MEA Technetium 99m Market is estimated to be valued at USD 685.3 Mn in 2026 and is expected to reach USD 1,220.9 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 8.6% from 2026 to 2033. Technetium-99m is a short-lived form of Technetium-99, an isotope of technetium. It is found as a component of nuclear waste and is used as a medical diagnostic tool. It is used as a radioactive tracer and can be detected in the body by medical equipment such as gamma cameras and Single Photon Emission Computed Tomography (SPECT).
Analysts’ Views on MEA Technetium 99m Market:
Increasing organic strategies such as development of isotopes used for nuclear medicine applications. For instance, on January 9, 2023, BEST Cyclotron Systems Inc., the company which offers radioisotopes and production capabilities for nuclear medicine and radiotherapy with its range of cyclotron systems, announced that it has designed and installed a variety of cyclotrons for medical, industrial, and research applications ranging from energy 1 MeV to 70 MeV. The isotope Technetium-99m (Tc-99m), one of the isotopes most frequently utilized in nuclear medicine applications can be produced using one of these BEST cyclotrons, B-15, one of the isotopes most frequently utilized in nuclear medicine applications
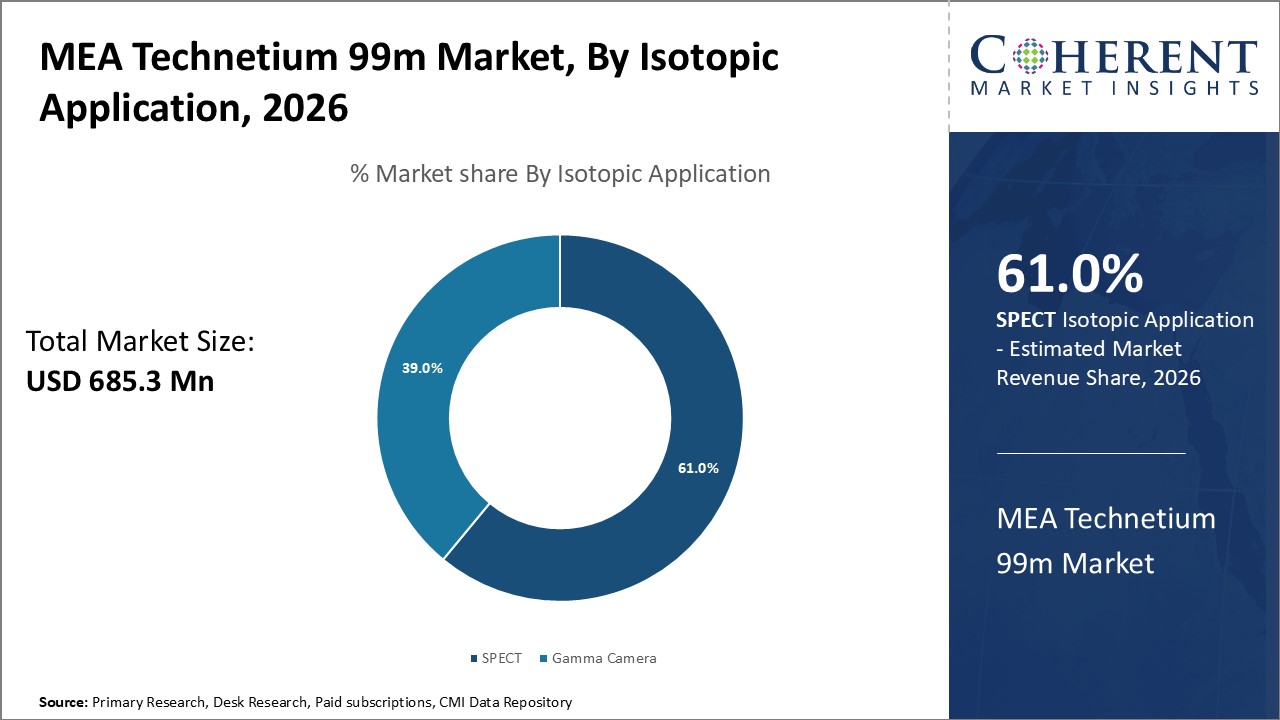
Figure 1. MEA Technetium 99m Market Share (%), by Isotopic Application, 2026
To learn more about this report, Request Free Sample
MEA Technetium 99m Market- Drivers
Expansion Of Nuclear Medicine Facilities And Advancements In Diagnostic Imaging
Increasing demand for Technetium 99m due to the expansion of nuclear medicine facilities and advancements in diagnostic imaging is expected to drive the MEA Technetium 99m Market growth by 2030. For instance, in 2019, Hamad Medical Corporation (HMC), the principal public healthcare provider in the State of Qatar, announced the opening of a radiopharmaceutical lab in Qatar. The new laboratory, located at HMC’s National Center for Cancer Care and Research (NCCCR), will provide cancer patients with diagnostic and therapeutic care locally, saving these patients the need to travel abroad for this type of care.
Increasing Adoption Of Inorganic Growth Strategies Such As Partnership
Major players in the market are focused on adopting partnership strategies to enhance their market share. For instance, in March 2020, NorthStar Medical Technologies, LLC, a commercialized nuclear medicine company, partnered with Capella Imaging, LLC, a BioGenerator company, under which NorthStar has licensed exclusive rights to FibroScint (Tc-99m F4A), a fibrin-targeted diagnostic imaging agent for cardiac imaging
Hospitals and healthcare facilities had to implement strict infection control measures and limit patient visits to minimize the risk of virus transmission. This led to a decrease in the number of routine diagnostic imaging procedures, which impacted the demand for technetium 99m and other radiopharmaceuticals used in nuclear medicine. Furthermore, supply chain disruptions and transportation restrictions caused challenges in the distribution and availability of Technetium 99m. The international movement of radioactive materials faced delays and logistical issues, affecting the timely delivery of Technetium 99m generators to healthcare facilities in the Middle East.
MEA Technetium 99m Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 685.3 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.6% | 2033 Value Projection: | USD 1,220.9 Mn |
| Segments covered: |
|
||
| Companies covered: |
GE Healthcare, IBA, Sumitomo Corporation, BWX Technologies, Inc., Advanced Cyclotron Systems, Inc., Navidea Biopharmaceuticals, Inc., BEST Cyclotron Systems Inc., NorthStar Medical Radioisotopes, and Siemens Healthineers. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
MEA Technetium 99m Market Segmentation:
The MEA Technetium 99m Market report is segmented into isotopic application, end user, and country
Based on Isotopic Application, the market is segmented into gamma camera and SPECT. Out of which, the SPECT segment is expected to hold a dominant position in the MEA Technetium 99m Market during the forecast period, and this is attributed because of the three-dimensional imaging technique that provides cross-sectional views of radioisotope distribution within the body.
Based on End User, the market is segmented into hospitals and diagnostic Centers. Out of which, the Hospitals segment is expected to dominate the market over the forecast period and this is attributed because of the wide range of nuclear medicine services
Based on Country, the market is segmented into Saudi Arabia, Algeria, Bahrain, Cyprus, Egypt, Iran, Iraq, Israel, Jordan, Kuwait, Lebanon, Libya, Morocco, Mauritania, Oman, Palestine, Qatar, Syria, Tunisia, Turkey, and Yemen.
MEA Technetium 99m Market: Key Developments
In March 2020, Navidea Biopharmaceuticals, Inc., one of the leaders in precision medicine with immuno-targeted products, received the U.S. Patent extension for Lymphoseek (technetium [Tc 99m] tilmanocept), a radioactive diagnostic agent which accumulates in lymphatic tissue and selectively binds to mannose-binding receptors (CD206) located on the surface of macrophages and dendritic cells, until 2026.
In 2020, the UAE's Abu Dhabi Health Services Company (SEHA), a healthcare network established a centralized radiopharmacy facility, which included Technetium 99m production capabilities. The facility aimed to ensure the availability of radiopharmaceuticals, including Technetium 99m, for healthcare facilities across the UAE.
In February 2020, BWX Technologies, Inc., one of the leading suppliers of nuclear components and fuel successfully labeled technetium-99m derived from its proprietary molybdenum-99 production process and its proprietary Tc-99m generators with cold kits.
In November 2019, researchers from the Institute of Nuclear and Radiological Sciences and Technology, Greece, reported the synthesis and characterization of two novel derivatives of undecanonoic and hexadecanonoic acid with the aim of developing 99mTc-labeled fatty acids intended for myocardial metabolism imaging.
MEA Technetium 99m Market: Key Trends
Increasing facility expansion by the research centers
Increasing research and development activities is expected to drive the market growth over the forecast period. For instance, in 2020, Saudi Arabia's King Faisal Specialist Hospital & Research Centre (KFSH&RC) collaborated with a South Korean company to establish a radiopharmaceutical production facility in the country. The facility aimed to produce Technetium 99m locally and reduce dependence on imports.
MEA Technetium 99m Market: Restraint
Shutting Down of Major Producing Reactors And Adoption of Alternative Imaging Modalities
Availability of 99mTc depends on the production of Mo-99 from the small number of nuclear reactors worldwide. The major producing reactors have already aged and are on the verge of shutting down. This creates an issue related to production disruption assuming the need for extended shutdowns for planned or unplanned maintenance work, which is expected to hinder the growth of the MEA technetium-99m market.
Adoption of alternative imaging modalities is also expected to limit the growth of the market. The expected shortage of Tc-99m in the near future has led to an increase in the adoption of PET or other imaging modalities.
MEA Technetium 99m Market- Key Players
Major players operating in the MEA Technetium 99m Market include GE Healthcare, IBA, Sumitomo Corporation, BWX Technologies, Inc., Advanced Cyclotron Systems, Inc., Navidea Biopharmaceuticals, Inc., BEST Cyclotron Systems Inc., NorthStar Medical Radioisotopes, and Siemens Healthineers.
*Definition: Technetium-99m (Tc-99m) is a radioactive isotope of technetium, a chemical element that is widely used in nuclear medicine imaging procedures due to its favorable properties.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients