The global methicillin-resistant staphylococcus aureus (MRSA) drugs market size is expected to reach USD 1,267 Mn in 2026 and is projected to grow at a compound annual growth rate (CAGR) of about 4.3% during the forecast period (2026-2033), surpassing USD 1,705 Mn by 2033. This growth is attributable to rising prevalence of drug-resistant infections, growing awareness regarding antimicrobial resistance, and continuous development of novel MRSA antibiotics and combination therapies aimed at combating methicillin-resistant Staphylococcus aureus (MRSA).
|
Current Event |
Description and its Impact |
|
Global and National AMR Policy Strengthening |
|
|
Stringent Antimicrobial Regulatory Proposals |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
As per Coherent Market Insights’ latest methicillin-resistant staphylococcus aureus (MRSA) drugs market analysis, glycopeptides and lipoglycopeptides remain the most widely used drug class for MRSA, accounting for a revenue share of 36% in 2026. This strong market preference reflects both clinical efficacy and guideline recommendations.
Glycopeptides, such as vancomycin, and lipoglycopeptides, like dalbavancin and oritavancin, work by blocking bacterial cell wall synthesis. This makes them very effective against serious infections, including bloodstream infections, skin and soft tissue infections, and complicated hospital-acquired infections. They are being widely used for Methicillin-resistant Staphylococcus Aureus treatment because of their proven efficacy against resistant strains of Staphylococcus aureus, including those unresponsive to conventional beta-lactam antibiotics.
For instance, dalbavancin, a long‑acting lipoglycopeptide, has been effective in real-world use for complex bone and joint infections, including cases with previous treatment failures. It has achieved high clinical cure rates even outside the traditional indications for MRSA‑associated infections. In addition, these drugs are considered safe, come in both intravenous and long-acting forms, and are recommended in global treatment guidelines, which has made them widely used by healthcare providers.
To learn more about this report, Request Free Sample
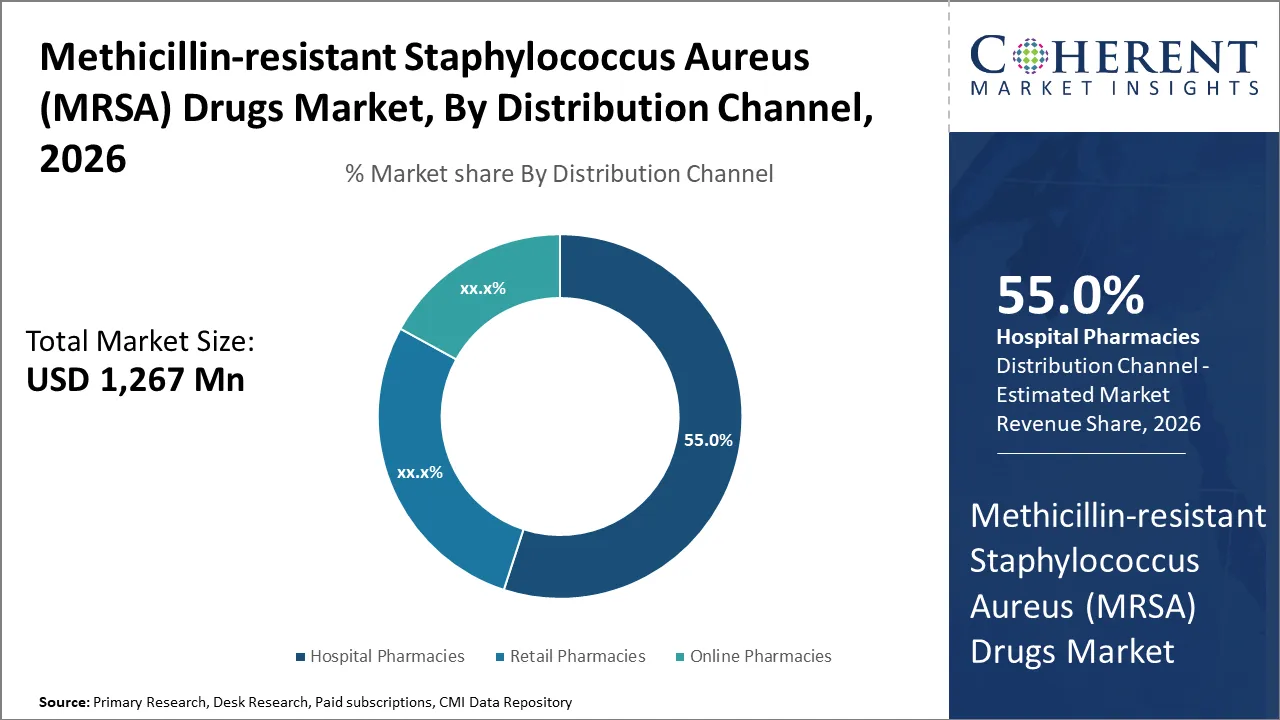
Based on distribution channel, hospital pharmacies segment is expected to account for the largest market share of 55% in 2026. This is mostly due to high number of MRSA patients admitted to hospitals, where these drugs are essential for treatment. Hospitals provide easy access to advanced medications and ensure proper administration under medical supervision. This makes hospital pharmacies the preferred distribution channel for MRSA infection drugs.
In a large surveillance study of MRSA bacteremia in the U.S., more than 50% of cases were classified as healthcare‑associated (i.e., hospital or post‑hospital onset) across multiple years of data. This indicates that most serious MRSA infections requiring systemic antibiotic treatment originated in or were linked to hospital care. As a result, majority of methicillin-resistant staphylococcus aureus (MRSA) drug sales are slated to remain concentrated in hospital pharmacies.
The Methicillin-resistant Staphylococcus Aureus (MRSA) drugs market is experiencing strong growth due to innovations in next‑generation antibiotics and targeted therapies. New antibiotics, such as long-acting lipoglycopeptides like dalbavancin and oritavancin, and improved oxazolidinones, are providing more effective treatment options against resistant MRSA strains. These drugs offer easier dosing, better safety, and the potential to reduce hospital stays.
For instance, in May 2025, Innoviva Specialty Therapeutics, Inc. commercially launched ZEVTERA (ceftobiprole) in the United States. ZEVTERA is an advanced‑generation cephalosporin antibiotic approved to treat three types of bacterial infections. It works by killing bacteria that are resistant to many other antibiotics, including MRSA.
Similarly, rapid molecular diagnostic tools allow doctors to quickly identify MRSA infections and start targeted therapy sooner, improving patient outcomes as well as reducing complications. These advancements are driving the MRSA drugs market by addressing the critical challenge of antibiotic resistance.
Due to the increase in multidrug-resistant MRSA strains, combination therapy is becoming more common in clinical practice and influencing market trends. Using β-lactam antibiotics together with established MRSA drugs like daptomycin or vancomycin has shown better bacterial clearance and less chance of resistance, especially in complicated bloodstream infections or pneumonia. This approach boosts antibacterial effects and helps maintain the effectiveness of existing medications.
A large retrospective cohort study published found that adults with MRSA bloodstream infections treated with daptomycin plus a β lactam had significantly lower odds of composite clinical failure than standard monotherapy. This benefit was driven by reductions in persistent or recurrent bacteremia.
To learn more about this report, Request Free Sample
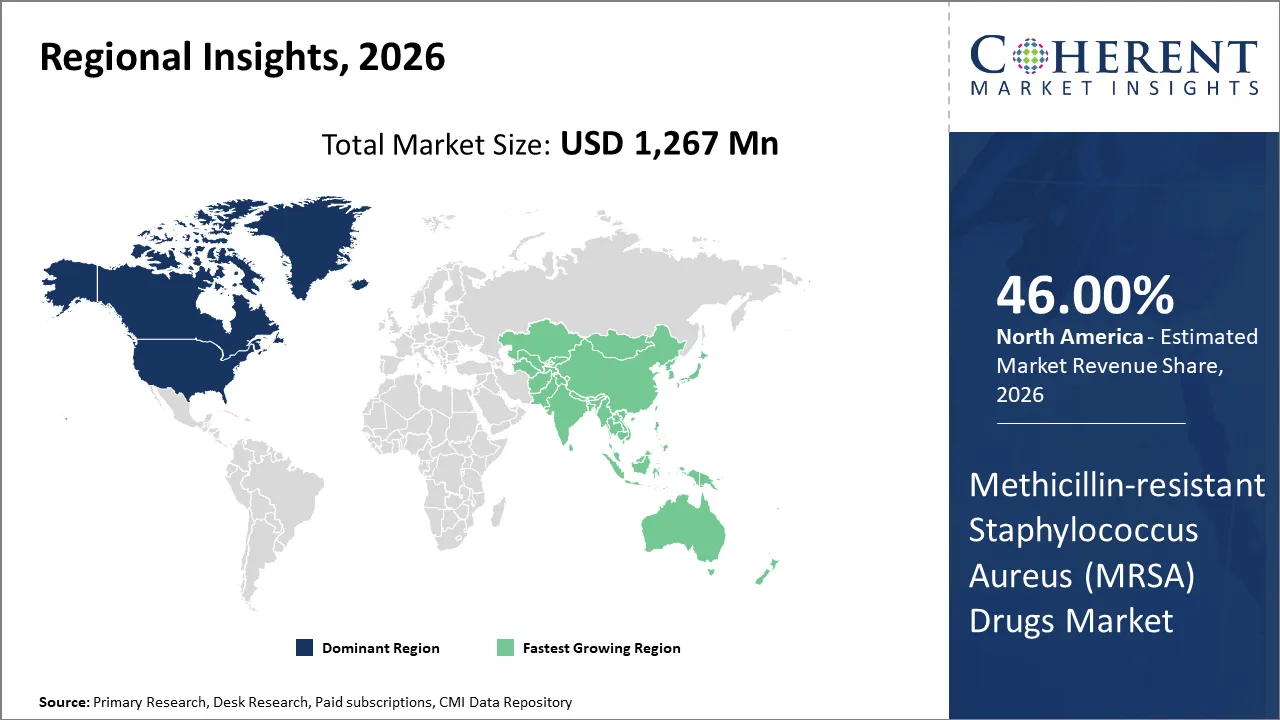
North America is slated to lead the global methicillin resistant staphylococcus aureus (MRSA) drugs market, holding a share of 46% in 2026. This is attributable to high infection burden, well established healthcare infrastructure, extensive clinical research, and strong antimicrobial stewardship programs. A 2025 study by the U.S. Centers for Disease Control and Prevention reports that about 2% of people carry MRSA, with higher rates in some groups. Advanced diagnostic capabilities and high healthcare spending in the U.S. and Canada help drive consistent demand for effective MRSA treatments.
A major driver in this region is the high rate of hospital-acquired MRSA infections, which account for a large portion of healthcare-associated infections in acute care settings. This creates steady demand for specialized MRSA treatments. At the same time, increasing awareness of community-acquired MRSA and the broader use of infection control measures in hospitals and outpatient clinics are helping the MRSA drugs market grow in North America.
In recent years, several pharmaceutical companies across North America have advanced MRSA drug pipelines. For example, in October 2023, Basilea Pharmaceutica submitted a new drug application (NDA) to the U.S. FDA for indications including MRSA related bacteremia and skin infections, reflecting intensified R&D focus. This trend toward novel agents and expanded indications is expected to further bolster regional market growth through the forecast period.
Asia Pacific region is anticipated to emerge as a central hub for methicillin resistant staphylococcus aureus drug manufacturers, representing a global market share of 20% in 2026. This can be attributed to expanding healthcare infrastructure, increasing prevalence of antibiotic resistant infections, and rising public and clinical awareness of antimicrobial resistance challenges. In addition, increasing approval of latest drugs for MRSA infections is expected to foster market growth in China.
China, India, and Southeast Asian nations face significant MRSA burdens in both hospital and community settings due to factors like high population density and variable infection control practices. Local efforts to strengthen laboratory surveillance and antibiotic stewardship are contributing to improved diagnosis and treatment pathways, which in turn support market growth for MRSA therapeutics.
A key trend driving growth in Asia Pacific is the escalating prevalence of MRSA and emphasis on surveillance initiatives. Epidemiological studies report that MRSA remains a common cause of nosocomial and community infections across the region, with prevalence rates in some Southeast Asian countries ranging significantly due to diverse healthcare settings and infection control capacities.
For example, a 2022 review of MRSA genotypes in Southeast Asia reported MRSA infection prevalence in the region generally between ~20%–30% in studies reviewed. Governments and healthcare systems are increasingly implementing national antimicrobial resistance monitoring programs to better track MRSA trends, which supports broader adoption of appropriate drug therapies.
The U.S. remains the market leader in methicillin-resistant staphylococcus aureus drugs, attributed to strong healthcare infrastructure, advanced diagnostic capabilities, high healthcare spending, and presence of leading pharmaceutical companies. Rising incidence of hospital-acquired and community-acquired MRSA infections is driving demand for effective therapeutics.
MRSA causes a high number of serious infections and deaths in the U.S. annually. For instance, according to the U.S. Centers for Disease Control and Prevention (CDC), MRSA is responsible for more than 70,000 severe infections and 9,000 deaths per year in the United States. This high infection burden is fueling demand for methicillin-resistant staphylococcus aureus (MRSA) drugs in the nation.
A key trend in the U.S. MRSA therapeutics market is the development and approval of novel therapeutics targeting resistant strains. U.S.-based companies are developing new drugs for MRSA-related bacteremia and skin infections. Such innovations, combined with increasing hospital focus on infection control and patient safety, are expected to drive adoption of advanced MRSA treatments across hospitals, clinics, and outpatient care centers.
The methicillin-resistant staphylococcus aureus drugs market in China is projected to grow rapidly due to rising awareness of antibiotic resistance, increasing hospital and surgical volumes, and government initiatives to strengthen antimicrobial stewardship. Rising prevalence of healthcare-associated infections is also fueling demand for effective MRSA therapeutics.
Epidemiological studies in China indicate that methicillin‑resistant Staphylococcus aureus (MRSA) accounts for a significant portion of S. aureus isolates in hospital settings, with prevalence reported between approximately 28% and 40%. This underscores the continued need for therapies specifically effective against MRSA infections.
A major growth driver in China is the expansion of hospital infection control programs and antibiotic monitoring. Hospitals are increasingly implementing strict infection prevention measures and adopting advanced MRSA treatment protocols. In addition, pharmaceutical companies are introducing locally produced and cost-effective MRSA drugs that meet international quality standards, improving access in both urban and semi-urban areas. Rising medical awareness and government support for antimicrobial resistance programs are expected to sustain strong market growth throughout the forecast period.
Some of the major players in methicillin-resistant staphylococcus aureus (MRSA) drugs market are Merck & Co., Inc., Pfizer Inc., AbbVie Inc., Teva Pharmaceutical Industries Ltd., Melinta Therapeutics, Inc., Cumberland Pharmaceuticals Inc., Nabriva Therapeutics Plc., Paratek Pharmaceuticals, Inc., Debiopharm International SA., Innovation Pharmaceuticals Inc., and Basilea Pharmaceutica Ltd.
Top industry players are heavily investing in research and development to create advanced treatment for Methicillin-resistant Staphylococcus Aureus infections. They are also adopting inorganic strategies like mergers, partnerships, acquisitions, and collaborations to expand their regional presence and gain a competitive edge over the market. For instance,
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,267 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.3% | 2033 Value Projection: | USD 1,705 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Merck & Co., Inc., Pfizer Inc., AbbVie Inc., Teva Pharmaceutical Industries Ltd., Melinta Therapeutics, Inc., Cumberland Pharmaceuticals Inc., Nabriva Therapeutics Plc., Paratek Pharmaceuticals, Inc., Debiopharm International SA., Innovation Pharmaceuticals Inc., and Basilea Pharmaceutica Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients