Global personalized cell therapy market is estimated to be valued at USD 21.95 Bn in 2026 and is expected to reach USD 103.51 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 24.8% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
Advancements in cell and gene therapy coupled with increasing investment by key players can drive the market growth. Global personalized cell therapy market is expected to witness significant growth over the forecast period due to factors such as rising prevalence of cancers and genetic disorders, increasing demand for targeted therapies and development of novel advanced therapies. Improvements in healthcare systems worldwide can also drive the market growth.
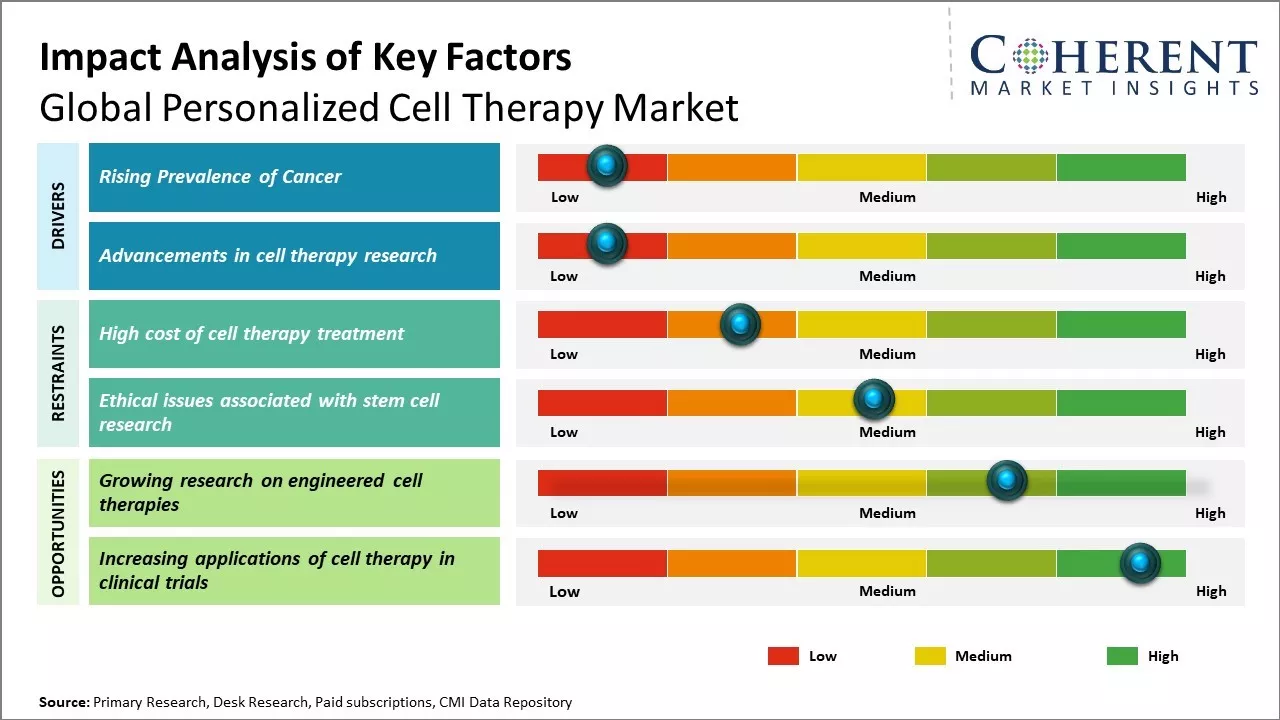
Rising Prevalence of Cancer
Rising prevalence of cancer around the world can drive the global personalized cell therapy market growth. This increase in cancer incidence has boosted investments in developing advanced treatment methods like personalized cell therapy. In personalized cell therapy, a patient’s own immune cells are engineered to target the specific cancer type and mutation. These living drugs are then infused back into the patient to fight the cancer in a personalized way. Compared to traditional chemotherapy, targeted cell therapies have shown promising results with fewer side effects. Many such therapies have recently received regulatory approvals to treat various blood cancers like lymphoma and leukemia. Researchers are also working on customizing these living cell therapies for solid tumors like lung cancer, breast cancer, and others. Growing success and efficacy of cell therapies in clinical trials and first approvals have boosted confidence among the medical community, patients as well as investors. Biotech and pharmaceutical companies are now dedicating more resources to develop the next generation of personalized cell therapies at a larger scale.
For instance, according to the data published by World Health Organization in February 2025, according to data by The International Agency for Research on Cancer (IARC), ahead of World Cancer Day, released new estimates showing a significant global burden of cancer in 2022. There were approximately 20 million new cases and 9.7 million deaths from cancer worldwide. The data highlights disparities, with underserved populations facing a disproportionate impact. Lung, breast, and colorectal cancers were the most common types globally, with lung cancer leading in both new cases and deaths. Inequities in cancer care financing were also evident, with many countries not adequately covering essential cancer and palliative care services as part of universal health coverage.

Get actionable strategies to beat competition: Request Free Sample
Advancements in cell therapy research
Advancements in cell therapy research can transform medicine and treat previously incurable diseases. Scientists are making progress in developing customized treatments that use a patient's own cells to fight various life-threatening illnesses. This pioneering work in personalized cell therapies can drive the market growth. Researchers have had some early successes using cells from bone marrow, blood and skin to treat cancers like leukemia and lymphomas. For example, stem cell therapies are being tested for conditions like heart disease, diabetes, Parkinson's and Alzheimer's with hope that damaged or dead cells can be replaced. The pandemic has also accelerated vaccine research using cell culture technology paving way for new strategies against various viruses.
As the science advances, more cell therapy clinical trials are planned for a wider range of health issues. According to World Health Organization, 500 trials studying over 25,000 patients was conducted by 2023 across key markets of U.S., Europe, China, and India. The data demonstrates growing interest from pharmaceutical companies and research organisations to develop these next-generation treatment options. As trials show success, regulatory approvals will increase bringing novel therapies to patients faster and increasing investments in this sector.
Key Takeaways from Analyst:
Global personalized cell therapy market growth is driven by rising prevalence of cancer and other chronic diseases worldwide r as cell therapies are increasingly used for disease treatment. Advancements in cell therapies such as CAR T-cell therapy and growing understanding of their mechanisms can expand applications to new disease areas. Younger patient demographics are more open to novel treatment options and see cell therapies as less invasive compared to other alternatives.
High costs and lack of treatment standardization can hamper the market growth. Manufacturing complexities and costs associated with developing personalized therapies puts them out of reach for most patients. Absence of treatment protocols means clinical efficacy can vary greatly. North America dominates the market due to strong regional presence of leading players and availability of late-stage clinical trial data. Asia Pacific is poised to be the fastest growing region due to rising healthcare investments, emerging economies and growing biotech industry in countries like China and India.
Market Challenges: High cost of cell therapy treatment
The high cost of cell therapy treatment can hamper the global personalized cell therapy market growth. Cell therapy involves complex manufacturing processes that require specialized equipment and facilities. This makes the treatment expensive, putting it out of reach for many patients. Developing personalized therapies that precisely meet an individual patient's needs makes the process even more complex and costly. Currently, the average cost of a single dose of cell therapy ranges from US$ 300,000 to US$ 500,000 in most countries. Some of the most advanced personalized cell therapies can cost over US$ 1 million per patient.
While these treatments have shown promising results for curing various terminal diseases, the high price tag means only a small fraction of patients who could potentially benefit are able to access them. Insurance coverage for these novel treatments is still limited in many parts of the world as these are still considered experimental. This financial burden often has to be borne by the patient themselves. High treatment costs also restrict patient access in developing nations with limited healthcare budgets. In 2021, according to the report by the United Nations Development Programmed, more than half of world's population does not have full coverage of essential health services. These lack access to advanced therapies due to their countries' inability to afford them.
Market Opportunities: Growing research on engineered cell therapies
Engineered cell therapies have become a promising area of medical research globally in recent years. With continuous advancement in stem cell biology and gene engineering, researchers are now able to develop personalized cell therapies that can cure diseases that were considered incurable until now. One of the most prominent examples is use of chimeric antigen receptor T cell (CAR-T) therapy in treating some types of cancers. In CAR-T therapy, a patient's own T cells are engineered to recognize and attack cancer cells. This therapy has shown tremendous potential in treating certain leukemias and lymphomas with remission rates as high as 90% in clinical trials. As the research progresses, many new cell therapy candidates are under development for a wide range of chronic and degenerative conditions such as heart disease, diabetes, Parkinson's disease, and arthritis. Scientists are engineering various cell types like stem cells, cartilage cells, insulin-producing cells and brain cells through genetic modification and cocooning them in biomaterials to effectively regenerate damaged tissues and restore organ function in the body. For instance, in 2020, according to a study published by Stem Cell Reviews and Reports Journal, regenerative trials using engineered mesenchymal stem cells coated in biomaterials showed promising results in repairing knee cartilage for osteoarthritis patients.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
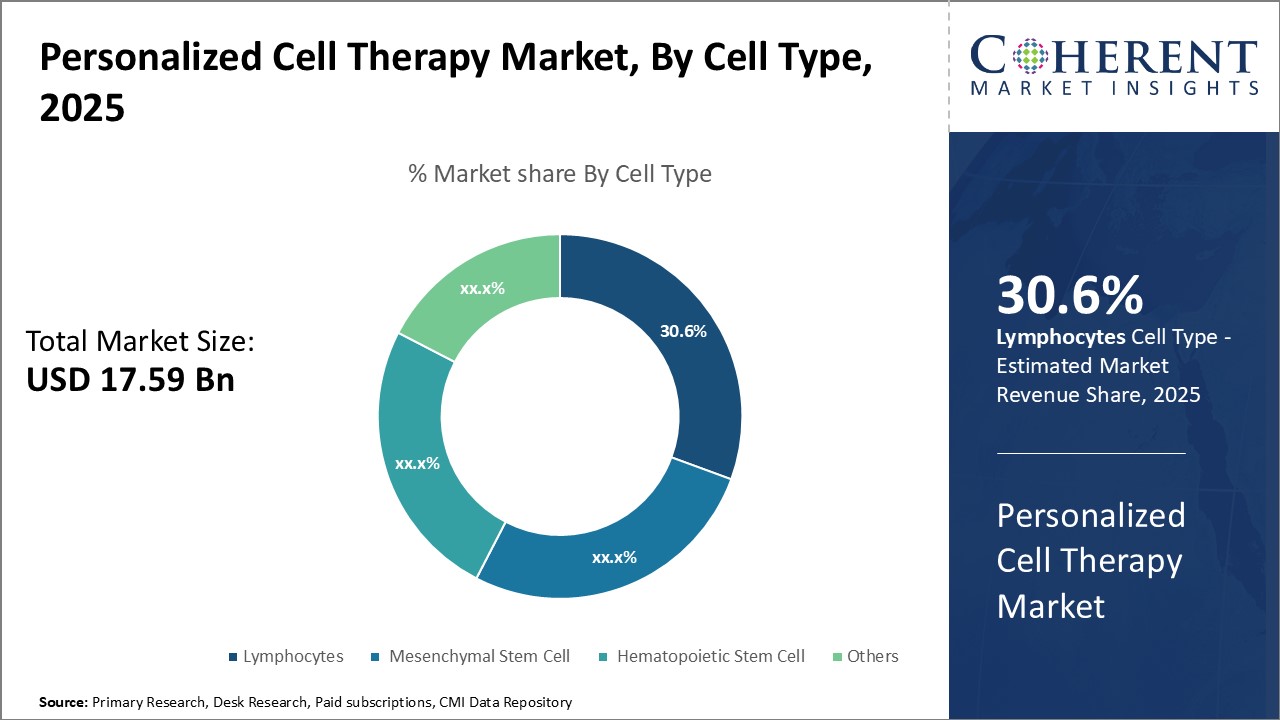
By Cell Type - Immunotherapy drives the lymphocyte segment growth
In terms of cell type, lymphocytes segment is estimated to contribute the highest market share of 31.6% in 2026, owing to advancements in immunotherapy techniques. Lymphocytes include T cells and B cells which play a crucial role in identifying and destroying abnormal cells. Technological developments have enabled researchers to culture and grow these cells outside the body and reintroduce them after modifying to target specific diseases. Chimeric antigen receptor T-cell (CAR-T) therapy is a prominent example, where T cells are extracted from patients, engineered to include receptors that recognize tumor antigens, and infused back to trigger an immune response against cancerous cells. This revolutionary cell therapy I can be used for treating certain blood cancers and represents a major breakthrough for personalized care. Growing clinical evidence of lymphocyte therapy success along with its targeted approach with fewer side effects compared to other alternatives can drive the segment growth.
To learn more about this report, Request Free Sample
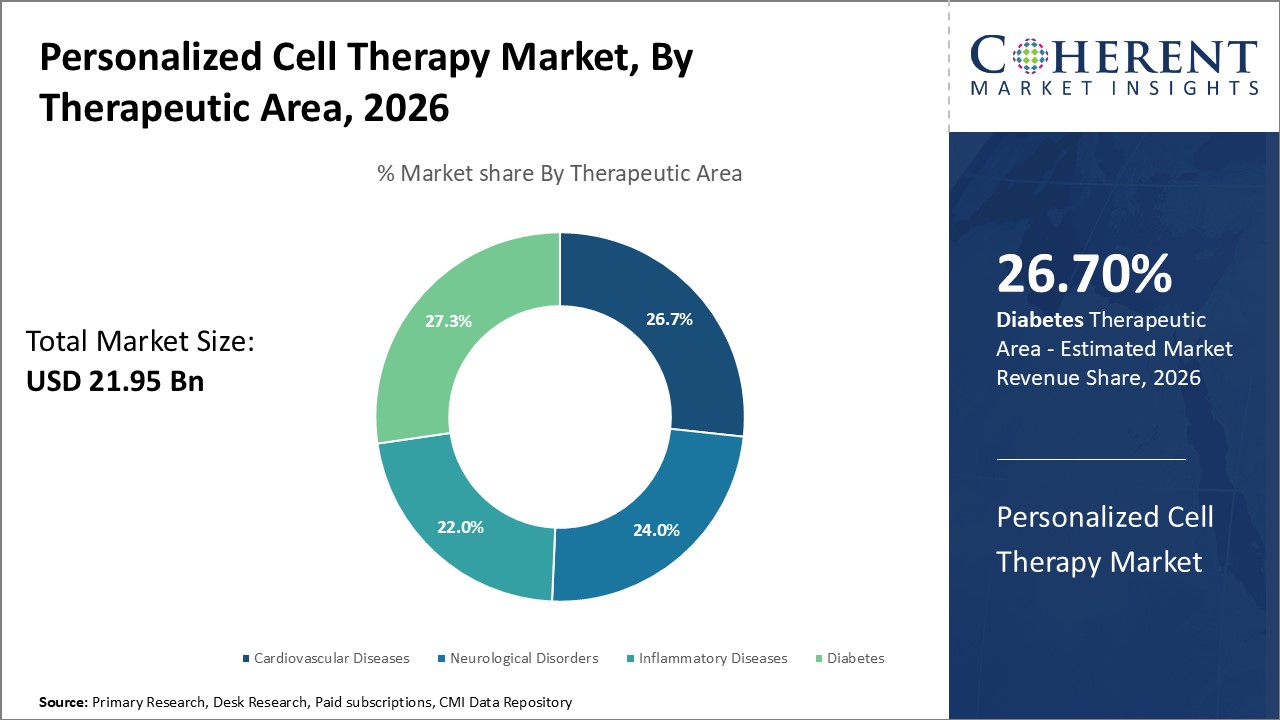
By Therapeutic Area - Neurogenesis stimulates interest in neurological disorders segment
In terms of therapeutic area, neurological disorders segment is estimated to contribute the highest market share of 26.7% in 2026, owing to lack of effective treatments for conditions like Alzheimer's, Parkinson's, spinal cord injuries and growing interest in tissue regeneration approaches. Stem cell therapy is actively explored as it holds potential to reduce neural damage, stimulate neurogenesis and aid regeneration of neural networks in the brain and spinal cord. Mesenchymal stem cells extracted from bone marrow or adipose tissue secrete neurotrophic factors and anti-inflammatory cytokines upon administration in the body. Preclinical research indicates these cells can modulate immune response, promote angiogenesis around sites of injury, and support growth of new neurons. Several clinical trials are underway to evaluate efficacy and safety profile of stem cell therapy for neurological conditions. Induced pluripotent stem cell (iPSC) technology enables generation of patient-specific neurons and glial cells in vitro, which represents an attractive avenue for developing personalized cell therapies with potential to slow or even reverse disease progression.
By End Users- Hospital expertise drives market share in healthcare institutions.
In terms of end-users, hospitals segment is estimated to contribute the highest market share of 41% in 2026, owing to their expertise in handling complex cell-based therapies. Hospitals have advanced cell processing facilities, well-trained healthcare staff adept at cell culture, cryopreservation and controlled reintroduction techniques. These can also effectively manage adverse events, closely monitor patients, and submit comprehensive data for regulatory approvals and research publications. Most clinical trials involve hospital-based administration to leverage their experience and infrastructure for cell therapy research. Additionally, hospitals provide multi-disciplinary teams of oncologists, neurologists, surgeons, and others required for treating conditions like cancer and neurological dysfunctions. The level of specialization, high-acuity services and multimodal treatment protocols under one roof attract patients and researchers to hospitals for personalized cell therapies. Moreover, hospitals encourage robust industry-academia partnerships and facilitate technology transfers crucial for advancing next-gen cell-based products.
Need a Different Region or Segment? Request Free Sample
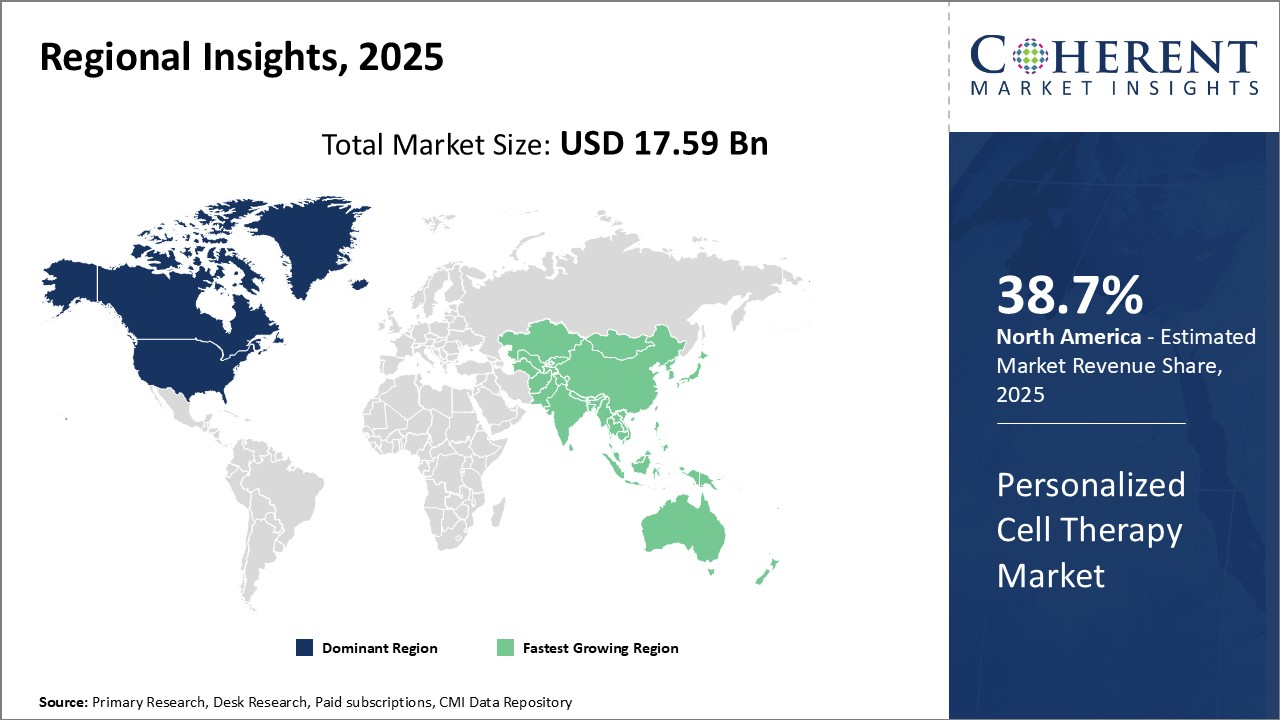
North America dominates the global personalized cell therapy market, with an estimated market share of 38.7% in 2026, due to significant industry presence and extensive research activities. American biotechnology and pharmaceutical companies have pioneered numerous cell therapy innovations over the past few decades. Major market players like Novartis, Gilead Sciences, and Juno Therapeutics have their headquarters in the U.S.
Moreover, large population size and higher healthcare expenditure in the U.S. translate to a sizable patient pool requiring cutting-edge treatments. Personalized cell therapies have found numerous clinical applications in oncology, cardiology, neurology and other therapeutic areas in the country. Support from the National Institutes of Health and the Food and Drug Administration has accelerated research and development as well as expedited approval processes for advanced therapies.
Asia Pacific is expected to be the fastest growing regional market for personalized cell therapies. Countries like China, Japan, South Korea and India are emerging as attractive destinations for outsourced clinical research and manufacturing capabilities. Rising affordability of novel drugs alongside growing medical infrastructure and awareness will fuel the mass adoption of personalized medicine in Asia in the near future.
Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 21.95 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 24.8% | 2033 Value Projection: | USD 103.51 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Thermo Fisher Scientific, Inc., Novartis AG, Gilead Sciences, Inc., Pfizer Inc., Amgen Inc., Bluebird Bio, Inc., Personalis, Inc., Adaptimmune Therapeutics PLC, BioNTech SE, Sangamo Therapeutics, Inc., Regeneron Pharmaceuticals, Inc., CRISPR Therapeutics AG, Caribou Biosciences, Inc., Mustang Bio, Inc., Fate Therapeutics, Inc., Atara Biotherapeutics, Inc., Holostem Terapie Avanzate S.r.l |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Global Personalized Cell Therapy Market involves medical treatments that use a patient's own cells to treat diseases like cancer. In cell therapy treatments, cells are removed from a patient and genetically modified outside the body to produce substances that boost, restore, or replace impaired immune system functions. The modified cells are then infused back into the patient to fight disease. The global market involves research on cell therapies tailored specifically for individual patients based on their genetic profiles and medical conditions.
Share
Share
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients