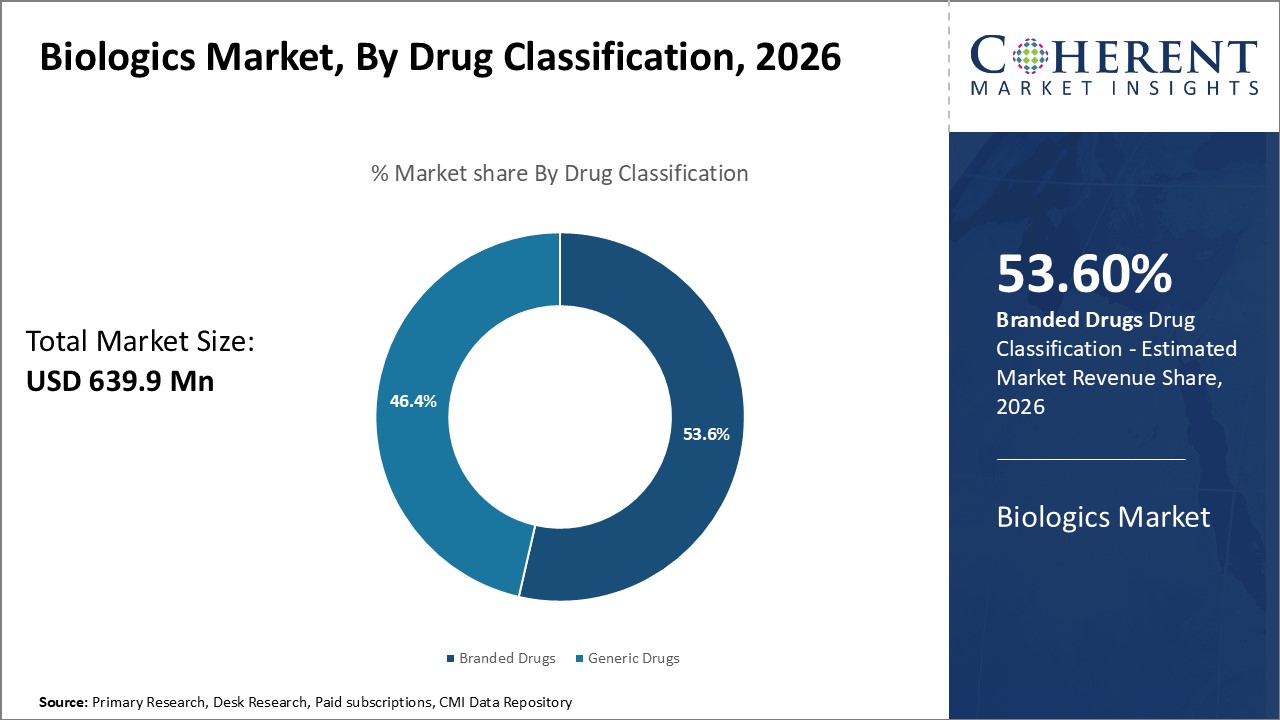
The biologics market is estimated to be valued at USD 639.9 Mn in 2026 and is expected to reach USD 1,311.8 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 10.8% from 2026 to 2033.
To learn more about this report, Request Free Sample
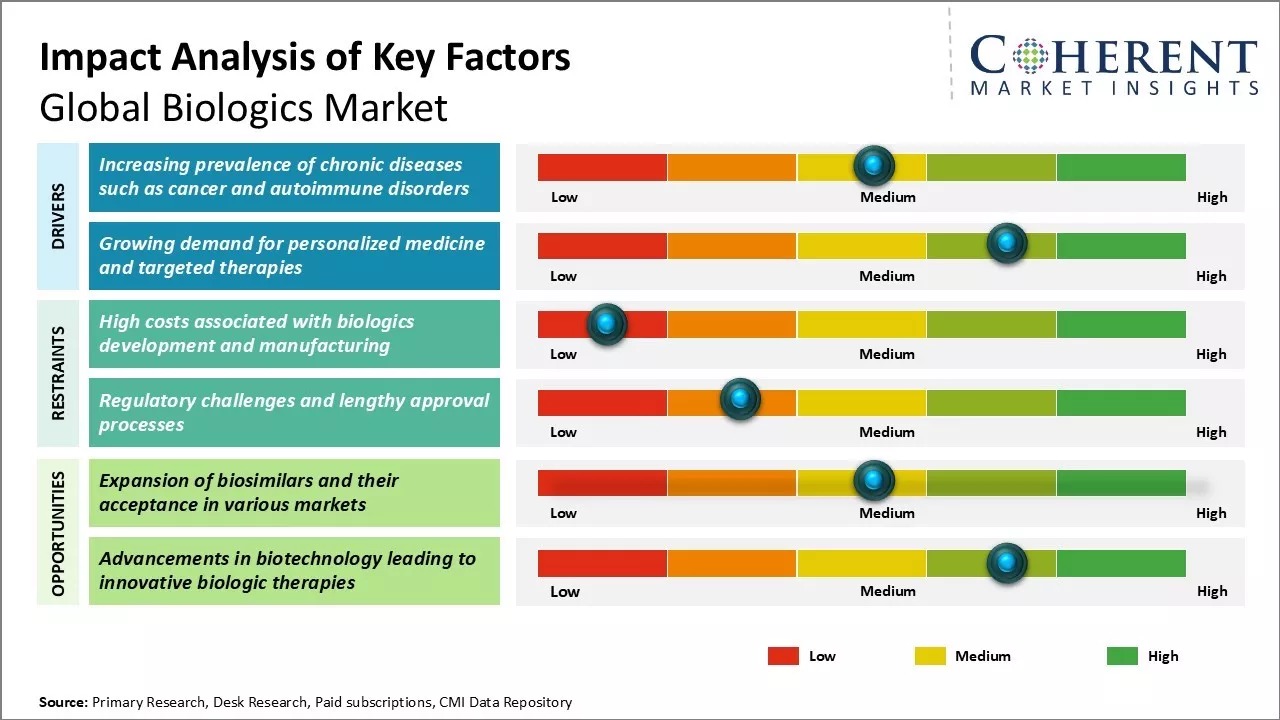
This growth is primarily driven by the increasing demand for monoclonal antibodies and recombinant proteins, which are crucial in the treatment of chronic diseases such as cancer, diabetes, and cardiovascular diseases. As the prevalence of these diseases continues to rise globally, the need for advanced biologic therapies that offer more targeted and effective treatment options becomes even more critical.
However, high costs of developing and manufacturing biologics remains a major challenge for widespread adoption.
|
Current Events |
Description and its impact |
|
Regulatory Shifts in Biosimilar Development |
|
|
Geopolitical Export Controls and Trade Dynamics |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The increasing prevalence of chronic diseases globally is a significant factor propelling the growth of the biologics market. Chronic illnesses such as diabetes, rheumatoid arthritis, cancer, and autoimmune disorders have shown a marked rise over the past decade, with complex pathophysiologies that often require advanced therapeutic solutions. Biologics, which include monoclonal antibodies, recombinant proteins, and gene therapies, offer targeted treatment options that can modulate disease-specific pathways more effectively than traditional small-molecule drugs.
For instance, according to a 2025 National Institute for Health Care Management report, chronic diseases affected approximately 60% of Americans, with many individuals suffering from more than one chronic illness.
In terms of product, the monoclonal antibodies segment is estimated to contribute the highest market share of 33.6% in 2026, owing to the increasing prevalence of chronic diseases globally.
Monoclonal antibodies have revolutionized the treatment of various conditions such as cancer, autoimmune disorders, infectious diseases, and others. Their high specificity allows them to precisely target molecular abnormalities, driving disease processes. The capability of monoclonal antibodies to modulate immune responses makes them very effective for conditions where traditional small molecule drugs have limitations.
To learn more about this report, Request Free Sample
In terms of drug classification, the branded drugs segment is estimated to contribute the highest market share of 53.6% in 2026, owing to strong marketing by pharmaceutical companies and extensive clinical evidence for branded products.
Large pharmaceutical corporations invest heavily in promotional campaigns and branding activities to generate greater awareness about their products among physicians and patients. Clinicians tend to have more confidence in prescribing branded drugs due to availability of data from extensive clinical trials detailing their safety, efficacy and dosing.
In terms of route of administration, the oral segment is estimated to contribute the highest market share of 39.6% in 2026, owing to the major benefits it offers over parenteral drugs especially for chronic conditions.
The oral route provides unmatched convenience as pills can be self-administered at home without any medical supervision or health staff involvement. This improves treatment accessibility. Oral drugs face less stigmatization compared to injections making them socially discreet. Their self-administrable nature increases adherence to medication schedules.
Artificial Intelligence (AI) is reshaping the biologics market by accelerating innovation, optimizing drug development, and enhancing patient outcomes. Its integration within the industry is not just a technological upgrade but a paradigm shift influencing every stage of biologic product lifecycles.
To learn more about this report, Request Free Sample
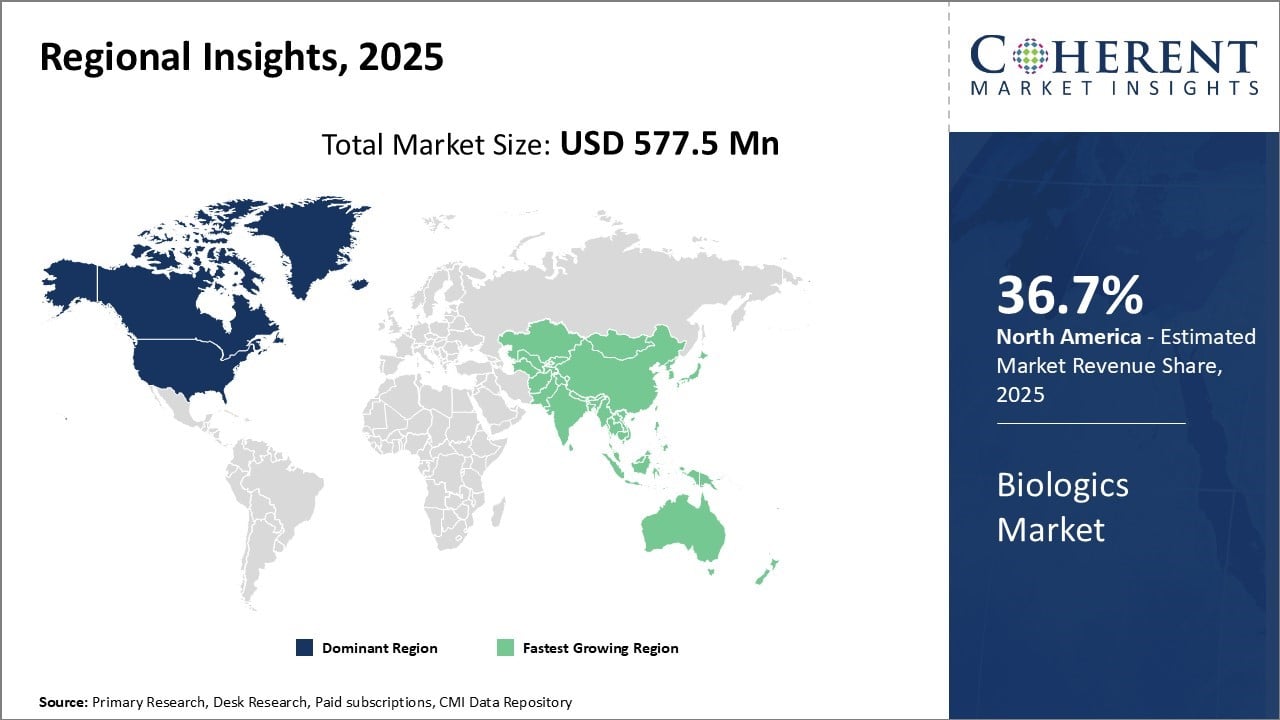
North America dominates the biologics industry with an estimated market share of 36.7% in 2026, due to strong presence of global pharmaceutical players like AbbVie Inc. and a well-established healthcare infrastructure. Major markets like the U.S. have fostered innovations in biologics through favourable research policies like the 21st Century Cures Act and financial support for R&D activities.
For instance, in 2021, biosimilar approvals and product launches contributed to approximately USD 7 billion in annual savings—bringing cumulative U.S. savings to over USD 13 billion since 2015. Under current adoption projections, biosimilars could deliver more than USD 38 billion in savings from 2021–2026.
Asia Pacific region exhibits the fastest growth due to rising healthcare expenditures, growing disease burden, and increasing focus on ensuring access to advanced treatment options. Counties like China and India are emerging as global biopharma hubs leveraging their large generics manufacturing base and a skilled workforce.
For instance, India celebrated approximately 25 years in the biosimilar space in 2026, having approved over 95 biosimilars to date—establishing itself as a major global player in biologics and biosimilars manufacturing.
The U.S. biologics sector is powered by robust research & development (R&D) infrastructure, significant public funding (e.g. the Biologics Price Competition and Innovation Act), and leading pharmaceutical firms investing heavily in novel biologics, cell and gene therapies—reinforced by a supportive regulatory framework that speeds approvals and fosters innovation.
China is rapidly emerging as a powerhouse in biologics, fueled by proactive government initiatives under ‘Made in China 2026’ and the 14th Five-Year Plan, fast-tracked regulatory approvals (cutting average approval times to approximately 90 days), and increasing biotech R&D spending (over USD 15 billion in 2022)—all catalyzing domestic and global growth.
India is expanding in biologics via streamlined regulatory pathways, targeted government schemes (e.g. Bio-E3, PLI incentives), rising chronic disease prevalence, and strong biosimilar manufacturing capabilities (driven by Biocon, Dr. Reddy’s, Intas), supported by increasing R&D investment—from US USD 1.3 billion in 2020 to approximately USD 5 billion by 2026.
Germany’s biologics sector is supported by a high chronic disease burden—nearly 493,000 new cancer cases annually—and substantial biotech R&D investment equivalent to approximately USD 10.6–USD 11.3 billion per year. Major firms like Bayer, Boehringer Ingelheim, and Merck KGaA, bolstered by strong public–private innovation ecosystems, continue to drive growth in the biopharmaceutical space.
|
Brand Names of Approved Gene or Cell Therapies |
Price Range (USD) |
|
Kymriah (Tisagenlecleucel)- Leukemia, lymphoma |
375,000 - 475,000, Single infusion gene therapy |
|
Yescarta (Axicabtagene ciloleucel) - Large B-cell lymphoma |
Approximately 400,000, CAR-T cell therapy requiring specialized HCP support |
|
Zolgensma (Onasemnogene abeparvovec) Spinal muscular atrophy |
Approximately 2.1 million (one-time), One-time gene therapy for pediatric patients |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 639.9 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.8% | 2033 Value Projection: | USD 1,311.8 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
F. Hoffmann-La Roche Ltd., AbbVie Inc., Amgen Inc., Johnson & Johnson Services, Inc., Merck & Co., Inc., Pfizer Inc., Sanofi S.A., Gilead Sciences, Inc., Novartis AG, Bristol-Myers Squibb Company, Regeneron Pharmaceuticals, Inc., Takeda Pharmaceutical Company Limited, Biogen Inc., Eli Lilly and Company, and Celltrion Healthcare Co., Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: The biologics market refers to the global industry involved in the development, manufacturing, and commercialization of biologic therapies—medicines derived from living organisms or their cells, including monoclonal antibodies, vaccines, gene and cell therapies, and therapeutic proteins—used to treat a wide range of diseases such as cancer, autoimmune disorders, and infectious diseases.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients