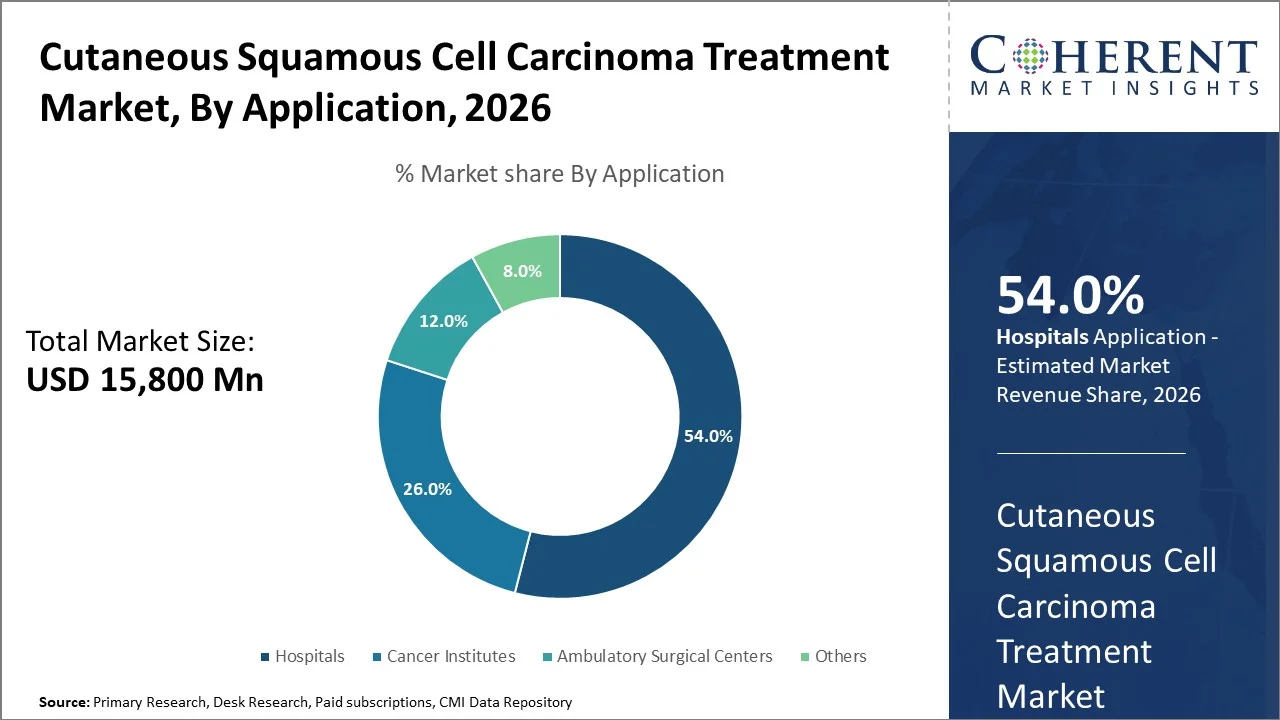
The global cutaneous squamous cell carcinoma treatment market size is estimated to reach USD 15,800 Mn in 2026 and is projected to grow at a CAGR of 7.2% during the forecast period (2026-2033), totaling USD 25,800 Mn by 2033. This can be attributed to rising prevalence of non-melanoma skin cancers and advancements in cutaneous SCC treatment drugs.
The global cutaneous squamous cell carcinoma treatment market is expected to grow steadily during the forecast period. This is due to increasing incidence of non-melanoma skin cancers, expanding awareness regarding early diagnosis and treatment options, rising demand for novel skin cancer drugs, and continuous advancements in targeted and immunotherapy-based treatments.
Rising adoption of minimally invasive surgical procedures and increasing availability of novel biologics and immune checkpoint inhibitors are also supporting market expansion. Similarly, development of advanced cutaneous SCC therapy options is expected to bode well for the market during the assessment period.
Cutaneous squamous cell carcinoma (cSCC) is a non-melanoma skin cancer (NMSC) and one of the most commonly diagnosed skin cancers, second only to basal cell carcinoma. Most cSCC cases are localized and highly treatable with surgical intervention. According to pooled clinical evidence, approximately 4–5% of patients experience local recurrence, around 3–4% develop nodal metastases, and about 1–2% die from the disease. Increasing incidence of cSCC, particularly in aging populations and high UV-exposure regions, is expected to support market growth.
By treatment, surgical segment is projected to account for a prominent market share of 60% in 2026. This is attributable to its status as the first-line and most effective curative approach for localized cutaneous squamous cell carcinoma. Surgical procedures such as excision and Mohs micrographic surgery offer high cure rates and precise tumor removal while preserving surrounding healthy tissue.
The strong preference for surgery is further supported by its ability to provide immediate tumor clearance and a lower recurrence risk compared to non-surgical options in early-stage cases. It also benefits from wide availability across healthcare settings, making it the standard treatment choice for most diagnosed patients.
To learn more about this report, Request Free Sample
According to Coherent Market Insights’ latest cutaneous squamous cell carcinoma treatment market analysis, hospitals segment is set to lead the market with a share of 54% in 2026. This is mostly due to their advanced clinical infrastructure, availability of specialized dermatology and oncology departments, and the ability to perform complex surgical procedures such as Mohs micrographic surgery under one roof.
In addition, hospitals remain the primary treatment setting for moderate to advanced cSCC cases because they offer multidisciplinary care, better patient monitoring, and improved access to reimbursement-supported therapies. On the other hand, cancer institutes segment is expected to grow at a higher CAGR due to advanced therapy adoption and trials.
Rising Incidence of Skin Cancers: There is a rise in cases of skin cancers, especially cutaneous squamous cell carcinoma, across nations like the U.S. and China due to prolonged exposure to ultraviolet radiation. For instance, the estimated annual incidence of cutaneous squamous cell carcinoma exceeds one million cases in the United States. This, in turn, is creating high demand for cutaneous squamous cell carcinoma treatment drugs and surgeries.
Advancements in Immunotherapy and Targeted Therapies: There is a rising shift toward checkpoint inhibitors, monoclonal antibodies, and PD-1/PD-L1 therapies, which have significantly improved survival outcomes in advanced cutaneous squamous cell carcinoma (cSCC). These therapies are increasingly becoming the preferred systemic treatment option for advanced or unresectable cases. They are also being explored in combination with surgery and radiation. This evolving treatment landscape is expected to create lucrative growth opportunities in the cutaneous squamous cell carcinoma treatment market during the forecast period.
Shift Towards Minimally Invasive and Non-Surgical Treatments: Rising preference for minimally invasive and non-surgical approaches is emerging as a key trend in the cutaneous squamous cell carcinoma treatment market, especially for selected low-risk or superficial lesions. There is increasing interest in options such as cryotherapy, photodynamic therapy (PDT), laser-based treatments, and certain topical therapies.
These approaches are valued for their favorable cosmetic outcomes, reduced recovery time, and lower risk of complications, making them suitable for patients who are not ideal candidates for surgery or in cases where less aggressive management is appropriate. However, surgical excision remains the standard and most widely adopted treatment for invasive cutaneous squamous cell carcinoma.
Innovations in the Cutaneous Squamous Cell Carcinoma (CSCC) treatment market are rapidly shifting toward peri-operative immunotherapy integration. Immune checkpoint inhibitors are now being used not only for advanced disease but also around surgery to prevent recurrence and improve long-term disease control. This reflects a major transition from reactive cancer treatment to risk-based, early immune intervention strategies.
A key recent breakthrough is the This approval is based on the Phase III C-POST trial. The study demonstrated a significant reduction in the risk of recurrence or death, with a hazard ratio of approximately 0.32. Overall, this highlights a major clinical shift toward post-operative immune-based strategies for preventing relapse.
|
Current Event |
Description and its Impact |
|
FDA expands immunotherapy use to prevent recurrence after surgery (2025) |
|
|
Strong clinical trial data is reshaping treatment standards (C-POST trial, 2025) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report

To learn more about this report, Request Free Sample
North America is expected to dominate the global cutaneous squamous cell carcinoma treatment market with a 42% share in 2026, owing to frequent regulatory approvals, high burden of skin cancer, and strong healthcare infrastructure. Rising adoption of advanced therapies such as immunotherapy, early diagnosis rates, and availability of FDA-approved drugs are improving treatment outcomes across the region.
There is a significant increase in the development and approval of novel medications for cutaneous squamous cell carcinoma across North America. For instance, in 2024, the U.S. Food and Drug Administration (FDA) approved cosibelimab-ipdl (Unloxcyt) for metastatic or locally advanced cSCC. Likwise, in 2025, the FDA expanded the use of cemiplimab (Libtayo) for adjuvant treatment in high-risk cSCC patients following surgery and radiation. These approvals will likely boost North America cutaneous squamous cell carcinoma treatment market growth during the forthcoming period.
Asia Pacific is anticipated to emerge as the most lucrative market for cutaneous squamous cell carcinoma treatment companies, accounting for a global market share of 22% in 2026. This can be attributed to improving healthcare infrastructure, increasing awareness about skin cancer, and expanding access to dermatological treatments. Countries such as China, India, and Japan are witnessing a steady rise in skin cancer diagnosis due to better screening programs and growing healthcare penetration. Furthermore, government initiatives aimed at strengthening cancer care services are supporting market expansion.
The United States is expected to remain the leading market for cutaneous squamous cell carcinoma treatments, owing to rising UV exposure-related skin cancer cases, high adoption of advanced immunotherapies, strong dermatology care infrastructure, and increasing geriatric population. In addition, early diagnosis rates and widespread availability of FDA-approved targeted and immune checkpoint therapies are further strengthening market growth.
A key growth driver is the high incidence of non-melanoma skin cancers, especially among the aging population with prolonged sun exposure history. The American Cancer Society reports that cutaneous squamous cell carcinoma (cSCC) is the second most common form of human skin cancer, with at least 200,000 to 400,000 new cases of cSCC expected each year in the US. This high burden is driving demand for surgical procedures such as Mohs surgery as well as advanced systemic treatment options.
China’s cutaneous squamous cell carcinoma treatment market is witnessing strong growth due to improving oncology care infrastructure, rising skin cancer awareness, expanding access to dermatology services, and increasing adoption of standardized cancer treatment protocols. In addition, rapid urbanization and growing environmental exposure factors are contributing to a higher burden of skin-related malignancies, supporting market expansion.
A key growth driver is the rapidly aging population, which is increasing the number of patients susceptible to skin cancer and related complications. According to recent demographic data, over one-fifth of China’s population is aged 60 and above. This demographic shift is expected to increase the burden of age-related conditions, including skin cancers such as cutaneous squamous cell carcinoma (cSCC), thereby fueling demand for surgical and non-surgical treatment options in advanced cases.
Some of the major players in Cutaneous Squamous Cell Carcinoma Treatment Market are Eli Lilly and Company, Sanofi S.A., Cadila Healthcare Limited, Cipla Limited, Castle Biosciences, Regeneron Pharmaceuticals, Inc., Merck & Co., Inc., Vidac Pharma, LEO Pharma A/S, Merck Sharp & Dohme, and Amgen Inc., among others.
Leading companies in the cutaneous squamous cell carcinoma (cSCC) treatment market, such as Regeneron Pharmaceuticals, Merck & Co., Bristol Myers Squibb, and Novartis, are adopting several key strategies to strengthen their position as well as improve patient outcomes. A major focus is on developing and expanding immunotherapy and targeted therapy options, especially PD-1/PD-L1 inhibitors like cemiplimab and pembrolizumab, which are increasingly used for advanced or metastatic cSCC cases.
Companies are also investing heavily in clinical trials to explore combination therapies and improve treatment effectiveness across different patient groups. Some players are increasingly focusing on personalized medicine approaches using biomarkers to identify patients who are most likely to respond to immunotherapy, helping improve treatment outcomes and market competitiveness.
For example,
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 15,800 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.2% | 2033 Value Projection: | USD 25,800 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Eli Lilly and Company, Sanofi S.A., Cadila Healthcare Limited, Cipla Limited, Castle Biosciences, Regeneron Pharmaceuticals, Inc., Merck & Co., Inc., Vidac Pharma, LEO Pharma A/S, Regeneron Pharmaceuticals, Inc., Merck Sharp & Dohme, and Amgen Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients