The Global Ingestible Smart Pills Market is anticipated to grow at a CAGR of 5.6%, with a USD 1.6 Bn in 2026 and is expected to reach approximately USD 2.3 Bn in 2033. This growth in the market is driven by the rising burden of chronic diseases noncommunicable diseases account for about 75% of global deaths, with 18 million premature deaths before age 70 worldwide (WHO) and increasing demand for digital health and remote patient monitoring solutions, supported by global health strategies to promote digital health adoption. (Source: WHO)
In 2026, the smart pills segment dominates the ingestible smart pills market due to increasing demand for minimally invasive and patient-friendly diagnostic solutions. Technologies such as capsule endoscopy enable detailed visualization of the gastrointestinal tract without the need for sedation or invasive procedures, improving patient comfort and compliance. According to the National Institutes of Health, capsule endoscopy has a diagnostic yield of over 60–70% for small bowel disorders, significantly higher than many traditional methods. Additionally, rising prevalence of gastrointestinal diseases, including Crohn’s disease, is further driving adoption, as smart pills allow continuous, real-time monitoring and more accurate disease management across clinical settings.
Two key factors are expected to drive continued growth in this segment. First, the healthcare industry is increasingly favoring minimally invasive gastrointestinal diagnostic procedures that offer visual evidence of hard-to-reach areas of the bowel without resorting to traditional invasive methods. Second, AI-assisted analysis of capsule data and improvements in workflow efficiencies have enhanced the detection of bleeding, Crohn's disease, polyps, and other abnormalities related to iron deficiency. This progress has led to greater confidence among clinicians in the use of smart pills.
The most significant development in this segment occurred on September 23, 2025, when Medtronic announced that its PillCam capsule for endoscopy had been used by over 4 million patients worldwide. This announcement was followed by the FDA's clearance on March 12, 2026, of the Deep Capsule, an AI-assisted analysis tool for small-bowel capsule endoscopy, designed to enhance the speed at which clinicians can review and interpret capsule data.
The imaging sector is expected to dominate in 2026, driven by the widespread adoption and commercial availability of capsule-based visualization for assessing small bowel and colon issues. This prominent status in imaging is supported by a strong demand for cancer diagnostics; the American Cancer Society projects 158,850 new cases of colorectal cancer in the U.S. for that year. Additionally, the FDA's approval of Deep Capsule® AI-assisted capsule endoscopy image analysis software on March 12, 2026, will further position imaging at the forefront of clinical workflows.
A growing population within the digestive disease sector is influenced by two key factors: many patients stand to benefit significantly from the development of non-invasive gastrointestinal assessment tools, and the high incidence of gastrointestinal bleeding is driving demand for increased access to swallowable imaging technologies for bowel evaluation. This condition results in over 1.5 million healthcare visits and approximately 500,000 hospital admissions annually in the U.S.
To learn more about this report, Request Free Sample
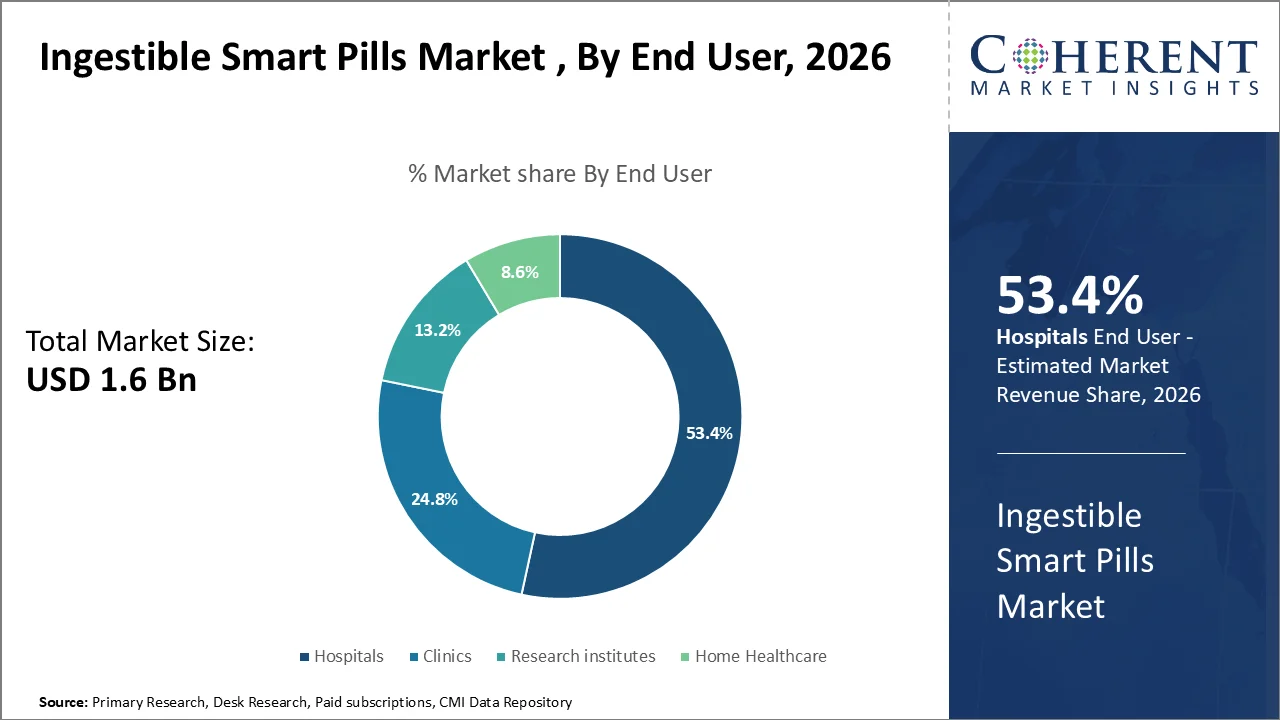
In 2026, hospitals will be the largest segment of end users, primarily due to their concentration of gastroenterologists, robust diagnostic infrastructure, and high patient volume necessary for ingestible smart pill procedures. The American Hospital Association’s 2026 hospital statistics indicate that there are 5,121 community hospitals in the U.S., with 3,567 of these being affiliated with hospital systems. This underscores the importance of hospitals as the most suitable environments for capsule-based diagnostics and follow-up care.
Hospital demand continues to rise, with the AHA reporting a 5.3% increase in inpatient volumes and a 9.8% increase in outpatient visits for 2025, which strengthens the case for hospital-based GI diagnostics. Reimbursement remains favorable, CMS indicates that capsule endoscopy interpretation is reimbursable in both inpatient and outpatient hospital settings, while hospital operating payment rates for FY 2026 are projected to increase by 2.6% for qualifying acute care hospitals.
The York and Scarborough Teaching Hospitals NHS Foundation Trust issued a protocol on February 3, 2025, for patients at risk of colorectal disease, comparing colon capsule endoscopy (CCE) to standard colonoscopy. Additionally, on April 17, 2026, CapsoVision announced that its AI-based pathology tools significantly reduce the time clinicians need to review images from CCE, making it a more financially attractive option for physicians.
The year 2026 represents a major change in the field of ingestible smart pills, as we transition from traditional imaging techniques to functional sensing capabilities and AI-based data interpretations. On June 27, 2025, Atmo Biosciences received U.S. FDA 510(k) clearance for its ingestible capsule, which can detect gas. This approval was supported by a multi-site pivotal study that successfully met all endpoints and will assist in diagnosing gut motility disorders. Additionally, Mass General Brigham reported that their ingestible biosensor pill was able to identify cases of acute mesenteric ischemia in preclinical models with 90% accuracy. Furthermore, the use of AI assistance in analyzing small bowel images obtained through capsule endoscopy reduced the review time from 120.9 minutes to 77.9 minutes, enhancing the clinical scalability of this technique. (Atmo Biosciences)
|
Current Events |
Description and its Impact |
|
FDA Approval of AI-Assisted Capsule Endoscopy Tool |
|
|
European Commission’s Clarification on AI Regulations in Medical Devices |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
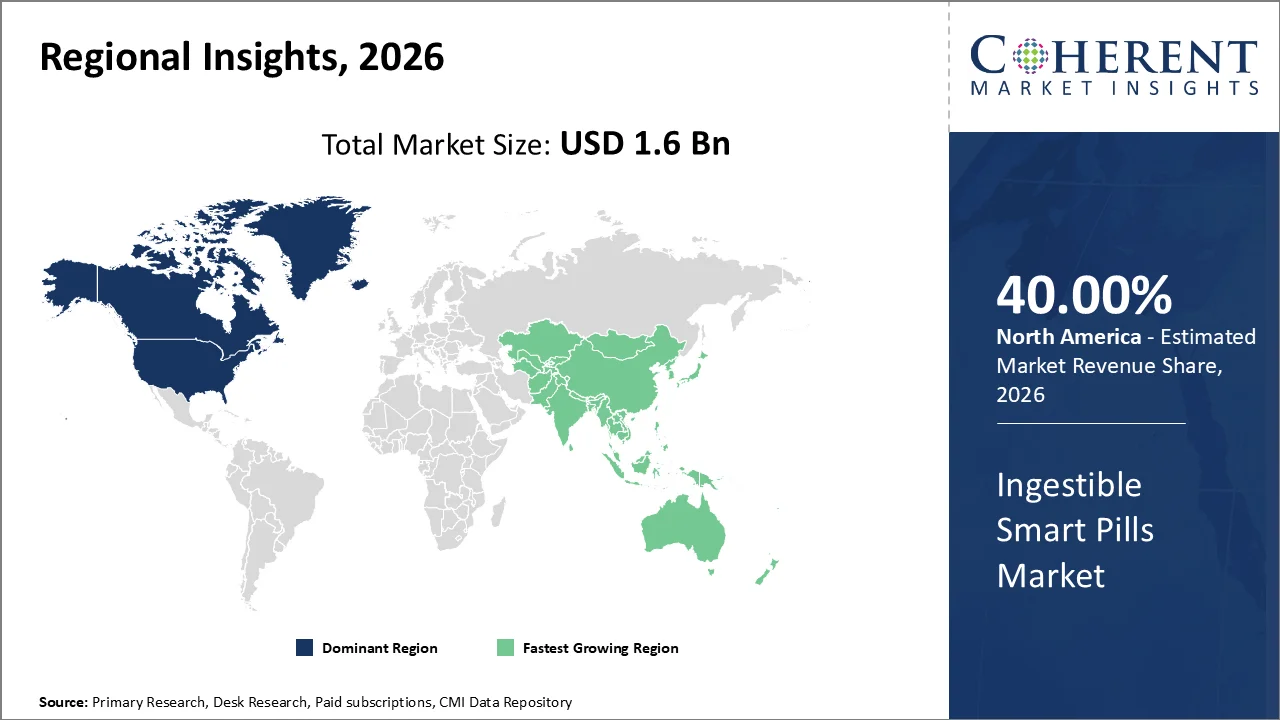
In 2026, North America is projected to hold the largest share of the global ingestible smart pill market, accounting for 40%. The U.S. healthcare system provides significant scale and depth in reimbursement options and has been an early adopter of AI-enabled diagnostic platforms. This trend facilitates the integration of innovative technologies, such as ingestible smart pills, into patient care and drives the expected growth of the market. According to the American Society of Gastrointestinal Endoscopy, over 20 million GI endoscopic procedures are performed each year in the United States, highlighting a significant need for advanced bowel visualization tools. Additionally, the U.S. Office of the National Coordinator reports that by 2025, 76% of hospitals will be using all four measured interoperability domains. As a result, there will be an increasing integration of image-rich capsule diagnostics into standard workflows, driven by the growing acceptance of routine capsule diagnostics.
The Asia Pacific region is projected to have the highest growth rate in 2026, with a market share of 24.3%. This growth is attributed to a rapidly increasing aging population, which is enhancing both the number of individuals and the capacity of health systems, along with a growing awareness of minimally invasive gastrointestinal (GI) diagnostics. The World Health Organization (WHO) reports that the Western Pacific Region currently has over 245 million people aged 65 and older. According to the United Nations Economic and Social Commission for Asia and the Pacific (ESCAP), 60% of the world's older population resides in the Asia and Pacific Regions. Furthermore, by 2025, individuals aged 65 and over are expected to account for 10.8% of the total population in that region. This demographic trend continues to expand the potential patient population for imaging technologies within smart pills.
The United States is the leading market in North America thanks to very high levels of healthcare expenditure combined with a large number of hospitals and a lot of activity related to commercializing various products in the hospital sector.The CMS referenced a next-generation multitarget stool DNA test with a specificity of 93.4% in the proposed 2026 final rule, highlighting the ongoing federal emphasis on promoting non-invasive methods for detecting colorectal cancer. Additionally, the ONC has indicated that certified EHR users must provide standardized APIs for FHIR, thereby facilitating better data exchange for image-rich and advanced diagnostic tools.
China has the potential to serve as a major growth engine for the Asia-Pacific region due to its large, aging population and improving health-related engagement from the population through the development of health literacy. A report from the National Bureau of Statistics stated that there were 223 million individuals age 65 and older by the end of 2025 and they represent about 15.9% of the overall population of China. According to the National Health Commission, the level of health literacy in China has grown to 33.69% by 2025 and that increase has helped develop greater awareness of the need for screening and have increased acceptance of new noninvasive diagnostic technologies.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.6 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.6% | 2033 Value Projection: | USD 2.3 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
BodyCap Medical, Given Imaging Ltd., Olympus Corporation, Proteus Digital Health, Inc., CapsoVision, Inc., Chongqing Jinshan Science and Component (Group) Co., Ltd., Medimetrics, IntroMedic Co., Ltd., Check-Cap Ltd., Boston Scientific Corporation, HQ Inc., Smart Pill Inc., Medtronic plc, Philips Healthcare, etectRx, Inc., Novartis, Siemens Healthcare, Pentax Medical Company (HOYA Corporation), GE Healthcare, and Stryker Corporation. |
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Major companies are BodyCap Medical, Given Imaging Ltd., Olympus Corporation, Proteus Digital Health, Inc., CapsoVision, Inc., Chongqing Jinshan Science and Component (Group) Co., Ltd., Medimetrics, IntroMedic Co., Ltd., Check-Cap Ltd., Boston Scientific Corporation, HQ Inc., Smart Pill Inc., Medtronic plc, Philips Healthcare, etectRx, Inc., Novartis, Siemens Healthcare, Pentax Medical Company (HOYA Corporation), GE Healthcare, and Stryker Corporation.
On June 26, 2025, the University of Maryland announced a significant advancement in ingestible capsule research: a 3D-printed capsule featuring integrated microneedles and a tunable drug reservoir capable of regulating drug release from seconds to months within the gastrointestinal (GI) tract.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients