Degenerative disc disease is a spinal condition caused by the breakdown of intervertebral discs. The symptoms observed for degenerative disc disease are tingling or numbness in legs or difficulty in walking. This disease is diagnosed with physical examination, through X-rays or magnetic resonance imaging (MRI). The degenerative disc disease is most commonly seen in geriatric population and has caused lower back pain and neck pain in adults.
The U.S. degenerative disc disease treatment market is estimated to have around 2.644 Mn patients suffering from degenerative disc disease (DDD) in 2021 and is expected to exhibit a CAGR of 5.9% during the forecast period (2021-2028).
Figure 1.U.S. Degenerative Disc Disease Treatment Market Share (%) in Terms of Value, By Treatment 2021
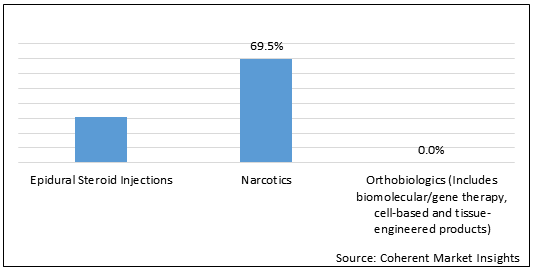
To learn more about this report, Request Free Sample
Increasing cause of lower back pain & neck pain in adults and increasing geriatric population is expected to drive the market growth during the forecast period.
Increasing cause of lower back pain & neck pain in adults and increasing geriatric population in the U.S. is expected to drive the growth of the degenerative disc disease treatment market over the forecast period. For instance, according to the article published in the National Center of Biotechnology Information in May 2017, among adults 60% to 80% would experience back pain and 20% to 70% would experience neck pain that would interfere with their daily activities during their lifetime in the U.S.
U.S. Degenerative Disc Disease Treatment Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2020 | Market Size in 2021: | US$ 2.644 Mn |
| Historical Data for: | 2017 to 2020 | Forecast Period: | 2021 to 2028 |
| Forecast Period 2021 to 2028 CAGR: | 5.9% | 2028 Value Projection: | US$ 3.958 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AnGes Inc., Ankasa Regenerative Therapeutics, Inc., BIOPHARM GMBH, BioRestorative Therapies Inc., Biosplice Therapeutics, Inc., Bone Therapeutics SA, Cerapedics Inc, CO.DON, Creative Medical Technology Holdings, Inc., DiscGenics Inc., Eli Lilly and Company, Ferring Pharmaceuticals, FibroGenesis, Geistlich Pharma AG, Harbor View Medical Services, PC, Intralink-Spine, Inc., Isto Biologics, Johnson & Johnson, Kolon TissueGene Inc., Kunovus, Lineage Cell Therapeutics, Inc., Mesoblast, NC Biomatrix BV, Notogen Inc., Pfizer, Inc., R3 Stem Cell, Smith & Nephew, Spine BioPharma, Stayble Therapeutics AB, US Stem Cell Inc., and VIVEX Biologics, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Figure 2 .U.S. Degenerative Disc Disease Treatment Market Share (%), By Disease Indication, 2021
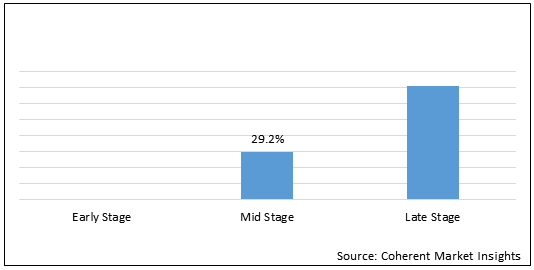
To learn more about this report, Request Free Sample
Increasing research and development for the treatment of degenerative disc disease is expected to drive the market growth during the forecast period.
Key players operating in the market are focusing on development of novel therapeutics for the treatment of degenerative disc disease which is expected to offer a lucrative opportunity for the market players to launch their novel products in the market. For instance, in March 2020, DiscGenics, Inc. announced that it had completed enrolment in its Phase I/II first in human U.S. clinical study of IDCT, an allogeneic, injectable Discogenic Cell therapy for degenerative disc disease (DDD)
U.S. Degenerative Disc Disease Treatment Market– Impact of Coronavirus (COVID-19) Pandemic
Since the COVID-19 virus outbreak in December 2019, the disease has spread to over 100 countries across the globe with the World Health Organization declaring it as a public health emergency. According to the World Health Organization’s report, the manifestation of Coronavirus (COVID-19) has resulted in more than 33.6 million infected individuals in the U.S. as of July 16, 2021. COVID-19 can affect the economy in three main ways; by directly affecting production and demand, by creating disruptions in distribution channels, and through its financial impact on firms and financial markets. For instance, in the U.S., according to the U.S. Consumer Spending Report, there has been a huge plunge in consumer spending as people stayed home and avoided shopping, traveling or gathering in crowds and caused the economy to sink at an estimated annual rate of 32% in the April-June 2020 quarter. The direct impact of COVID-19 pandemic related to possible delays of surgical care in infected patients seems relatively small in the grand scheme. Overall, the indirect effects of the COVID-19 pandemic upon spinal care are noticeable. Such a deleterious impact can be traced to several factors such as the reallocation of hospital resources (such as nursing staff and ICU beds) to the care of patients with the Coronavirus as well as cancellation of elective surgeries and office visits in order to reduce the chances of disease spread. The combination of both of these has led to sudden and drastic changes in the daily routine of not only spine surgeons but also of other professionals involved in spine care, such as pain management and rehabilitation physicians, physical therapists, and occupational therapists. Such effects have been even more pronounced after several U.S. states and counties decreed ‘shelter-in-place’ policies. The COVID-19 pandemic has also greatly affected the daily operations of several healthcare industries. Cancel and delay in surgery tend to percolate throughout the whole healthcare environment, with negative consequences upon ongoing clinical trials mainly related to cessation of patient enrollment as well as cancelled follow-up visits, imaging, and laboratory tests. All of these tend to extend the required time for the completion of clinical studies, data analysis and the U.S. Food and Drug Administration approval, with increased total costs for bringing such new technologies and drugs to the market for the treatment of degenerative disc disease.
U.S. Degenerative Disc Disease Treatment Market: Restraint
The major factors that may hinder growth of the U.S. degenerative disc disease treatment market include potential side effects of drugs used in the treatment of degenerative disc disease and high cost of surgery. For instance, according to the article published in the Spine Health in 2015, the cervical artificial disc replacement surgery cost around US$ 35,000 in the U.S. Moreover, according to the New Choice Health, Inc., a medical adviser company, the average cost of disc replacement surgery procedures ranges from US$ 16,700 – US$ 45, 800 in the U.S.
Key Players
Major players operating in the U.S. degenerative disc disease treatment market include AnGes Inc., Ankasa Regenerative Therapeutics, Inc., BIOPHARM GMBH, BioRestorative Therapies Inc., Biosplice Therapeutics, Inc., Bone Therapeutics SA, Cerapedics Inc, CO.DON, Creative Medical Technology Holdings, Inc., DiscGenics Inc., Eli Lilly and Company, Ferring Pharmaceuticals, FibroGenesis, Geistlich Pharma AG, Harbor View Medical Services, PC, Intralink-Spine, Inc., Isto Biologics, Johnson & Johnson, Kolon TissueGene Inc., Kunovus, Lineage Cell Therapeutics, Inc., Mesoblast, NC Biomatrix BV, Notogen Inc., Pfizer, Inc., R3 Stem Cell, Smith & Nephew, Spine BioPharma, Stayble Therapeutics AB, US Stem Cell Inc., and VIVEX Biologics, Inc.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients