Tissue Plasminogen Activator Market is estimated to be valued at USD 3.00 Bn in 2026 and is expected to reach USD 4.34 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 5.4% from 2026 to 2033.
Analysts’ Views on Global Tissue Plasminogen Activator Market:
Robust pipeline of tissue plasminogen activator drugs and increasing initiatives and awareness programs related to ischemic stroke and myocardial infarction are expected to drive the global tissue plasminogen activator market growth over forecast period.
Figure 1. Global Tissue Plasminogen Activator Market Share (%), By Drug Type, 2026
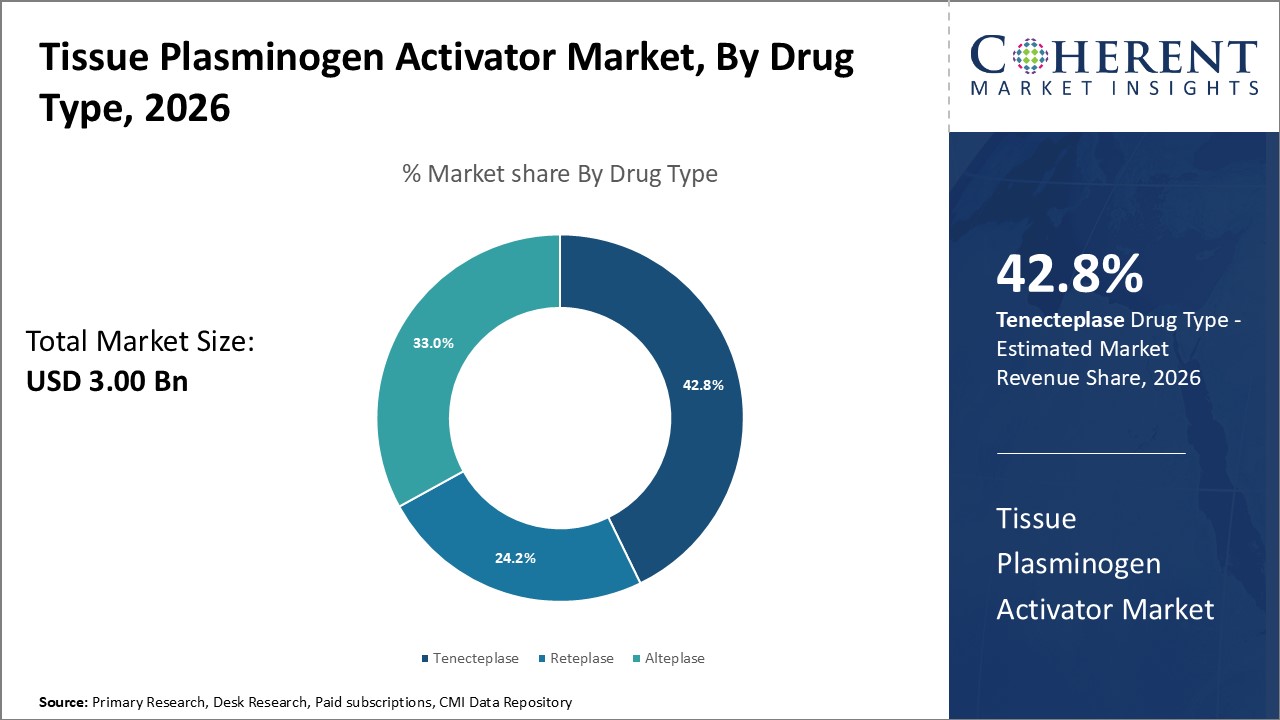
To learn more about this report, Request Free Sample
Global Tissue Plasminogen Activator Market – Drivers
Figure 2. Global Tissue Plasminogen Activator Market Share (%), By Region, 2026
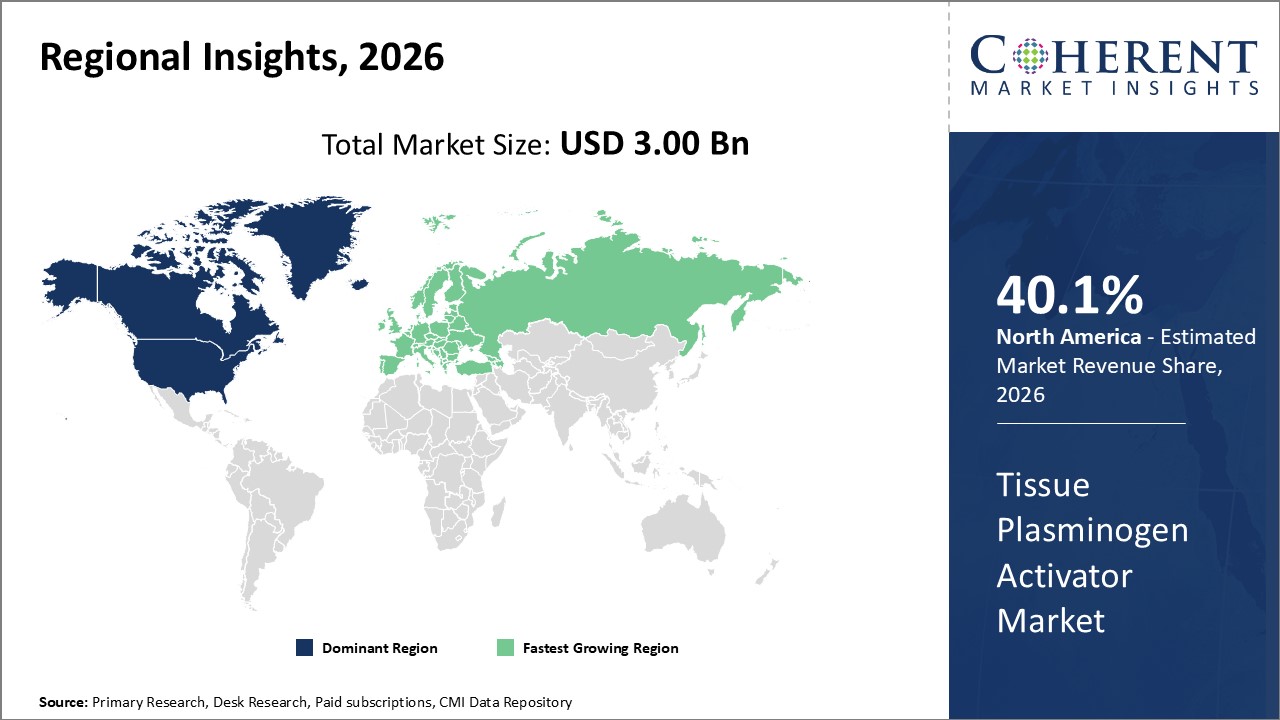
To learn more about this report, Request Free Sample
Global Tissue Plasminogen Activator Market- Regional Analysis
Among region, North America is estimated to hold a dominant position in the global tissue plasminogen activator market over the forecast period. North America is estimated to hold 40.1 % of the market share in 2026. The market is expected to witness significant growth in the near future due to rising product approvals by regulatory authorities in U.S. For instance, in June 2021, the U.S. Food and Drug Administration (FDA) had authorized or approved Ryplazim (plasminogen, human-tvmh) a drug developed by ProMetic Biotherapeutics Inc., a biopharmaceutical company for the treatment of individuals with plasminogen deficiency type 1, commonly known as hypoplasminogenemia, a disease that can impede normal tissue and organ function and can lead to blindness.
Tissue Plasminogen Activator Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 3.00 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.4% | 2033 Value Projection: | USD 4.34 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
F. Hoffmann-La Roche AG, Boehringer Ingelheim GmbH, CHIESI Farmaceutici S.p.A., Gennova Biopharmaceuticals Ltd, Abbott Laboratories, and Reliance Life Sciences |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Tissue Plasminogen Activator Market- Segmentation
Global tissue plasminogen activator market report is segmented into by drug type, by application, and by region.
Global Tissue Plasminogen Activator Market - Cross Sectional Analysis
Among application segment, ischemic stroke segment is expected to be dominant in the Europe region, owing to increasing prevalence of stroke. For instance, according to Age and Ageing journal, in March 2020, the number of strokes in the U.K. every year is expected to increase by 61%, while the number of stroke survivors is expected to double.
Global Tissue Plasminogen Activator Market: Key Developments
In July 2021, Boehringer Ingelheim GmbHThe drug demonstrated a promising safety and effectiveness profile in an interim review of the study, which has completed the exploratory open-label Phase IIb phase with 62 patients.
In May 2021, Biogen Inc., a pharmaceutical company that focuses on neuroscience drugs, and TMS Co., Ltd., a clinical stage biotechnology company, announced that Biogen Inc. exercised its option to acquire TMS-007, a clinical-stage drug for acute ischemic stroke, from TMS Co., Ltd. The acquisition of TMS-007 by Biogen Inc. was based on favorable findings from a Phase 2a research. TMS-007 is a small molecule plasminogen activator with a unique hypothesized mode of action linked to dissolving blood clots and perhaps preventing local inflammation at the site of thrombosis.
In February 2020, Reliance Life Sciences Private Limited, a research-driven organization, launched three biosimilars to the market: interferon beta, reteplase, and human chorionic gonadotropin. Reteplase is a purified version of human tissue plasminogen activator that is used to treat myocardial infarction, ischemic stroke, and pulmonary embolism in the event of an emergency.
Global Tissue Plasminogen Activator Market: Key Trends
Global Tissue Plasminogen Activator Market: Restraint
To counterbalance the restraint, key players should focus on receiving funds to increase their research and development activities.
Global Tissue Plasminogen Activator Market - Key Players
Major players operating in the global tissue plasminogen activator market include F. Hoffmann-La Roche AG, Boehringer Ingelheim GmbH, CHIESI Farmaceutici S.p.A., Gennova Biopharmaceuticals Ltd, Abbott Laboratories, and Reliance Life Sciences.
*Definition: TPA tissue plasminogen activator is produced by using recombinant technology techniques known as recombinant tissue plasminogen activator (rtPA). Specific rtPAs are tenecteplase, alteplase, and reteplase. These enzymes are used in clinical treatment of thrombotic stroke or embolic stroke. tPA is a drug that dissolves blood clots. It is a thrombolytic agent which can be administrated into the veins.
Share
Share
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients