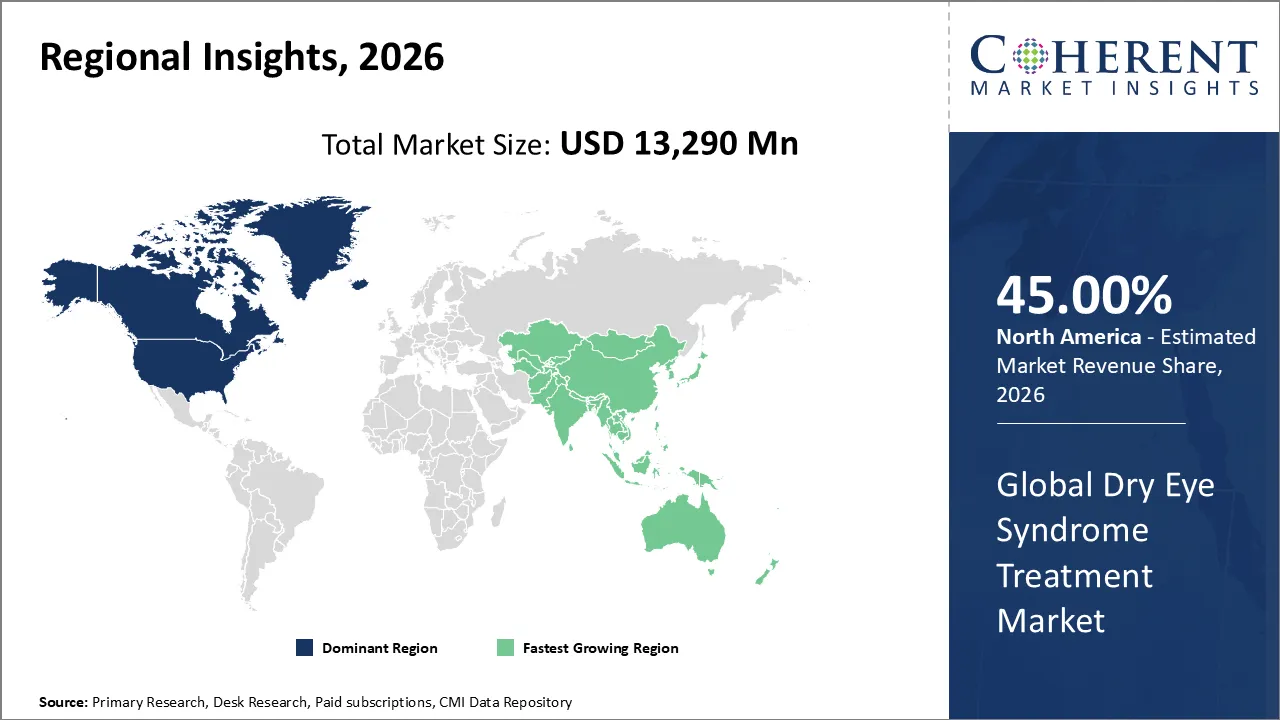
Dry eye syndrome treatment market is estimated to be valued at USD 13,290 Mn in 2026 and is expected to reach USD 38,420 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 16.4% from 2026 to 2033.
The growing rate of prevalence of dry eye syndrome, especially in the aging population and digital screen viewers, is a major factor fueling market demand for dry eye syndrome treatment products. The rising number of digital device users who are watching television and using computers and other digital devices is predominantly responsible for causing more cases of eye strain and imbalance in the tear film. This in turn is fueling the market for dry eye syndrome products. In addition, increased awareness about early treatment and management of chronic conditions is also a factor.
|
Current Events |
Description and Its impact |
|
Regulatory Framework Evolution and Drug Approvals |
|
|
Technological Innovation and Treatment Advancement |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
By product type, the artificial tears segment is expected to capture the highest market share of 61% in 2026, owing to its first-line treatment for mild to moderate dry eye syndrome. Artificial tears restore tear film stability and ocular discomfort due to environmental factors and time spent in front of digital screens.
For instance, in February 2025, Alcon announced the U.S. launch of SYSTANE PRO Preservative-Free (PF), a new innovative artificial tear solution, which is designed to deliver long-lasting relief by hydrating, restoring, and protecting all forms of dry eyes through its unique triple action, preservative-free formula.
Based on the distribution channel, retail pharmacies are expected to provide a 58% share in 2026, owing to their easy accessibility and presence almost everywhere; both prescription and over-the-counter dry eye treatment products are readily available. A retail pharmacy will be the point of purchase for patients after consulting with an ophthalmologist, who can offer expert advice on what should be purchased and whose products would be immediately available.
For instance, in March 2025, Harrow Therapeutics launched the VEVYE® Access for All program, which is focused on reducing barriers to treatment for people suffering from dry eye disease in the United States. This is done by making its cyclosporine solution available at retail pharmacies.
To learn more about this report, Request Free Sample
North America is likely to dominate the dry eye syndrome treatment market in 2026, with an estimated share of around 45% of the overall market. The leadership position of the region in the market can be attributed to the high prevalence of dry eye disease, either directly or indirectly, due to the aging population and due to prolonged use of digital screens. Besides, strong presence of leading pharmaceutical and ophthalmic companies, early adoption of advanced treatment options, and favorable reimbursement policies have resulted in its growth.
The Asia Pacific region is expected to witness the fastest growth in the dry eye syndrome treatment market during the forecast period. High growth in the Asia Pacific region is primarily due to the increasing screen time, growing awareness regarding eye care, rise in the middle-class population, and improved healthcare in countries such as China, India, Japan, and South Korea.
For instance, in January 2025, Santen Pharmaceutical launched a new preservative‑free artificial tear formulation in China, designed specifically for patients with moderate to severe dry eye disease.
The U.S. dry eye syndrome treatment market is rising significantly, driven by the increasing prevalence of digital eye strain, the aging population, and the pre-emptive adoption of advanced treatment methods. The rise in overall patient and practitioner awareness levels of dry eye syndrome treatment is fueling routine examinations, which is further boosting the overall treatment rates.
For instance, In May 2025, the U.S. Food and Drug Administration (FDA) approved Alcon’s Tryptyr, a novel eye drop treatment that stimulates natural tear production by activating corneal sensory nerves
India’s dry eye syndrome treatment market is growing at a rapid rate due to the increasing trend of screen usage, urban populations, and the health of the eyes. With greater awareness of the availability of ophthalmological care and retail pharmacy outlets, the availability of treatment is increasing across the country, especially in Tier 1 and 2 cities.
For instance, in May 2024, Entod Pharmaceuticals launched vitamin D3-enriched lubricating eye drops in India, a new vitamin-enriched product to cater to ophthalmic care, thus ensuring targeted relief for patients experiencing dry eyes.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 13,290 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 16.4% | 2033 Value Projection: | USD 38,420 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AbbVie Inc., Novartis AG, Bausch Health Companies Inc., Johnson & Johnson, Inc., I-MED Pharma inc., Santen Pharmaceutical Co., Ltd, AFT Pharmaceuticals, Novaliq GmbH, Otsuka Pharmaceutical Co., Ltd., Sun Pharmaceutical Industries, Inc., Teva Pharmaceutical Industries Ltd., Mitotech, SA, Viatris Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
One of the main factors propelling the market's expansion is the rising incidence of dry eye syndrome, especially among the elderly and those who use digital devices for extended periods of time. A rise in cases has been caused by increased screen time, environmental factors, and changes in lifestyle, which has increased demand for prescription drugs, artificial tears, and other therapeutic solutions. Adoption of dry eye treatments is further strengthened by growing awareness of the importance of early diagnosis and management of chronic eye conditions.
The market also offers growth opportunities through the introduction of innovative products and treatment modalities. The development of preservative-free and lipid-based artificial tears, nanomicellar drug delivery systems, and new anti-inflammatory agents like cyclosporine and lifitegrast can capture new patients looking for step-change improvement in product benefits and comfort profiles. Device-based treatment modalities, punctal plugs, thermal pulsation devices, and AI-based diagnostics also provide growth opportunities and differentiation for new and existing customers across retail and healthcare settings.
The dry eye syndrome treatment market is entering a phase of genuine clinical maturation rather than just incremental product expansion. What differentiates the current environment from the past decade is the realignment of treatment pathways around evidence‑based care rather than symptomatic relief alone. For example, adoption data from eye care clinics in North America shows that preservative‑free artificial tears now account for over 60% of product recommendations, reflecting clinicians’ growing preference for formulations backed by real‑world tolerability data.
Geographically, the divergence in treatment sophistication is stark: North America and Europe are pushing biologic and neurostimulatory approaches into mainstream practice, while Asia Pacific markets are leapfrogging older therapies, adopting nanomicellar delivery systems and device‑aided treatments ahead of some Western counterparts, driven by aggressive clinical adoption in China and Japan. This isn’t just regional growth; it’s a strategic rebalancing of therapeutic hierarchies.
Ultimately, the market is shedding its identity as a collection of sundry lubricants and becoming a disease‑centric therapeutic category. Success in this space will increasingly hinge on clinical validation, differentiated mechanisms of action, and measurable patient outcomes — not merely expanded product portfolios or broader distribution alone.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients