Metal Non-Covered Biliary Stent Market is estimated to be valued at USD 423.2 Mn in 2026 and is expected to reach USD 587.6 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 4.8% from 2026 to 2033.
Analysts’ Views on Global Metal Non-covered Biliary Stent Market:
Key market players are investing in research and development and commercialization of products for treatment of biliary disorders, and this is expected to drive the global metal non-covered biliary stent market growth during the forecast period. Rising geriatric population lead to the increase in the surgeries and increasing incidence of biliary duct disorders and increasing research and development funding are also expected to drive the market growth. For instance, in October 2022, according to the World Health Organization (WHO), one in six individuals globally will be 60 or older by 2030. By 2030, there will be 1.4 billion people over the age of 60 indicating increase from1 billion in 2020. The number of individuals in the world who are 60 or older will double to 2.1 billion by 2050.
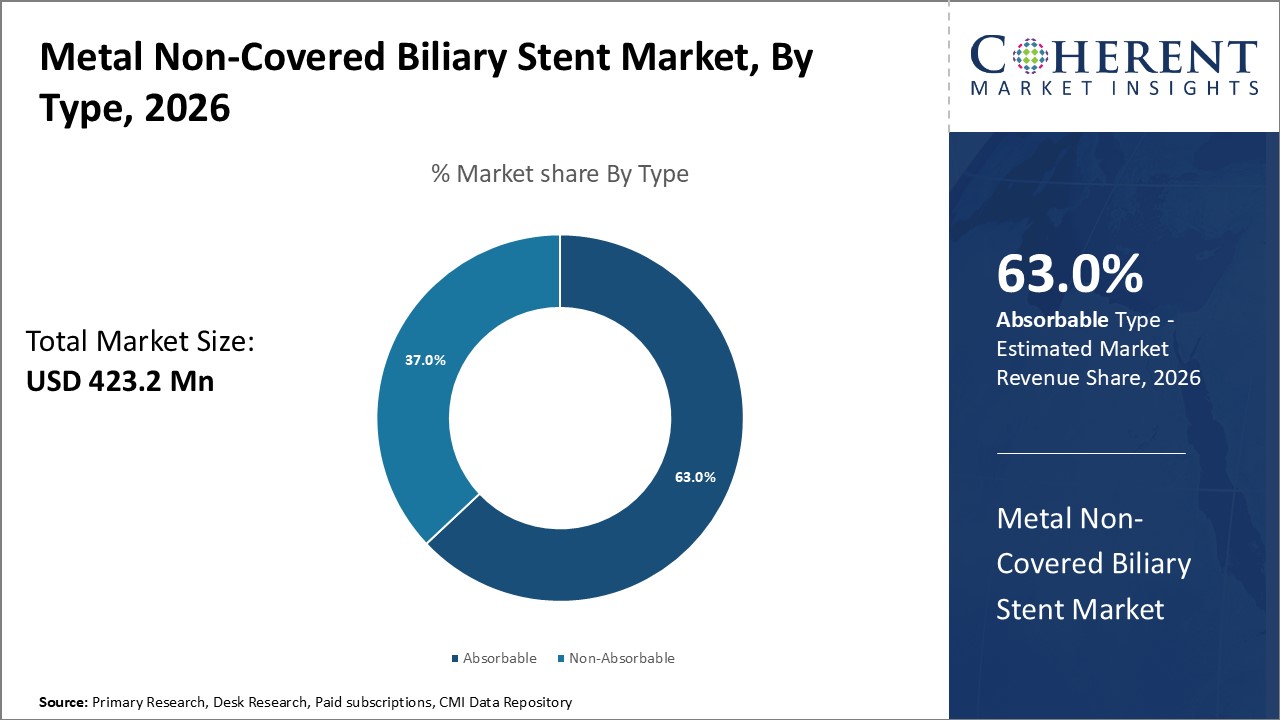
Figure 1. Global Metal Non-covered Biliary Stent Market Share (%), By Type, 2026
To learn more about this report, Request Free Sample
Global Metal Non-covered Biliary Stent Market– Drivers
Increasing Prevalence of Biliary Diseases
Increasing prevalence of biliary diseases is expected to drive the global metal non-covered biliary stent market growth over the forecast period. For instance, in June 2022, according to the National Cancer Institute, in U.S, the rate of new cases of liver and intrahepatic bile duct cancer from 2020 was 9.3 per 100,000 men and women per year in 2020. The death rate was 6.6 per 100,000 men and women every year. According to the source in 2023, it is estimated that there will be 41,210 new cases of liver and intrahepatic bile duct cancer and 29,380 deaths due to liver and bile duct cancer in U.S.
Technological Advancements
Increasing technological advancement in the detection and treatment of gastrointestinal diseases such as bile duct obstruction that provides several advantages such as lesser chances of infection, quicker recovery, and shorter hospital stays is expected to drive the market growth over the forecast period. For instance, in June 2020, Cook, a medical device company, announced the launch of two products named transluminal biliary biopsy forceps set (BBFS) and a new 40-centimeter delivery system line extension of the existing Zilver 635 Biliary Stent (ZIB), and both are commercially available in the U.S. and Canada. The Zilver 635 Biliary Stent is used in cases of abnormal constrictions of the biliary tree called biliary neoplasms.
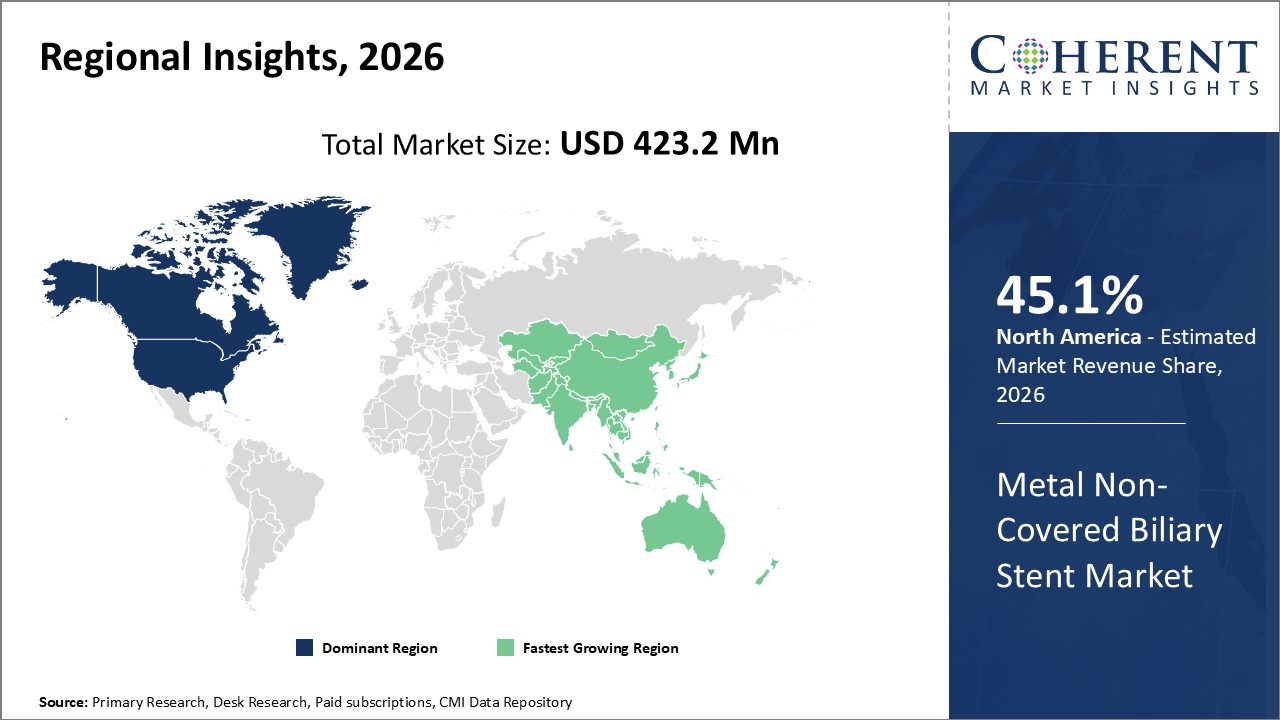
Figure 2. Global Metal Non-covered Biliary Stent Market Share (%), By Region, 2026
To learn more about this report, Request Free Sample
Global Metal Non-covered Biliary Stent Market- Regional Analysis
Among region, North America dominated the global metal non-covered biliary stents market, owing to increasing number of pancreatic and bile cancers among the population. For instance, in June 2021, according to the American Cancer Society, pancreatic cancer accounted for approximately 3% of all cancers in the U.S and approximately 7% of all cancer deaths in 2020. The presence of advanced healthcare infrastructure coupled with the presence of advanced technology for the diagnosis and treatment is expected to boost demand for the metal non-covered biliary stents in North America.
Although procedure volume and chance of training for fellows were decreased, the number of GI endoscopy centers that still offer ERCP to treat biliary blockage brought on by cholangitis is on the decline.
Metal Non-covered Biliary Stent Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 423.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.8% | 2033 Value Projection: | USD 587.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AMG International, BD (Becton Dickinson and Company), Boston Scientific Corporation or its affiliates, Cook, Merit Medical Systems, Pnn Medical A/S, TAEWOONG, Teleflex Incorporated, CONMED Corporation, Olympus Corporation, and AbbottAMG International, BD (Becton Dickinson and Company), Boston Scientific Corporation or its affiliates, Cook, Merit Medical Systems, Pnn Medical A/S, TAEWOONG, Teleflex Incorporated, CONMED Corporation, Olympus Corporation, and Abbott |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Metal Non-covered Biliary Stent Market- Segmentation
Global metal non-covered biliary stent market report is segmented into type, application, end user, and region.
Among Type, the global metal non-covered biliary stent market is segmented into absorbable and non-absorbable. Out of which, absorbable segment is expected to dominate the global metal non-covered biliary stent market during the forecast period due to the immediate technical and clinical success rates of absorbable metal biliary stents .
Among Application, the global metal non-covered biliary stent market is segmented into malignant obstruction, biliary leaks, benign biliary strictures, and others. Out of which, benign biliary strictures segment is expected to dominate the market over the forecast period due to the prevalence of biliary strictures due to chronic pancreatitis.
Among End User, the global metal non-covered biliary stent market is segmented into hospitals, clinics, and ambulatory surgical centers. Out of which, hospitals segment is expected to dominate the market over the forecast period due to the prevalence of biliary diseases and pancreatic cancer that leads to hospitalization.
Among Region, the global metal non-covered biliary stent market is segmented into North America, Latin America, Europe, Asia Pacific, Middle East, and Africa. Out of which, the North America is expected to dominate the market over the forecast period, owing to increasing prevalence of cervical cancer and biliary diseases, and various initiatives by the government regarding awareness about the biliary duct cancer.
Among all segmentation, the application segment has the highest potential due to rising prevalence of biliary strictures due to chronic pancreatitis over the forecast period. For instance, in February 2021, according to a study published by Journal of United European of Gastroenterology, the average incidence rate of chronic pancreatitis in 2020 was, 12.6 per 100,000 years per person for the general population, 8.6 per 100,000 person-years for women, and 16.7 per 100,000 person-years for men.
Global Metal Non-covered Biliary Stent Market- Cross Sectional Analysis
Among type, the absorbable segment held a dominant position in North America region over the forecast period due to increasing number of biliary diseases patients that includes gall bladder stones, pancreatitis, and others and this is owing to the increasing biliary duct disorder cases and the absorbable stents provide good reliability for the treatment. For instance, in March 2022, according to an article published by JAWA Network, approximately 20 million people in the US. Acute cholecystitis is diagnosed in approximately 200,000 people in the U.S. every year.
Global Metal Non-covered Biliary Stent Market- Key Developments
In May 2021, Cook, a medical device company that develops, manufactures, and distributes devices for minimally invasive medicine, acquired Cantel Medical, a global company that provides innovative infection prevention products and services for patients, to expand its biliary stent and another product portfolio to provide services to customers globally. Thus, such development in the region is likely to boost demand for pancreatic and biliary stents.
On February 24, 2023, Olympus Corporation, a global medtech company that makes people’s lives healthier, safer, and more fulfilling, acquired Taewoong Medical Co., Ltd, (Taewoong Medical), a manufacturer of medical devices such as gastrointestinal (GI) metallic stents1, to strengthen its GI EndoTherapy product portfolio capabilities, and thus, improving patient outcome through comprehensive solutions. Taewoong Medical will receive approximately US$ 370 million in cash, out of which, US$ 255.5 million will be paid at the closing and up to US$ 114.5 million will be paid if future milestones is achieved.
In May 2021, AMG International, a wholly owned subsidiary Q3 Medical Devices Limited (Q3), an Ireland-based company which deals with the innovation of biodegradable implants and micro invasive technology, further established its leadership role in the fast-paced development of novel biodegradable implants. The company announced that it had received CE Mark approval for its second fully biodegradable product named UNITY-B balloon expandable biodegradable biliary stent (BEBS) for endoscopic use. The UNITY-B will compliment ARCHIMEDES, the world's first CE approved pancreatico biliary biodegradable implant.
In May 2020, Q3 Medical Group, a medical device company entered a distribution agreement with Medtronic, a medical device company for its AECHIMEDES, a biodegradable biliary and pancreatic stent. This stent is specially designed as an alternative to the traditional plastic biliary stents. The aim behind this six-year exclusive agreement is to distribute ARCHIMEDES biodegradable stent in the western EU, Japan, and the U.S. with rights to other regions.
Global Metal Non-covered Biliary Stent Market- Key Trends
Partnership by the Key Players
Key players are focusing on collaboration as well as partnership, for the market growth over the forecast period. In July 2022, Micro-Tech Endoscopy, a global manufacturer of GI endoscopy devices and non-vascular stents, announced a distribution partnership with Thoracent, Inc., an innovator in pulmonology. Thoracent, Inc. will be the U.S. distributor of Micro-Tech's Y-Shaped tracheal biliary stent system, a flexible, self-expanding device with an innovative over-the-wire delivery system that is designed to aid the management of airway tumors in the trachea without invasive surgery.
Global Metal Non-covered Biliary Stent Market: Restraints
High cost of Biliary Stent Insertion Procedures
Certain factors such as high cost of biliary stent insertion procedures is expected to hinder the market growth over the forecast period. So, low cost plastic stents can be used for the treatment. For instance, in August 2021, according to a study published by Journal of Endoscopy International Open, an average cost of the metal non-covered biliary stent procedures for the treatment of biliary obstruction is US$ 9,304 but has more accuracy as compared to the plastic stent.
Global Metal Non-covered Biliary Stent Market- Key Players
Major players operating in the global metal non-covered biliary stent market include AMG International, BD (Becton Dickinson and Company), Boston Scientific Corporation or its affiliates, Cook, Merit Medical Systems, Pnn Medical A/S, TAEWOONG, Teleflex Incorporated, CONMED Corporation, Olympus Corporation, and Abbott.
Definition: A biliary metal stent is a flexible metallic tube that is specially designed to hold bile duct open, which has been blocked or partially blocked. When a blockage occurs in the bile duct, fluids like bile (bilirubin) are unable to flow into the duodenum to aid in digestion. Metal stents have a larger diameter with a lower risk of stent occlusion.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients