnia should be clinically evaluated in special centers, thus promoting drug dispensing in hospitals.
Idiopathic hypersomnia is increasingly recognized as part of the broader group of central disorders of hypersomnolence, yet it remains significantly underdiagnosed, creating a growing treatment gap and driving demand for pharmacological therapies.
As per the U.S. National Institute of Neurological Disorders and Stroke (NINDS), hypersomnias are disorders of the neurological system that are chronic in nature and marked by daytime sleepiness impacting everyday activities.
The growth of the market is propelled by the rising use of wake-promoting drugs like modafinil-based medications and new central nervous system stimulants.
The American Academy of Sleep Medicine (AASM) recommends wake-promoting agents as first-line therapy for excessive daytime sleepiness in central hypersomnolence disorders, supporting consistent prescription growth.
|
Current Event |
Description and its Impact |
|
Growing Regulatory Recognition and FDA Support for Orphan Drug Development in Rare Sleep Disorders (2025–2026) |
|
|
Expansion of National Sleep Health Policies and Diagnostic Guidelines (U.S. & EU, 2025–2026) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
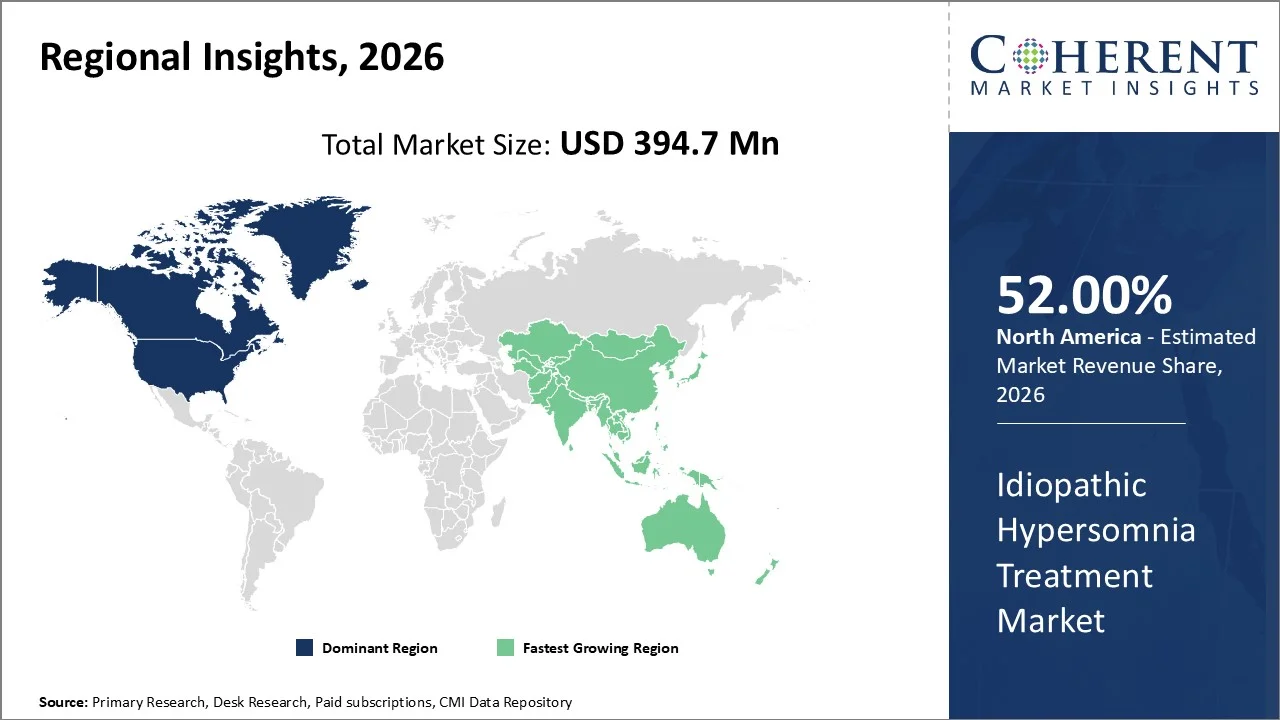
North America is estimated to contribute to 52% of the market share in 2026 due to high levels of early diagnosis, well-established centers for sleep disorders, and faster adoption of new drugs like sodium oxybate. The North American Idiopathic Hypersomnia Treatment Market is considered dominant due to the higher levels of disease awareness, advanced sleep disorder diagnostic facilities, and the presence of treatments to stimulate wakefulness and central nervous system function.
The region benefits from innovative clinical sleep testing facilities (polysomnography labs and MSLT availability), which support higher diagnosis rates compared to other regions. Moreover, strong presence of pharmaceutical companies and faster regulatory approvals (FDA pathways for sleep disorder drugs and orphan indications) are accelerating treatment adoption.
In March 2025, Jazz Pharmaceuticals announced new Phase 4 DUET study results, showing that Xywav significantly improved excessive daytime sleepiness and symptom severity in patients with idiopathic hypersomnia, reinforcing its clinical effectiveness in real-world settings.
Asia-Pacific region will emerge as the fastest growing region for treatment of idiopathic hypersomnia during the forecast period due to increasing awareness about sleep disorders, improvement in diagnostics, and development of health care infrastructure in developing countries like India and China, as well as in developed nations like Japan and South Korea.
In April 2026, Alkermes plc announced the initiation of its Phase 3 “Brilliance Studies” program evaluating alixorexton, an investigational oral orexin-2 receptor agonist for narcolepsy and idiopathic hypersomnia.
The United States idiopathic hypersomnia treatment market is witnessing steady growth, driven by increasing diagnosis rates of central disorders of hypersomnolence and strong innovation in wake-promoting therapies. Growing awareness of sleep-related issues combined with the developments in neuropharmacology in the treatment of orexin pathway disorder is fueling the demand for drugs.
In March 2026, Eli Lilly and Company acquired Centessa Pharmaceuticals to gain access to cleminorexton, an orexin receptor-2 agonist in Phase 2 trials for idiopathic hypersomnia.
Japan idiopathic hypersomnia treatment market is expected to show significant growth in the coming years, owing to the aging demographics and greater awareness about sleep disorders. Japan being among the fastest aging nations in the world, there exists growing demand for treatments that address excessive daytime sleepiness.
Additionally, global pipeline advancements such as the development of orexin agonists by firms such as Alkermes plc, are anticipated to impact future approval processes and marketing strategies in Japan.
Some of the major key players in Idiopathic Hypersomnia Treatment are Takeda Pharmaceutical Company Limited, Teva Pharmaceutical Industries Ltd., Jazz Pharmaceuticals, Inc., BIOPROJET, Avadel Pharmaceuticals, Plc. GlaxoSmithKline plc, Pfizer Inc., Theranexus, Fisher & Paykel Healthcare Limited, Drive DeVilbiss Healthcare LLC, and Merck & Co. Inc.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 394.7 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.7% | 2033 Value Projection: | USD 709.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Takeda Pharmaceutical Company Limited, Teva Pharmaceutical Industries Ltd., Jazz Pharmaceuticals, Inc., BIOPROJET, Avadel Pharmaceuticals, Plc. GlaxoSmithKline plc, Pfizer Inc., Theranexus, Fisher & Paykel Healthcare Limited, Drive DeVilbiss Healthcare LLC, and Merck & Co. Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients